Timing of Guided Bone Regeneration: A Case Report
Careful diagnosis and timing boost the chances for success of restoration with GBR prior to or in combination with implant placement.
By Michael Sonick, DMD | Debby Hwang, DMD
The introduction and refinement of guided bone regeneration (GBR) allowed clinicians to generate more stable, cosmetically appealing implant-supported prostheses. Although ridge morphology is no longer the sole determinant of implant feasibility due to advancements in bone grafting, it retains a tremendously profound influence on treatment sequencing and end-stage survival. Still accurate is the notion that the more intact the patient’s residual ridge is, the better his or her regenerative and restorative prognosis. Conscientious planning and clinical fortitude, however, may transcend initially inauspicious bone morphology to some extent. When carefully considered and timed, GBR is highly successful, producing new functional bone that anchors implants as well as solid native osseous tissue does. This article reviews the defect types amenable to GBR and illustrates the decision-making process and surgical techniques used to reach a satisfactory implant restoration.
The Therapeutic Phase
The formative stage of dental treatment, related to implants or otherwise, is unquestionably the most crucial therapeutic phase. Planning a case is not always straightforward or logistical, and the best approach may not be the immediately intuited or most expedient one. In spite of ideally executed surgery, a sloppy, rushed, or poorly communicated treatment design exponentially multiplies the risk of failure and patient dissatisfaction. Consider this idea in the context of implant rehabilitation in the esthetic zone, which involves substantial time, physical, emotional, and monetary investments for both the patient and doctor. In order to reach a pleasing, enduring result, the orchestration of cosmetic implant dentistry must be fastidious.
Each step in the process focuses on creating and maintaining enough hard tissue and mucosa for an ideal prosthesis. Because the tooth-to-alveolar bone volume ratio is relatively large in the anterior maxilla, there may be inadequate residual ridge left after dental extraction to support an implant fixture, let alone any surrounding soft tissue. Remodeling creates an average loss of 3 mm to 6 mm horizontally and 1 mm to 2 mm vertically after 4 months, which corresponds roughly to the conventional implantation time.1,2 Notably, the buccal plate is two to three times thinner than its lingual counterpart and, thus, undergoes nearly twice as much lateral reduction as a vertical deficit 2 mm greater than the palatal side.1,3,4 The cardinal tenet of tooth removal, then, is to minimize bone loss. Curtailing post-extraction resorption involves an atraumatic surgical technique along with socket preservation, or placement of bone graft material into the void. Socket preservation cuts horizontal deterioration by at least half, reducing the ridge by 1 mm to 1.5 mm instead of the 3 mm to 6 mm seen naturally, and it may prevent vertical resorption entirely.2,5,6
Mere socket preservation, however, may not be enough. True implant site development, particularly in the current state of the field, involves the growth of ridge dimensions beyond those that existed originally. The presence of an implant does not necessarily abate bone loss; indeed, biologic width reformation, occlusal forces, oral hygiene, and medical conditions contribute to osseous remodeling around the titanium fixture. The savvy clinical team foresees the postsurgical resorptive course and compensates by attempting to build extra ridge wherever and whenever possible. For certain defect types, guided bone regeneration (GBR) using a membrane alone or in conjunction with a bone graft accomplishes this reasonably well, achieving success rates above 90% in studies up to 5 years, and augments by 3 mm to 5 mm supracrestally.7,8 Although it relieves supracrestal deficiencies, GBR best resolves space-making defects, which occur within an envelope of bone.9,10 The intact surrounding walls provide viable cells, blood supply, containment for graft particles, and a buttress to support the regenerative barrier and prevent collapse. Predictability of augmentation is directly proportional to the amount of bone that is enclosing the space. Of course, the gross size of the defect itself is also a critical factor. It is evident that the smaller the gap is that needs to be bridged by new tissue, the easier it is to fill. A defect spanning a single tooth or with mild horizontal bone loss has a very good prognosis, while one spanning two teeth or with moderate horizontal or mild vertical deficiency garners a good prognosis. The prognosis of a three-tooth encompassing, moderately resorbed edentulous ridge drops to average; restoration of a four-tooth or severely vertically compromised defect is unlikely. Thus, indications for GBR consequently include mild horizontal and mild vertical defects (less than 3 mm) and small dehiscence/fenestration defects, the latter of which achieve consistent coverage over 90%.11
A more specific treatment protocol is demonstrated here. In the case of a fenestration or dehiscence defect that would allow an implant to lie within the bony envelope, simultaneous implant placement and GBR should commence. Similarly, the same treatment is advised for a horizontal deficiency that would expose less than 50% of the implant diameter and leaves the implant within the bony envelope. If, however, a portion of the implant would penetrate beyond the envelope of bone via a dehiscence/fenestration defect, the restorative position must be evaluated. A favorable position suggests concomitant fixture placement and regeneration. On the other hand, a less-than-ideal position or horizontal defect that exposes more than 50% of the fixture diameter or leaves a portion uncovered beyond the osseous walls disqualifies implantation, instead designating GBR and an adequate healing period prior to placement. Vertical defects, notoriously difficult to resolve consistently, may benefit from a tenting device placed at the time of GBR—such a mechanism may be a screw, pin, or, in cases when less than 3 mm of height is needed, even the implant itself.
Knowing these limitations when preparing an esthetic implant rehabilitation helps to stage the case properly. After maturation, post-atraumatic tooth extraction, and socket preservation (customarily a course of 3 months), the dentist must methodically evaluate the residual ridge via palpation, bone sounding, conventional radiographic study, and possibly CT scan analysis. An obvious defect precludes implantation and instead calls for regenerative therapy first, followed by later placement (ie, pre-implant GBR). A more subtle, practically unnoticeable defect justifies concomitant fixture placement and GBR (ie, peri-implant GBR) if the following three prerequisites are met:
• Primary implant stability may be attained.
• Ideal positioning for restoration is possible.
• Deficiency is space-making and self-limiting.
As outlined above, defects treatable by simultaneous implantation and GBR include dehiscence/fenestrations confirmed within the envelope of bone as well as circumferential defects from incomplete healing after extraction or diameter discrepancies between the osteotomy site/socket and fixture. Minor dehiscence/fenestration notwithstanding, any vertical defect favors a staged grafting plan as do other deficits that are not space-making. Certainly, it is important to note that if the surgeon harbors any doubt regarding the feasibility of peri-implant GBR or if there are high cosmetic demands, he or she may opt for GBR and delay implant placement until after site maturation. Executed judiciously, GBR creates a ridge that can achieve an implant survival rate equivalent to that of native bone.
The following case illustrates decision-making with respect to the timing of GBR and implant insertion. It involves a maxillary anterior canine, tooth No. 6, and therefore requires an elevated level of esthetics and function.
Case: Pre-Implant Guided Bone Regeneration
Patient History
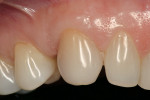
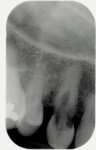
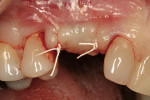
A medically and periodontally stable 48-year-old, non-smoking woman presented with a previously traumatized tooth No. 6, failing due to internal resorption (Figure 1 and Figure 2).
Treatment Sequence
• Extraction and socket preservation of tooth No. 6
• 3-month healing period
• GBR at No. 6 site
• 6-month healing period
• Implant placement in No. 6 site
• 3-month healing period
• Guided soft-tissue growth
• 1-month healing period
• Final implant No. 6 crown restoration
Extraction and Socket Preservation
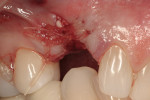
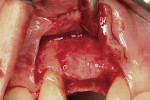
After oral sedation with 0.25 mg triazolam 1 hour prior to surgery and local anesthetic induction using 2% lidocaine with 1:100,000 epinephrine and 0.5% bupivacaine with 1:200,000 epinephrine, a sulcular incision was placed circumferentially around tooth No. 6. Using a piezoelectric unit, No. 6 was atraumatically extracted. Degranulation of the socket with a carbide finishing bur and irrigation with 0.125 mg chlorhexidine occurred, followed by decortication of the socket with a round bur to induce bleeding points. Freeze-dried bone allograft (FDBA) was placed into the alveolus and covered by an absorbable collagen plug (in this case, CollaPlug®, Integra LifeSciences, www.integra-ls.com). The site was closed secondarily with 5-0 chromic gut suture (Figure 3).
Ridge Assessment Post-Extraction
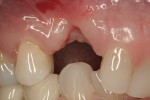
Noticeable vertical and mild horizontal resorption was detected 3 months after extraction (Figure 4). Due to the supracrestal nature of the ridge defect, it was decided to proceed first with GBR and delay implant placement.
Pre-Implant GBR
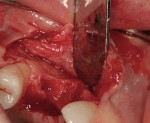
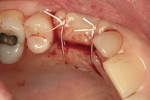
After oral sedation with 0.25 mg triazolam 1 hour prior to surgery and local anesthetic induction using 2% lidocaine with 1:100,000 epinephrine and 0.5% bupivacaine with 1:200,000 epinephrine, a palatally oriented horizontal incision was made in the edentulous No. 6 ridge with a sulcular extension to the mesio-buccal aspect of tooth No. 7 and disto-buccal aspect of tooth No. 5. To increase visualization, a vertical incision with a small right angle coronal modification was placed at the disto-buccal line angle of No. 5; a straight vertical cut followed at the mesio-buccal line angle of No. 7. A full-thickness flap was elevated past the mucogingival junction, and periodontal scoring took place near the base of the flap to facilitate the coronal advancement necessary for primary closure. Degranulation of the residual ridge using a pear-shaped carbide finishing bur and Prichard curette proceeded. Mild vertical and horizontal defects (≤ 3 mm) were detected (Figure 5 and Figure 6).
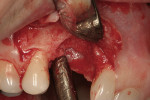
Decortication of the ridge was performed using a round bur to achieve bleeding points. FDBA was placed on the ridge to augment it buccally and coronally. A non-absorbable, titanium-reinforced expanded polytetrafluoroethylene (ePTFE) membrane (in this case, Gore-Tex® Titanium Reinforced, W.L. Gore & Associates, www.gore.com) was trimmed and used to cover the grafted area and secured with two titanium tacks at its apical aspect (Figure 7). The area was secured using 4-0 expanded ePTFE sutures in interrupted and horizontal mattress configurations (Figure 8). Primary coverage was achieved. After 6 months of uneventful healing, Stage 1 implant placement was initiated.
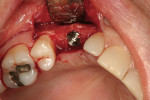
No. 6 Fixture Placement
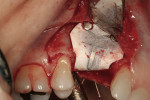
After oral sedation with 0.25 mg triazolam and local anesthetic induction using 2% lidocaine with 1:100,000 epinephrine and 0.5% bupivacaine with 1:200,000 epinephrine, a flap was created using the same incision design and elevation/dissection method documented above. However, no vertical incision was made at the mesio-buccal line angle of tooth No. 7. Initial exposure of the ridge showed the continued presence of the ePTFE barrier, which was detached using forceps after the tacks were removed (Figure 9). Degranulation of the site with a pear-shaped carbide finishing bur divulged sufficient supracrestal vertical and horizontal bone regeneration. Significant horizontal and vertical regeneration was seen (Figure 10). To preserve and expand the bone laterally, the osteotomy was created using expander drills (BTI®, www.bti-implant.com). A rough-surfaced, internal hex 4-mm (diameter) by 11.5-mm (length) implant was placed into the prepared ridge (in this case, NanoTite® Parallel Walled Certain® Implants, Biomet 3i Implant Systems, www.biomet3i.com) (Figure 11). FDBA was placed on the intact buccal surface to augment the ridge further. Primary stability was achieved, and the flap was primarily closed with 4-0 ePTFE sutures in an interrupted fashion after placement of the cover screw. The area was re-temporized with a resin-bonded fixed partial denture (FPD).
Guided Soft-Tissue Growth
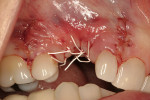
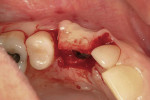
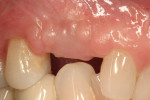
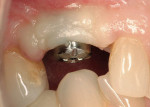
A mucosal discrepancy continued to exist 3 months post-implant placement (Figure 12). In order to enlarge the soft-tissue volume and allow for proper drape around the final prosthesis, guided soft-tissue growth was performed. After oral sedation with 0.25 mg triazolam and local anesthetic induction using 2% lidocaine with 1:100,000 epinephrine and 0.5% bupivacaine with 1:200,000 epinephrine, a flap was created using the same incision design and elevation/dissection method documented above, except for the absence of any vertical cuts placed at tooth No. 5 or No. 7. The implant cover screw was substituted with a one-piece, 4.1-mm (platform) by 5-mm (emergence profile) by 2-mm (height) healing abutment and the flap positioned over the abutment (in this case, Certain® EP Healing Abutment, Biomet 3i Implant Systems, www.biomet3i.com) and secured with 4-0 ePTFE suture in a simple interrupted pattern (Figure 13, Figure 14 and Figure 15). This effectively tented up the mucosa, allowing soft tissue to fill in the created void.
Implant Exposure and Final Prosthetics
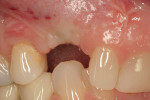
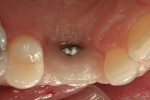
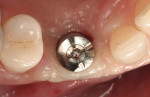
Adequate soft tissue was seen 1 month after surgery (Figure 16). A small exposure of the healing abutment occurred but did not appear to cause or reflect infection or dampen results (Figure 17). Using a tissue-punch technique, a two-piece CAD/CAM healing abutment (in this case, Certain® Encode® 2-PC EP Healing Abutment, Biomet 3i Implant Systems, www.biomet3i.com) with dimensions of 4.1 mm (platform) by 5 mm (emergence profile) by 4 mm (height) was placed on the No. 6 implant, after which end-stage restorative procedures began (Figure 18 and Figure 19). Three weeks after the final impression was taken, a temporary crown was placed on the implant to further mold the soft tissue (Figure 20). Due to excessive buccal soft tissue over the temporary prosthesis, mucosal abrasion with diamond burs was utilized to contour the soft tissue.
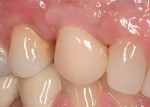
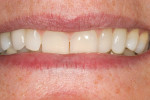
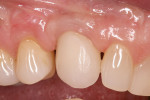
Roughly 17 months after treatment began, the patient received a final No. 6 implant crown (Figure 21). The marginal height of the No. 6 implant crown closely approximated that detected at original presentation (Figure 22). The patient was satisfied with the functional and esthetic result.
Postoperative Instructions
After each surgical procedure, the patient was instructed to take ibuprofen 600 mg every 4 to 6 hours, hydrocodone 7.5 mg/acetaminophen 750 mg every 4 to 6 hours as needed for pain, and doxycycline 100 mg once daily for 10 days. The patient was instructed not to brush at or near the surgical site but instead to rinse with 0.12% chlorhexidine or warm saline twice daily. The patient was also directed not to chew in the affected area for at least 2 weeks. Suture removal occurred 10 to 14 days after surgery.
Discussion
As the case report above demonstrates, it is possible to create stable, esthetically acceptable restorations using GBR prior to or in combination with implant placement.12 Correct diagnosis and timing boost the chances for success. Along with a well-calculated therapeutic design, success is contingent on the effort and persistence of both the treatment team and the patient. The most promising end result may require multiple surgeries, punctuated by extensive healing periods. In the example of a long span of adjacent implants, there still exists no assurance of ideal mucosal esthetics in the anterior, especially for replacement of large-rooted teeth; "black triangles" may be very difficult to avoid. In terms of function and stability, however, GBR performed pre- and peri-implantation is a favorable, predictable technique.
Acknowledgment
Restorative work courtesy of Dr. Joseph Worthington.
References
1. Schropp L, Kostopoulos L, Wenzel A. Bone healing following immediate versus delayed placement of titanium implants into extraction sockets: a prospective clinical study. Int J Oral Maxillofac Implants. 2003;
18(2):189-199.
2. Camargo PM, Lekovic V, Weinlaender M, et al. Influence of bioactive glass on changes in alveolar process dimensions after exodontia. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;90(5):581-586.
3. Araújo MG, Sukekava F, Wennström JL, et al. Ridge alterations following implant placement in fresh extraction sockets: an experimental study in the dog. J Clin Periodontol. 2005;32(6):645-652.
4. Botticelli D, Berglundh T, Lindhe J. Hard-tissue alterations following immediate implant placement in extraction sites. J Clin Periodontol. 2004;31(10):820-828.
5. Iasella JM, Greenwell H, Miller RL, et al. Ridge preservation with freeze-dried bone allograft and a collagen membrane compared to extraction alone for implant site development: a clinical and histologic study in humans. J Periodontol. 2003;74(7):990-999.
6. Lekovic V, Camargo PM, Klokkevold PR, et al. Preservation of alveolar bone in extraction sockets using bioabsorbable membranes. J Periodontol. 1998;69(9):1044-1049.
7. Chiapasco M, Zaniboni M, Boisco M. Augmentation procedures for the rehabilitation of deficient edentulous ridges with oral implants. Clin Oral Implants Res. 2006;17(Suppl 2):136-159.
8. Donos N, Mardas N, Chadha V. Clinical outcomes of implants following lateral bone augmentation: systematic assessment of available options (barrier membranes, bone grafts, split osteotomy). J Clin Periodontol. 2008;35(8 Suppl):173-202.
9. Tinti C, Parma-Benfenati S. Clinical classification of bone defects concerning the placement of dental implants. Int J Periodontics Restorative Dent. 2003;23(2):
147-155.
10. Wang HL, Boyapati L. “PASS” principles for predictable bone regeneration. Implant Dent. 2006;15(1):8-17.
11. Wang HL, Misch C, Neiva RF. “Sandwich” bone augmentation technique: rationale and report of pilot cases. Int J Periodontics Restorative Dent. 2004;24(3):232-245.
12. Anitua E. Plasma rich in growth factors: preliminary results of use in the preparation of future sites for implants. Int J Oral Maxillofac Implants. 1999;14(4):529-535.
About the Authors
Michael Sonick, DMD
Private Practice
Fairfield, Connecticut
Debby Hwang, DMD
Private Practice
Fairfield, Connecticut