Botulinum Toxin Type A in The Treatment of Dental Conditions
Andrew Blumenfeld
Temporomandibular disorder (TMD) is an umbrella term used to describe a number of diseases affecting masticatory function, which may include true pathology of the temporomandibular joint as well as masticatory muscle dysfunction.1,2 TMD manifests with facial pain, joint sounds, headache, peri-auricular pain, neck pain, and/or decreased jaw excursion. The precise differentiation of relevant etiologic factors is often difficult because of the lack of universally accepted diagnostic criteria;1,2 thus, there are no generalized treatment approaches to TMD3,4 and the various disease entities are often isolated and treated independently in clinical practice.
Periodontal and occlusal diseases having an etiology in dysfunction of the masticatory musculature are a key component of TMD (Figure 1 and Figure 2). Because the temporomandibular joint (TMJ) is one of the body’s most active joints, the significance of dysfunction of the muscles responsible for TMJ function is critical, and the majority of TMD cases include a myogenic component.3,5 Muscular spasticity secondary to bruxism, external stressors, oromandibular dystonia, and psychomotor behaviors are common etiologic factors of TMD.1,6 For example, parafunctional clenching can intensify myofascial pain, especially when the relaxed posture of the mandible does not match the occlusion, thereby further contributing to TMJ dysfunction.
Excessive pathologic nocturnal clenching of the jaw and grinding of the teeth, which in severe cases may manifest as dystonic bruxism, contributes to TMJ dysfunction in addition to the damage of the teeth, bone, joints, and gums.7 Without treatment, tooth decay is more prevalent because excessive forces cause microfractures and abfracturing of enamel, especially around existing restorations,8 and may also be followed by gingival recession. TMD caused by excessive biting forces has conventionally been treated with intraoral appliances, occlusal adjustments, dental restoration, and/or surgery. Many of these techniques currently in use (including orthognathic surgery, orthodontics, and neuromuscular rehabilitation of occlusion), are invasive, irreversible, and expensive for the majority of patients. There is also no guarantee that these major treatments will be effective4,9,10 and they may instead exacerbate and perpetuate pain symptoms and lead to irreversible damage.4,9 Restorations are often highly technique-sensitive and, in many cases, may involve the removal of healthy tooth material. Techniques currently employed for esthetic, conservative restorations may not withstand the parafunctional forces continually applied by some patients. Thus, many of these treatment options are not ideal for all patients, and muscular relaxation may pose a viable alternative. When a muscle relaxant is used with the muscles of mastication, this clenching reflex can be reduced or eliminated.11 Because a very small percentage of available force is required to masticate food, a slight relaxation of muscle function reduces bruxing and is usually insufficient to affect chewing and swallowing.12
Given these facts, prophylactic treatment for incipient TMD should be built around the reduction of both conscious and unconscious parafunctional destructive factors. Further, because extracapsular TMD is often transient, the least invasive initial treatment options are most desirable. In this context, one demonstrably effective technique for prophylactic management of parafunctional jaw muscle activity is to reduce the contraction/flexion cycles responsible for excessive destructive forces and gnathalogic diseases. Intraoral splints can be effective in preventing excessive wear and enabling the jaw to function in the most relaxed posture, yet dentists frequently report low compliance with intraoral splints and other protective removable appliances worn over the teeth even when they are effective.13 Patients do not like to have oral appliances impeding normal functions such as eating and speaking, and, on average, only 43% of patients will comply with intraoral splint treatments.11,13 Treatment of the myogenic component of TMD has been limited largely to supportive care. Physical therapies, oral pharmacotherapy, biofeedback, and other mo-dalities usually provide short-term and inadequate relief in more severe cases.3,4 Chronic systemic pharmacotherapy is largely unsatisfactory because of the modest improvements and frequent side effects, resulting in a low therapeutic ratio.10,14 Adverse side effects, such as the drowsiness that accompanies the use of muscle relaxants like diazepam, can curtail the activities of many patients and is an additional burden that may outweigh the therapeutic advantages afforded by these treatments.4 Furthermore, it has been estimated that three quarters of patients with severe chronic orofacial pain who are prescribed opioids do not achieve improvement in function or reduction in pain.10,15,16
There is a clear need to expand the current options available for preventive treatment of muscle-generated dental disease. The ideal treatment requires effective, safe agents with minimal side effects that are well-tolerated for long-term use and eliminate or reduce the need for other irreversible treatments or medications. Recently, intramuscular injections of botulinum toxin type A (Botox® Allergan, Inc, Irvine, CA) have been used throughout the United States as a novel approach to providing safe, effective, and well-tolerated long-term relief of intractable symptoms in patients who have failed conventional approaches to treatment.17-20 When injected into the masticatory muscles, botulinum toxin type A can eliminate or reduce the intensity of excessive jaw clenching by desensitizing muscular spindle cells, a main cause of force-related dental disease.21 Because the parafunctional clenching that contributes to force-related dental disease is usually transient, botulinum toxin type A may obviate the need for more aggressive and irreversible treatments and is also of benefit where compliance may be a problem.
Currently, seven botulinum neurotoxin serotypes (A, B, C1, D, E, F, and G), produced by Clostridium botulinum, are recognized.22 Although all inhibit acetyl-choline release, their intracellular target proteins, the characteristics of their actions and their potencies vary substantially.23 At the neuromuscular junction, the inhibition of acetylcholine release by botulinum toxin type A blocks or reduces the contraction of muscles, an effect which has been used therapeutically in disorders characterized by overactive muscle activity, such as cervical dystonia,24 blepharospasm,25 and spasticity of multiple origins.26 The Food and Drug Administration has approved Botox in the United States for the treatment of cervical dystonia, hyperhidrosis, strabismus, and blepharospasm and associated nerve disorders, and it is the muscle relaxant most widely recognized and used in these applications. Binder and colleagues, while treating patients for facial lines, noted improvement of migraine symptoms after Botox injections.27 This discovery led to further investigations of preventive treatment of migraine and other diseases including TMD.28 Given the extensive documentation of its utility in the literature to date, this review will be limited to consideration of the Botox formulation of botulinum toxin type A.
OROMANDIBULAR THERAPY: CLINICAL EXPERIENCE TO DATE
Since the early 1990s, botulinum toxin type A has been used in a large number of intraoral and perioral applications. Although many of these uses have remained unreported, and most reports are of small case-history studies, the success of these applications is compelling.
Pathologic Clenching
Excessive forces created by parafunctional clenching impede healing and reattachment of gum and bone in the mouth after trauma.29 The mechanism of action of botulinum toxin type A suggests that low doses can potentially limit this, and a reduction in clenching intensity will allow traumatized tissue to heal. Higher doses of botulinum toxin type A may potentially be used as a pharmaceutical splint, limiting muscle contraction before resetting and during rehabilitation after fracture of a facial bone (eg, fractured mandibular condyle). Because parafunctional clenching contributes to periodontal trauma, limiting clenching before and after periodontal surgery can benefit healing. Further, in this application, the use of a splint is often contraindicated because the teeth should be functional during healing. With significant bone loss, excessive forces may jeopardize dental stability and contribute to additional tooth loosening. The same applies in the patient with bone loss associated with either advanced periodontal disease or osteoporosis and a strong bite. Bite force is not diminished with reduced alveolar bone support. Maximum voluntary contraction has been shown to be initially reduced with botulinum toxin therapy,10 thus the use of botulinum toxin type A may offer an alternative to conventional splint therapy.
Where long-term temporization or a functional orthotic is used before occlusal reconstruction to ensure that the mandible and occlusion are correctly reset and that vertical dimension is maintained, the ideal mandibular position can vary considerably in all three dimensions. The ideal mandibular posture for successful occlusal reconstruction should be with the masticatory muscles relaxed. This can be easily achieved if clenching is reduced. Orthodontic treatments on patients who are clenchers or have a deep or crossed bite are prolonged if the vertical component of muscular force is greater than the force of the fixed or removable appliance. These cases often require the use of removable functional retainers in combination with regular fixed braces in an attempt to control the component of vertical force.12 Or-thodontic treatment time may be reduced, and patients are far more comfortable and functional (eating, speaking, swallowing), if clenching is reduced. Similarly, patients with chronic intermittent toothache may also stand to benefit from botulinum toxin type A if it can be used to verify that the problem is muscular and not pulpal (eg, complex toothache) in origin. For example, muscle pain from the anterior temporalis is often referred to the teeth. This should be treated before any major irreversible dental treatments are undertaken. In this context, the use of botulinum toxin type A may be a prophylactic and diagnostic one, eg, to help patients control parafunctional habits and thereby minimize the chances of temporomandibular and dental complications.
Oromandibular Dystonia
Oromandibular dystonia (OMD) is a movement disorder characterized by involuntary spasms and muscle contractions. It manifests as distorted oral position and function resulting in difficulty in speaking, swallowing, and eating. Although it is a neurologic disorder, it is included as a subset of TMD because of its involvement of the masticatory apparatus.30 Most of the reported literature on OMD have been open-label studies, but all have reported improvement with botulinum toxin injections.31-35 The largest study to date was a prospective open-label conducted by Tan and Jankovic that treated 162 patients with OMD over a 10-year period.35 Botulinum toxin type A was injected into the masseters (mean dose 54.2 ± 15.2 U/side) and/or the submentalis complex (mean dose 28.6 ± 16.7 U/side). Improvement in function (defined as a global score ? 3) for chewing and speaking was reported in 67.9% of the patients, and mean duration of clinical improvement was 16.4 ± 7.1 weeks.35
Mandibular Spasm
When the mandibular closing musculature remains semicontracted or in spasm, mouth opening is limited. Among other functional complications, this type of muscular spasm places limitations on completing the basic oral hygiene necessary to prevent oral disease.36 Other impairments can include: restrictions on dental treament (necessary dental treatment including x-rays is frequently limited by mechanical issues) difficulty with eating (the teeth cannot be separated sufficiently to bite many foods) and diminished oral utility (a broad spectrum of oral functions are impaired by restricted opening and the contraction of bite radius). Botulinum toxin treatment to the masticatory musculature diminishes the effects of hyperfunctional or spastic muscles.37
For example, several case reports have been published describing the effective use of botulinum toxin type A in five patients with hemimasticatory spasm, a condition involving jaw closure spasms of the temporalis and masseter muscles.37-41 All of the patients responded markedly to botulinum toxin injections.37 One small open-label study evaluated the effect of botulin-um toxin type A injection on pain in six patients with limited mouth-opening because of TMD.4 All of the patients had clinical remission of pain symptoms without any adverse effects during the 5- to 12-month follow-up period. Freund and Schwartz completed a large open-label trial with 46 patients suffering from TMD, and found that injections to the temporalis and masseter muscles with 150 U of botulinum toxin type A significantly improved pain, function, mouth opening, and tenderness to palpation.10
Bruxism
Various forms of bruxism have been described, which can affect the muscles solely and/or lead to forms of TMD involving joint damage.5 Many characteristics of bruxism mimic those of dystonia; Watts and coworkers have suggested that bruxism may actually be a form of dystonia.42 Patients with classic bruxism often have had symptoms for extended periods of time, frequently ranging from several years to several decades. These patients audibly grind their teeth, and in many cases have difficulty speaking, swallowing, or chewing. Like dystonia, pain is a large component, and symptoms are exacerbated by external factors such as fatigue, stress, and emotional extremes.5 If sleep bruxism is present, the most effective way to protect the teeth is with an occlusal appliance. The problem with occlusional covering appliances is that they do little or nothing to stop the bruxism and offer only a brief respite from headaches and bruxism-induced TMJ derangement or arthritis.43 If bruxism is a type of dystonia, it is possible that treatment success with intraoral or occlusal appliances may simply be a “sensory trick” that relieves dystonic symptoms.5 In general, however, because of the numerous presentations of bruxism, the current treatments available are neither uniform nor universally successful.5
Botulinum neurotoxin has shown prom-ise in alleviating the symptomatology of bruxism. One of the earliest reports on use of botulinum toxin type A for bruxism was by Van Zandijcke and Marchau,44 who described the successful treatment of a brain-injured patient with severe bruxism with 100 U of a botulinum toxin type A injections to the temporalis and masseter muscles. Ivanhoe and colleagues45 reported similar success with a 200 U dose of botulinum toxin type A in a separate brain-injury case report. A recent long-term, open-label trial studied 18 patients with a history of severe bruxism (14.8 ± 10 years) who were refractory to medical and dental procedures.46 Botulinum toxin type A injections were made into the masseters (mean dose: 61.7 U/side; range 25 U to 100 U), which yielded a total duration of therapeutic response of 19 weeks. Efficacy in the abolishment of grinding, on a scale of 0 to 4, averaged 3.4, with only one patient experiencing transient dysphagia.46
Dental Implants and Surgery
Oral and maxillofacial implants and fracture repair often require multiple fixation sites and hardware to overcome the strong forces of masticatory musculature. Overloading of these muscles can prevent or impede osseointegration of implants and/or fracture callus formation.47,48 The muscular relaxation achieved with botulinum toxin type A injections to the masticatory muscles can be therapeutically beneficial by allowing implant structures better unimpeded integration and fracture healing in a more stable environment. Kayikvioglu and colleagues conducted a small open-label study to prospectively examine the use of botulinum toxin type A as an adjunct to zygomatic fracture fixation surgery, in an attempt to reduce the number of fixation sites and to prevent dislocation of the zygomatic bone.48 Five male patients with zygomatic bone fractures were injected with 100 U of botulinum toxin type A into the masseter muscle of the fractured side. Patients were then operated on 12 to 48 hours after the injection and EMG confirmation of muscle denervation.48 The temporary paralysis of the masseter muscles allowed for fewer miniplate and/or microplate insertions in all patients, and resulted in no complications related to either the botulinum toxin injections or surgical procedures. Kayikvioglu’s group also found similar benefits of adjunct botulinum toxin treatment for surgical reduction of mandibular and condylar bone fractures.48
Prominent Gums
The display of excessive gingival tissue in the maxilla upon smiling, or “gummy smile,” is both an oral hygiene and cosmetic issue with no simple remedy. Although several surgical techniques have been reported in the literature for correction of hyperfunctional upper lip elevator muscles, such as the Rubinstein and Kostianovsky,49 Miskinyar,50 and Rees and LaTrenta51 techniques, they are not routinely used to treat gummy smile.52 In general, the most common surgical corrections currently used are the LeFort I maxillary osteo-tomies with impaction for skeletal vertical maxillary excess, and gingivectomies for delayed passive dental eruption with excessive gingival display.52
Excessive gum exposure is frequently attributable to over-contraction of the upper lip muscles, particularly the levator labii superioris alaeque nasi. When this is the case, a less invasive approach is to limit muscular over-contraction. If applied in small, carefully titrated doses, these muscles can be proportionately weakened with botulinum toxin type A, which will reduce exposure of the upper gums when smiling. In a small open-label trial, five patients with excessive gingival display resulting from hyperfunctional upper-lip elevator muscles were treated with Botox injections under electromyographic guidance.52 Patients received one 0.25 U per muscle bilaterally into the levator labii superioris, levator labii superioris alaeque nasi, and at the overlap areas of the levator labii superioris and zygomaticus minor muscles. All of the patients were pleased with the results and the effective increase in upper-lip length upon smiling averaged 124.2%.52 The duration of effect ranged from 3 to 6 months, and no adverse effects were reported or observed.
Masseteric Hypertrophy
Patients who are chronic jaw clenchers frequently present with masseter hypertrophy.53,54 The increased size of these muscles is evident in the patient’s facial appearance which is often substantially altered (eg, the jaw can appear swollen and misshapen). The common treatment before botulinum toxin was surgical resection, which often resulted in substantial contracture.43 In several small but well-documented clinical trials, the injection of small aliquots (eg, 30 U per side) of Botox into the masseter muscles resulted in a sustained reduction of masseter hyperactivity.53-56 Over time, in most patients, reduction in masseter hyperactivity has been found to yield a concomitant reduction in gross masseter size (maximum reduction 35.4%).53 If the underlying pathology responsible for the hyperactivity is resolved, the reduction in masseter hypertrophy remains an enduring effect even after Botox applications have ceased.55,56
Adverse Events
Botulinum toxin has high affinity receptor binding to cholinergic nerve endings and enzymatic specificity, which leads to a low adverse-event profile. The potential adverse effects of botulinum toxin in oromandibular disorders include facial nerve palsy, pain at the injection site, flu-like symptoms, non-targeted muscle weakness, dysphagia, and hematoma.1,4 These complications are generally transient, and resolve within a couple weeks.35
DISCUSSION
Preventative control of clenching parafunction and force-related impacts on the chewing apparatus is a highly significant issue in dental treatment today. Major aggressive treatments that involve dental or alveolar surgery should be minimized whenever possible. Surgical procedures that alter anatomic relationships without addressing factors contributing to pathogenesis may be more prone to failure and recurrence. It is clear that excessive loading on dental tissues is one of the causative factors that must be identified and addressed by all clinicians treating patients with dental pathology.
CONCLUSION
Though more extensive confirmation of its use in multiple dental applications is needed, it is evident that the potential use of botulinum toxin in the dental profession can be of great value. Botulinum toxin in the dental office offers a reversible alternative to more aggressive procedures such as full-mouth reconstruction, orthodontics, and orthognathic surgery. Dentists should be able to learn injection techniques using botulinum toxins with additional training, enabling more patients to be helped with more conservative, less expensive procedures.
ACKNOWLEDGMENT
The author would like to thank Carrie Pennell MS, PhD; James Boyd, DDS; Barry Glassman, DMD; and Howard Katz, DDS, for their assistance in providing the scientific information and data presented in this article.
DISCLOSURE
The author has received research grants and other stock monies from Allergan.
REFERENCES
1 Bentsianov BL, Francis A, Blitzer A. Botulinum toxin treatment of temporomandibular disorders, masseteric hypertrophy, and cosmetic masseter reduction. Operative Techniques in Otolaryngology-Head and Neck Surgery. 2004;15(2):110-113.
2 Olivo S, Bravo J, Magee DJ, et al. The association between head and cervical posture and temporomandibular disorders: A systematic review. J Orofac Pain. 2006;20(1):9-23.
3 Schwartz M, Freund B. Botulinum Toxin A Therapy for Temporomandibular Disorders. Philadelphia, Pa: Lippincott Williams & Wilkins, 2002.
4 Lee KM, Chow J, Hui E, et al. Botulinum toxin type A injection for the management of myofascial temporomandibular pain disorder. Asian Journal of Oral and Maxillofacial Surgery. 2005;17:100-103.
5. Schwartz M, Freund B. Treatment of temporomandibular disorders with botulinum toxin. Clin J Pain. 2002;18(6 Suppl): S198-S203.
6. Castenada R. Occlusion. In: Kaplan A, Assael L (eds). Temporomandibular Disorders. Philadelphia, Pa: Saunders, 1992.
7. Israel HA, Diamond B, Saed-Nejad F, et al. The relationship between parafunctional masticatory activity and arthroscopically diagnosed temporomandibular joint pathology. J Oral Maxillofac Surg. 1999;57(9): 1034-1039.
8. Christensen G. Destruction of human teeth. J Am Dent Assoc. 1999;130(8): 1229-1230.
9. Israel HA, Ward JD, Horrell B, et al. Oral and maxillofacial surgery in patients with chronic orofacial pain. J Oral Maxillofac Surg. 2003;61(6):662-667.
10. Freund B, Schwartz M, Symington JM. Botu-linum toxin: New treatment for temporomandibular disorders. Br J Oral Maxillofac Surg. 2000;38(5): 466-471.
11. Gobel H, Heinze A, Heinze-Kuhn K. Botulinum Toxin A is effective in cases of oromandibular dysfunction even if previous bite splint therapy has proved unsuccessful. Cephalalgia. 2001: 514-515.
12. Freund B, Schwartz M, Symington JM, et al. The use of botulinum toxin for the treatment of temporomandibular disorders: Preliminary findings. J Oral Maxillofac Surg. 1999;57(8):916-921.
13. Wig AD, Aaron LA, Turner JA, et al. Short-term clinical outcomes and patient compliance with temporomandibular disorder treatment recommendations. J Orofac Pain. 2004;18(3):203-213.
14. Tinter R, Jankovic J. Botulinum toxin type A in the management of oromandibular dystonia and bruxism. In: Brin MF, Jankovic J, Hallett M, eds. Scientific and Therapeutic Aspects of Botulinum Toxin. Philadelphia, Pa: Lippincott Williams & Wilkins, 2002.
15. Zenz M, Strumph M, Tryba M. Long-term oral opioid therapy in patients with chronic nonmalignant pain. J Pain Symptom Manage. 1992;7(2):69.
16. Zuniga JR. The use of nonopioid drugs in management of chronic orofacial pain. J Oral Maxillofac Surg. 1998;56(9): 1075-1080.
17. Allergan Inc. BOTOX Prescribing Information. Irvine, CA; 2002.
18. Borodic G, Acquadro M. Management of facial pain with botulinum toxin in a tertiary pain clinic. Naunyn Schmiedebergs Arch Pharmacol. 2002;365(Suppl 2):R14.
19. Chikhani L, Jedynak CP, Vidailhet M, et al. Hypertrophy of the masticatory muscles, bruxism and painful syndrome of the temporo-mandibular junction. Treatment with botulinum toxin type A. Mov Disord. 1995; Abstract #389.
20. Fross RD. Bruxism and masticatory myalgias: Use of botulinum toxin. Mov Disord. 2000;(Suppl 2):35.
21. Hui ACF. Botulinum toxin for treatment of masseteric hypertrophy. J Neurol. 2002;249(3):345.
22. Dolly O. Synaptic transmission: inhibition of neurotransmitter release by botulinum toxins. Headache. 2003;43:16-24.
23. Welch MJ, Purkiss JR, Foster KA. Sensitivity of embryonic rat dorsal root ganglia neurons to Clostridium botulinum neurotoxins. Toxicon. 2000;38:245-258.
24. Brashear A. The botulinum toxins in the treatment of cervical dystonia. Semin Neurol. 2001;21(1):85-90.
25. Jankovic J, Brin MF. Therapeutic uses of botulinum toxin. N Engl J Med. 1991;324:1186-1194.
26. Brashear A, Gordon MF, Elovic E, et al. Intramuscular injection of botulinum toxin for the treatment of wrist and finger spasticity after a stroke. N Engl J Med. 2002;347(6): 395-400.
27. Binder WJ, Brin MF, Blitzer A, et al. Botulinum toxin type A (BOTOX) for treatment of migraine headaches: An open-label study. Otolaryngol Head Neck Surg. 2000;123(6):669-676.
28. Freund BJ, Schwartz M. Relief of tension-type headache symptoms in subjects with temporomandibular disorders treated with botulinum toxin-A. Headache. 2002;42(10) :1033-1037.
29. Glaros A, Tabacchi K, Glass EG. Effect of parafunctional clenching on TMD pain and hearing loss. J Orofacial Pain. 1998;12(2): 145-152.
30. Blitzer A, Brin MF, Greene PE, et al. Botulinum toxin injection for the treatment of oromandibular dystonia. Ann Otol Rhinol Laryngol. 1989;98(2): 93-97.
31. Brin MF, Fahn S, Moskowitz C, et al. Localized injections of botulinum toxin for the treatment of focal dystonia and hemifacial spasm. Mov Disord. 1987;2(4): 237-254.
32. Hermanowicz N, Truong DD. Treatment of oromandibular dystonia with botulinum toxin. Laryngoscope. 1991;101(11):1216-1218.
33. Jankovic J, Orman J. Botulinum toxin for cranial-cervical dystonia: a double-blind, placebo-controlled study. Neurology. 1987;27: 616-623.
34. Laskawi R, Rohrbach S. Oromandibular dystonia. Clinical forms, diagnosis and examples of therapy with botulinum toxin. Laryngorhinootologie. 2001;80(12):708-713.
35. Tan EK, Jankovic J. Botulinum toxin A in patients with oromandibular dystonia: Long-term follow-up. Neurology. 1999;53(9):2102-2107.
36. Erdal J, Werdelin L, Prytz S, et al. Experience with long-term botulinum toxin treatment of oromandibular dystonia, guided by quantitative EMG. Acta Neurologica Scandinavica. 1996;94(6): 210.
37. Cersosimo MG, Bertoti A, Roca CU, et al. Botulinum toxin in a case of hemimasticatory spasm with severe worsening during pregnancy. Clin Neuropharmacol. 2004;27(1):6-8.
38. Auger RG, Litchy WJ, Cascino TL, et al. Hemimasticatory spasm: Clinical and electrophysiologic observations. Neurology. 1992;42(12):2263-2266.
39. Crucu G, Inghilleri M, Beredelli A, et al. Pathophysiology of hemimasticatory spasm. J Neurol Neurosurg Psychiatry. 1994;57(1): 43-50.
40. Kim HJ, Jeon BS, Lee KW. Hemimasticatory spasm associated with localized scleroderma and facial hemiatrophy. Arch Dermatol. 2000;57(4): 576-580.
41. Kim Y, Lee K, Na J, et al. A case of hemimasticatory spasm. J Korean Neurol Assoc. 1994;12:175-178.
42. Watts M, Tan EK, Jankovic J. Bruxism and cranial-cervical dystonia: is there a relationship? Cranio. 1999;17(3):196-201.
43. Clark GT. The management of oromandibular motor disorders and facial spasms with injections of botulinum toxin. Phys Med Rehabil Clin North Am. 2003;14(4): 727-748.
44. Van Zandijcke M, Marchau MM. Treatment of bruxism with botulinum toxin injections. J Neurol Neurosurg Psychiatry. 1990;53(6): 530.
45. Ivanhoe CB, Lai JM, Francisco GE. Bruxism after brain injury: successful treatment with botulinum toxin-A. Arch Phys Med Rehabil. 1997;78(11):1272-1273.
46. Tan EK, Jankovic J. Treating severe bruxism with botulinum toxin. J Am Dent Assoc. 2000;131: 211-216.
47. Nishimura K, Itoh T, Takaki K, et al. Periodontal parameters of osseointegrated dental implants: A four-year controlled follow-up study. Clin Oral Implants Res. 1997;8(4):272-278.
48. Kayikvioglu A, Erk Y, Mavif E, et al. Botulinum toxin in the treatment of zygomatic fractures. Plast Reconstr Surg. 2003;111(1):341-346.
49. Rubenstein A, Kostianovsky A. Cosmetic surgery for the malformation of the laugh: original technique. Prensa Med Argent. 1973;60:952.
50. Miskinyar SA. A new method for correcting a gummy smile. Plast Reconstr Surg. 1983;72:397-400.
51. Rees T, LaTrenta G. The long face syndrome and rhinoplasty. Persp Plast Surg. 1989;3: 116.
52. Polo M. Botulinum toxin type A in the treatment of excessive gingival display. Am J Orthod Dentofacial Orthop. 2005;127(2):214-218.
53. Kim HJ, Yum KW, Lee SS, et al. Effects of botulinum toxin type A on bilateral masseteric hypertrophy evaluated with computed tomographic measurement. Dermatol Surg. 2003;29(5):484-489.
54. Al-Ahmad HT, Al-Qudah MA. The treatment of masseter hypertrophy with botulinum toxin type A. Saudi Med J. 2006;27(3):397-400.
55. Mandel L, Tharakan M. Treatment of unilateral masseteric hypertrophy with botulinum toxin: Case report. J Oral Maxillofac Surg. 1999;57(8): 1017-1019.
56. Rijsdijk BA, Van Es RJJ, Zonneveld FW, et al. Botulinum toxin type A treatment of cosmetically disturbing masseteric hypertrophy. Ned Tijdschr Geneeskd. 1998;142(10):529-532.
57. Gray H. Anatomy of the Human Body. Philadelphia, PA: Lea & Febiger, 1918.
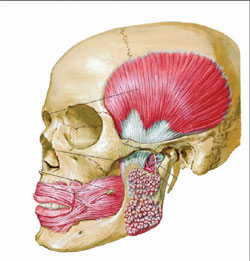 | 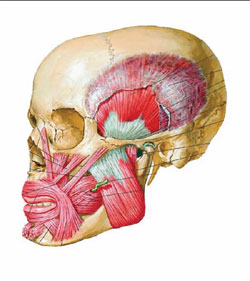 |
| Figure 1 The masseter is a thick, somewhat quadrilateral muscle, consisting of two portions, superficial and deep. The superficial portion, which is the larger of the two, arises by a thick, tendinous aponeurosis from the zygomatic process of the maxilla, and from the anterior two thirds of the lower border of the zygomatic arch; its fibers pass downward and backward, to be inserted into the angle and lower half of the lateral surface of the ramus of the mandible. The deep portion is much smaller, and more muscular in texture; it arises from the posterior third of the lower border and from the whole of the medial surface of the zygomatic arch; its fibers pass downward and forward, to be inserted into the upper half of the ramus and the lateral surface of the coronoid process of the mandible. The deep portion of the muscle is partly concealed, in front, by the superficial portion; behind, it is covered by the parotid gland. The fibers of the two portions are continuous at their insertion.57 (Netter illustration used with permission from Icon Learning Systems, a division of MediMedia USA, Inc. All rights reserved.) | Figure 2 The temporalis (temporal muscle) is a broad, radiating muscle situated at the side of the head. It arises from the whole of the temporal fossa (except the portion of it which is formed by the zygomatic bone) and from the deep surface of the temporal fascia. Its fibers converge as they descend and end in a tendon, which passes deep to the zygomatic arch and is inserted into the medial surface, apex, and anterior border of the coronoid process, and the anterior border of the ramus of the mandible nearly as far forward as the last molar tooth. The pterygoideus externus (external pterygoid muscle) is a short, thick muscle, somewhat conical in form, which extends almost horizontally between the infratemporal fossa and the condyle of the mandible. It arises by two heads; an upper from the lower part of the lateral surface of the great wing of the sphenoid and from the infratemporal crest; a lower from the lateral surface of the lateral pterygoid plate. Its fibers pass horizontally backward and lateralward, to be inserted into a depression in front of the neck of the condyle of the mandible, and into the front margin of the articular disk of the temporomandibular articulation. The pterygoideus internus (internal pterygoid muscle) is a thick, quadrilateral muscle. It arises from the medial surface of the lateral pterygoid plate and the grooved surface of the pyramidal process of the palatine bone; it has a second slip of origin from the lateral surfaces of the pyramidal process of the palatine and tuberosity of the maxilla. Its fibers pass downward, lateralward, and backward, and are inserted, by a strong tendinous lamina, into the lower and back part of the medial surface of the ramus and angle of the mandible, as high as the mandibular foramen.57 (Netter illustration used with permission from Icon Learning Systems, a division of MediMedia USA, Inc. All rights reserved.) |
| About the Author | |
Andrew Blumenfeld, MD | |



