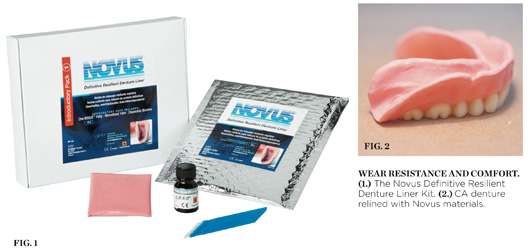
Novus Definitive Resilient Denture Liner
Proprietary technology promotes healthy tissue and patient compliance.
Novus® Definitive Resilient Denture Liner has been designed and formulated to remain permanently resilient for the life of the denture yet firm enough (with a durometer of up to 45) to ensure proper wear resistance and patient comfort. Novus can solve the most difficult denture cases involving retention, fit, and function and will dramatically reduce/eliminate sore spots, knife-edge ridges, and other problem areas often associated with patient discomfort. With Novus’s Polyphosphazene Technology, there are no hormonal or carcinogenic issues to worry about, and incorporating the product into the denture fabrication protocol will dramatically aid in the minimizing and elimination of tissue irritation. This benefit alone will work to promote more healthy tissue beneath the denture and better overall patient compliance and hygiene practices.
This product also features exception-al “problem-solving” characteristics for a dentist who is working with denture patients and has a modest dental laboratory on site. Specifically, it facilitates case scenarios such as the “Late Friday afternoon walk-in patient.” Most clinicians are all too familiar with the patient who is heading out of town for the weekend, or going on an extended vacation, and would like to alleviate their denture pain/irritation before leaving. Because most dental laboratories are not available on such late notice, this patient request would al-most be impossible (with previous denture reline/adjustment materials); however, with Novus, these cases can now be addressed with minimal equipment and/or material needs. By following a simple, routine series of steps, a dentist can now provide his or her patients with a last-minute denture reline that may perform better than any denture reline application received by the patient in the past (Figure 1).
A simplified review of these steps are as follows. Remove a portion of the old denture’s base material to allow for the desired thickness of Novus. Take an impression of the denture and invest it into a conventional investment flask. Apply a thin layer of fresh denture resin or Jet Novus Bond into the impression. Place the flask (with both denture and impression together) into a press for trial packing. Separate the flask and clean out any excess impression material. Place the Novus material onto the denture and put the flask back together. Process the flask following manufacturer’s directions. Separate the flask, remove the denture, and clean off any flash/excess material. Deliver the finished/relined denture to the patient (Figure 2).
Novus is passive to fungus overgrowth, requiring no periodic antimicrobial treatments often associated with silicone/urethane denture materials, which may exhibit fungus growth in a short period of time. This unique denture lining material will add to the health and comfort of the patient because it is non-porous and will not retain liquids, will resist odor-causing agents, and will prevent unsightly staining for the life of the denture. It does not harden like typical plasticized acrylics and will not require the periodic resurface coating that is often required with other conventional materials being used today. Additionally, Novus will not elicit “spongy” or “flexible” properties that give the denture wearer a feeling of constant movement or slippage; however, it will provide unsurpassed “shock absorption” during patient mastication and will require fewer adjustments over the life of the dental appliance.
For the dentist and laboratory technician, working with Novus is easy because it is easy to handle and can be ground, adjusted, and polished with standard rotary instruments. It is compatible with other acrylic materials/techniques and will bond both chemically and mechanically to other materials resulting in a permanent bond that ensures both denture performance and wear resistance. Novus can be processed, packed, and cured using conventional laboratory techniques, so it does not require the added costs of new equipment, software, and/or other time-consuming methods. Its properties allow it to be molded around overdenture abutments or implant heads/bars with deep undercuts to provide retention for removable prosthetic attachments. It is radiopaque and can be properly identified if any parts or pieces are accidentally ingested or inhaled by the patient. The product has been manufactured to provide unlimited shelf life if refrigerated and is available in three package sizes (5, 15, or 45 reline procedures) to accommodate any case volume.
Acknowledgement
This article was written by James Jesse, DDS, a member of Catapult Elite, a consortium of key opinion leaders in the dental industry.
For more information, contact:
Lang Dental Manufacturing Company, Inc.
Phone: 800-222-5264
Web: www.langdental.com
Disclaimer
The preceding material was provided by the manufacturer. The statements and opinions contained therein are solely those of the manufacturer and not of the editors, publisher, or the Editorial Board of Inside Dentistry.