When You Cannot Brush and Floss...
Ideally, we should cleanse our mouths and teeth after each snack or meal. Nuvora, Inc’s (Sunnyvale, CA) breakthrough Oral Health Lozenge (Figure 1) makes this possible. This patented,1 long-acting, convenient lozenge product is designed to slowly release active ingredients for about 1 hour to help dissolve biofilm, kill bacteria, capture malodorous compounds, neutralize acids, and stimulate saliva production in the oral cavity. According to the manufacturer, the Nuvora Oral Health Lozenge is the only oral healthcare product that has these multifunctional capabilities.
It has long been recognized that dental biofilm cannot be eradicated; however, plaque and its deleterious effects can be controlled with proper and diligent oral hygiene. The accepted oral hygiene regimen, recommended by dentists, is the combination of twice daily brushing and flossing. Recently, the American Dental Association has recognized the value of antiseptic mouth rinses in improving the efficacy of the daily oral hygiene regimen with the goal of preventing and inhibiting the maturation of the biofilm, which plays a major role in the etiology of gingivitis, periodontitis, and caries.2,3
We all must actively and diligently conduct a cleaning routine that controls the accumulation of dental plaque. However, many of us cannot accomplish this task consistently and effectively, partly because of the inadequacy of proper mechanical plaque control and lack of time. Compli-cating this problem is the fact that brushing, flossing, scraping the tongue, and gargling using a variety of tools and devices are commonly done in the privacy of one’s home and are typically considered socially unacceptable conduct in public. Therefore, many face the problem of not taking optimum care of our basic dental cleaning requirements. Even if we attempt an adequate level of plaque control, bacteria begin to recolonize the tooth surfaces immediately after a thorough dental prophylaxis, which in turn initiates the process of plaque reformation. Irrespective of which plaque hypo-thesis (nonspecific or specific) is favored, the goal is to remove excessive biofilm and the noxious products associated with it.
The Nuvora Oral Health Lozenge offers a convenient and pleasant-tasting solution for "in-between" cleaning by addressing a wide range of issues related to non-optimal oral hygiene. Biofilm protects pathogenic and non-pathogenic microorganisms from the outside by exhibiting a formidable permeation barrier. Nuvora’s SuRe™ (Sustained Release) Technology addresses the fact that significant time (up to 1 hour) is required for active agents to be in contact with the biofilm to effectively penetrate it, dissolve it, and then kill the bacteria that are protected inside the film. The active agents delivered by other products such as antiseptic mouth rinses, chewing gums, and breath mints do not last long enough in the mouth because they are flushed away or dissipate too quickly. Therefore, they do not achieve the required time necessary to allow the active agents’ penetration in-to and dissolution of the biofilm; at best, these short-lasting products affect only the biofilm surface. Therefore, the goal in every chemical cleaning process is to increase the contact time of the active agent with the biofilm and to enhance the biofilm’s permeability so that other active substances can penetrate the film to accomplish their targeted function. This can be achieved by plasticizing the biofilm or have permeation enhancers enter into the film, similar to the approach in transdermal drug delivery, where the skin’s permeability often has to be enhanced so that active agents can penetrate more easily and in sufficiently high amounts.4
These consecutive processes (partitioning, permeation, and dissolution) take time (about 30 to 60 minutes). Thus, short-lasting products may not reach the desired effectiveness (Figure 2). For example, the active ingredients provided by an antiseptic mouth rinse are in contact with the biofilm not longer than 30 seconds. During that time only the surface of the biofilm is affected. Chewing gums and mint candies last a little longer (less than 10 minutes) but often contain sugars, which provide nutrition to the developing biofilm and, therefore, do not achieve the desired cleansing effect. In contrast, Nuvora’s SuRe Technology assures that the contact time of active agents with the biofilm is sufficiently long enough (the lozenge lasts in the mouth for about 1 hour) to affect the biofilm and helps the user with daily oral care when brushing, flossing, or gargling are not feasible or convenient. One study demonstrated that microbial levels are reduced immediately after treatment by both antiseptic mouth rinse and lozenge. However, the bacterial reduction is greater for the lozenge. One hour after application, the bacterial rebound is larger for the antiseptic mouth rinse. The lozenge appears to create a reservoir effect that maintains a lower bacterial load level.5
SuRe Technology is the basis for Nuvora’s sophisticated and long-lasting lozenges. Essential oils combined with FDA-acceptable polymers form the semi-solid, sustained-release matrix of the lozenge, which holds other beneficial ingredients. The appropriate combination allows custom-tailoring of the desired dissolution time of the lozenge, and gives Nuvora control over the delivery-time interval and agent release rates. The beneficial agents can have antimicrobial, anti-fungal, local anesthetic, anti-inflammatory, and flavor-masking or enhancing activities; help alleviate dry mouth problems; and prevent caries.
The ingredients used in the Nuvora Oral Health Lozenge are either generally recognized as safe (GRAS) or are used in many other over-the-counter products. All Nuvora ingredients have been found to be safe in those other products. Because of the prolonged contact time with the biofilm-coated structures, the active agents are more efficient, and, therefore, the dosages of the Nuvora ingredients are up to 1,000 times smaller compared to those found in commercially available antiseptic mouth rinses. Despite the lower dosage in the lozenge, the longer contact time achieves a greater efficacy, according to the manufacturer.
References
1. United States Patent Applications 20040151771 and 20040247669.2. Special Supplement: Antimicrobial Mouthrinses as Part of Daily Oral Care. J Am Dent Assoc. 2006;137(Suppl 3).
3. Gunsolley JC. A meta-analysis of six-month studies of antiplaque and antigingivitis agents. J Am Dent Assoc. 2006; 137(12): 1649-1657.
4. Baker R, Kochinke F. "Transdermal Drug Delivery Systems." Controlled Release of Drugs: Polymers and Aggregate Systems. VCH, 277-305.
5. Internal Pilot Study JG2005 (Bacterial Load Assessment). Nuvora, Sunnyvale CA.
This article was written by Frank Kochinke, PhD.
For more information, contact:
Nuvora, Inc
Fax: 408-730-1924
Web: www.nuvorainc.com
E-mail: info@nuvorainc.com
DISCLAIMER
The preceding material was provided by the manufacturer. The statements and opinions contained therein are solely those of the manufacturer and not of the editors, publisher, or the Editorial Board of Inside Dentistry. The preceding is not a warranty, endorsement, or approval for the aforementioned products or services or their effectiveness, quality, or safety on the part of Inside Dentistry or AEGIS Communications. The publisher disclaims responsibility for any injury to persons or property resulting from any ideas or products referred to in the preceding material.
 | 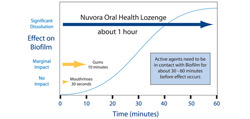 | |
| Figure 1 Nuvora Oral Health Lozenge with revolutionary SuRe Technology is the only product that assures sufficiently long contact time required to achieve desired cleaning and fresher breath. | Figure 2 Schematic depiction of different product technologies and their contact time with biofilm. |



