Periodontal Plastic Surgery I: Root Coverage
Michael Sonick, DMD; Debby Hwang, DMD
A smile is one of the most primitive forms of human communication, and it is not surprising that an esthetic one is the major ambition of patients. Face-lip, lip-tooth, and lip-gingiva relations all determine the appeal of a smile. The amount of tooth structure exposed during smiling depends on a number of factors: the degree of contraction of the muscles of facial expression, soft tissue level, skeletal characteristics, and the design of restorations, tooth shape, or tooth wear. Most people fail to expose much of the gingival tissue during smiling but those with a short upper lip, hypermobile lips, or large alveolar processes often do.1 Excessive gingival display occurs in patients with a moderately long upper lip only in the presence of unusual maxillary anterior supra-eruption or skeletal hyperplasia.
The Perfect Smile
Three general classifications of smile lines exist, based on the height of the upper lip relative to the maxillary anterior central incisors: high, average, and low. A high smile exposes the total length of the maxillary anterior teeth and a contiguous band of gingiva.2 An average smile exhibits 75% to 100% of the teeth and the interproximal gingiva only, while a low smile displays less than 75% of the anterior teeth. Roughly 70% of people have an average smile, 20% have a low smile, and 10% have a high smile. Men present more frequently with low smile lines, though perhaps this mirrors conformity to masculine archetypes.
The Perfect Gums
The clinician cannot underestimate the magnitude of the periodontal drape in the look of a smile. The gingiva frames the teeth, and its position, shape, and color establishes an esthetic facade. Ideally, the gingival margin of the maxillary lateral incisors lies 1 mm to 2 mm incisal to that of the central incisors and canines.3 The height of contour of the gingival margin of maxillary central incisors and canines occurs at the distal line angle; alternatively, the lateral incisor’s height of contour exists at the mesiodistal center.4
Tissue thickness and tooth shape govern the degree of gingival scallop. Thick tissue and square-shaped teeth support a flat gingival contour (more masculine), while thin tissue and triangular teeth favor a scalloped margin (more feminine).5 Papillae fill the embrasure spaces in healthy patients, after the morphology of the intact underlying bone. In the anterior region, papillae appear convex, reduced in width, and pyramidal or knife-edged. Papillae become flatter between posterior teeth. Healthy gingiva appears pink, with possible stippling, and entirely covers the cementum. The epithelial attachment of the gingiva to the tooth should lie at the cementoenamel junction (CEJ) or immediately above.
How to Achieve Gingival Beauty
Patients often complain about smiles that expose “too-long teeth.” These unsightly issues may stem from a flawed gingival drape. A “mucogingival deformity” arises when there exists a significant departure from the normal shape of the soft tissue, with or without bone loss. Examples include recession and altered passive eruption—basically too little and too much tissue, respectively. To correct such deviations from the norm, periodontal plastic surgery is performed, including grafts for root coverage as well as esthetic crown lengthening.
The following sections of this article address how to diagnose and what to do in cases of exposed roots due to insufficient gingiva. A future article will discuss the opposite scenario: a surplus of tissue.
Deficient Gingival Tissue
Buccal or lingual recession exists when the anatomical root becomes visible and exposes cementum. A related or separate entity, papillary recession occurs when the interdental tissue falls short of the contact point.6 Many therapies exist to cover root surfaces with success; papilla regeneration, on the other hand, is a much more unpredictable undertaking.
The prevalence of recession elevates with age; 90% of people 80 to 90 years old have at least 1 mm of exposure.7 Recession typically occurs on the buccal in males and in African-Americans, and on maxillary canines, premolars, and first molars along with mandibular central incisors in other individuals. Indications to treat recession involve esthetic disharmony, clearly, but include hypersensitivity and defect progression as well.
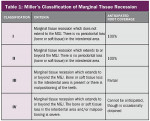
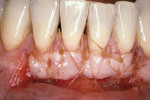
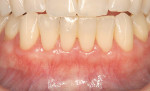
Treatment of defects first concerns the arrest of any etiological factors (Figure 1); success relies on choosing suitable cases to treat. Miller classified recession based on the relationship between the soft and hard tissues and, more importantly, correlated each level of recession to an estimated percentage of root coverage (Table 1).8 In essence, the higher the level of interproximal bone, the better the result.
Arguably, maintenance of a robust blood supply primarily determines graft survival.9 The adjacent and underlying bone provides the source, as do patent vessels in the surrounding mucosa. Thus, a substantial volume of bone and soft tissue become equally essential. Thick gingiva holds more intact vascular structures to feed the graft, whether the donor material is a repositioned flap or free tissue. There is no definitive set minimum measurement that denotes “thickness,” but generally, a flap that exceeds 1 mm favors success.10 Ultimately, if revascularization fails to occur, the graft dies, so close adaptation of the graft to the root and neighboring bone is a requirement.
Along with significant bone loss and relatively thin gingiva, the clinician must ensure that potential recipient sites do not possess the following, as these local factors hinder graft take: severe caries; existing cervical restorations; ectopic enamel; calculus; and hyperactive muscle attachment.
Once the surgeon clears the above requirements, he or she must screen for smoking, which affects wound repair. Tobacco use impairs oxygen exchange, collagen turnover, and immunologic response. To combat these problems, the patient should abstain from smoking at least 1 week before treatment and most likely refrain for the majority of the healing period, which is a minimum of 1 month postsurgery.11,12
Before consideration of the types of intervention, the clinician must first ask the following questions: Is the etiology addressed? Is there no or minimal bone loss? Is there thick tissue (at least 1 mm) at the recipient site? Is the patient a nonsmoker? Only when all answers are affirmative may root coverage surgery proceed.
Root Coverage Grafting
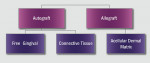
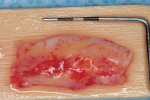
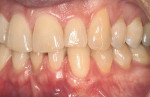
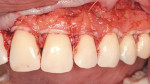
The final step is the selection of a suitable grafting regimen. What treatments exist? Which methods are best? There are two main types of root coverage procedures: reconstructive flaps (known also as “pedicle grafts”) (Figure 2) and free grafts (Figure 3).13 The first category uses tissue adjacent to the receded area and still attached at the base to cover the defect. This flapped mucosa may be rotated or simply advanced coronally to obscure the recession. This may or may not involve the papilla. Because there is a limit to the amount of available adjoining tissue and of lateral slide achievable, the rotational flap treats single receded areas with relative ease but multiple sites with difficulty. A coronally advanced flap (CAF), conversely, uses the gingiva immediately apical to the recession and does not compromise tissue overlying adjacent roots, permitting it to cover a more extensive region of recession.14-16
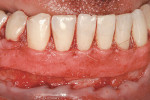
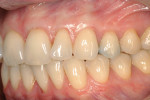
Alternately, the clinician may prefer to isolate tissue from a discrete secondary location. Compared to reconstructive flaps—rotational ones especially—free grafts usually provide more donor tissue but do not sustain a continuous blood supply. Free grafts consist of autogenous or allogenic tissue. Harvested typically from the host palate but occasionally other sites (eg, edentulous ridge, attached buccal gingiva), the free gingival graft (FGG) possesses a full layer of surface epithelium whereas the connective tissue graft (CTG) does not.17,18 That said, some clinicians leave a thin coronal strip of epithelium on the CTG to facilitate suturing (Figure 4A and Figure 4B).
Processed allograft from human dermis also serves as a free source. With cellular components removed but vasculature, collagen network, ground substance, and elastic fibers remaining, acellular dermal matrix (ADM) bars the need for a second surgical site and, as it is collected from cadavers, has a virtually limitless supply (Figure 5A; Figure 5B; Figure 5C; Figure 5D). With respect to defect elimination, case reports demonstrate high cosmetic success, but again, very few controlled trials support its use, as a recent meta-analysis suggested.19 In a retrospective analysis, Harris compared connective tissue graft to ADM mean root coverage after 4 years and discovered that only CTG sites retained a high percent of coverage at 97%.20 The average root coverage of teeth treated with ADM declined from 93% to 66% over the study period. Thus, acellular dermal matrix may lack endurance, perhaps due to the presence of elastic fibers that shrink.
Both reconstructive flaps and free grafts cover the root surface mostly with a long junctional epithelium or a scar; they do not restore the lost attachment apparatus (ie, bone, connective tissue, and cementum). To ameliorate this, some practitioners perform guided tissue regeneration (GTR) in conjunction with coronally positioned flaps. Membranes, either absorbable or nonabsorbable, impede epithelial down-growth and thus help to reestablish the connective tissue attachment via site repopulation with cementoblasts, osteoblasts, and periodontal ligament fibroblasts.21 In theory, such an attachment creates long-term stability. An investigation on GTR-mediated root coverage found 10-year maintenance of recession-depth improvement, though some attachment loss occurred over time.22 Harris, in contrast, saw significant and rather rapid deterioration of early GTR results, from 92% mean coverage at 6 months to just 60% at 2 years.23 No randomized controlled trials exist to verify these findings.
Bone graft may be used as an adjunct to membranes to encourage hard tissue regrowth over the recession defect, though studies fail to show any added benefit from demineralized freeze-dried bone allograft.24,25 Moreover, human histology presents minimal or inconsistent evidence of new bone and cementum from guided tissue regeneration.26 Accordingly, it is unfeasible to advocate use of membrane technology over traditional techniques, though it certainly remains a viable treatment option.
To be sure, there is a movement in periodontics to embrace the application of potentially regenerative materials to augment or even substitute for conventional therapy. This marks a conceptual evolution of the field from being anecdotally based to biologically rooted—a good thing—but at present, no longitudinal proof exists to substantiate claims of true regeneration or clinical superiority. As encouraging evidence mounts, however, better recommendations may be offered. Currently, groups employ several major biomimetic or human-derived products to improve root coverage, with mixed or unexceptional results (Table 2).27-32 This is not to say such materials do not work; there are promising scientific rationales for their function, but they may require improved clinical formulations.
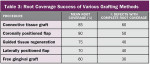
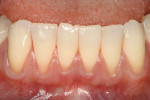
It is obvious that myriad ways exist to cover root recession. Which method gives the most predictable result? Strong data suggests that the CTG attains and sustains the greatest outcomes. Considered the “gold standard” of recession treatment, the CTG provides the highest frequency of 100% root coverage (Table 3).33 An example of a connective tissue graft is illustrated here (Figure 6A; Figure 6B; Figure 6C). In the end, the proper choice of therapy depends on defect morphology (Table 4).
No matter the treatment modality delivered, surgical overcorrection of a defect is not only desirable but also nearly mandatory. That is, the best way to guarantee a long-lasting, esthetic result is to attempt coverage beyond the borders of the recession. If the clinician employs a flap, he or she should make sure it extends past the defect. For instance, a coronally advanced flap ought to be positioned at least 2 mm coronal to the CEJ to ensure complete root coverage.34 If a free graft is used, it should be wide, with some thickness (and firmly adapted to the underlying tissue). Such overcorrection minimizes the effect of graft pullback and increases the likelihood of viability.
If therapy failed to cover the recession 100% initially, it is still possible to observe more root coverage over time, under the condition that grafting thickened the tissue. On occasion, a 1 mm coronal displacement of gingiva, dubbed “creeping attachment,” transpires 1 year postsurgery, barring inflammation.35-37 Contingent to a considerable degree upon thick tissue, the probability of creeping attachment increases for narrow initial defects, isolated defects, a lingual tooth position, good oral hygiene, and younger patients.38 Thicker gingiva at the very least resists further recession if not favors actual defect reduction.
Conclusion
If “I want a better smile” is the chief complaint of a patient, the practitioner must scrutinize not only the face, lips, and teeth, but also the periodontal drape. Treatment may include all the dental specialties as well as some medical ones to move and reshape teeth, shift the jaws, reconfigure facial structures, and position gingiva. The conscientious dentist realizes that even subtle revision of the soft tissue frame over the teeth causes visual tension and knows that gingival recession in particular ages people. Restoration of ideal mucosal contours via root coverage is crucial to the design of a pleasing smile. Keen diagnosis and elimination of receded areas may, at times, transform a listless face into a vibrant one and, consequently, bolster the patient’s self-worth, an incalculable reward.
References
1. Matthews TG. The anatomy of a smile. J Prosthet Dent. 1978;39(2): 128-134.
2. Tjan AH, Miller GD, The JG. Some esthetic factors in a smile. J Prosthet Dent. 1984;51(1):24-28.
3. McGuire MK. Periodontal plastic surgery. Dent Clin North Am. 1998;42(3): 411-465.
4. Lombardi RE. The principles of visual perception and their clinical application to denture esthetics. J Prosthet Dent. 1973; 29(4): 358-382.
5. Kois JC. Predictable single-tooth peri-implant esthetics: five diagnostic keys. Compend Contin Educ Dent. 2004;25(11):895-900.
6. Nordland WP, Tarnow DP. A classification system for loss of papillary height. J Periodontol. 1998;69(10):1124-1126.
7. Albandar JM, Kingman A. Gingival recession, gingival bleeding, and dental calculus in adults 30 years of age and older in the United States, 1988-1994. J Periodontol. 1999;70(1):30-43.
8. Miller PD Jr. A classification of marginal tissue recession. Int J Periodontics Restorative Dent. 1985;5(2): 8-13.
9. Mormann W, Ciancio SG. Blood supply of human gingiva following periodontal surgery. A fluorescein angiographic study. J Periodontol. 1977;48(11): 681-692.
10. Hwang D,Wang HL. Flap thickness as a predictor of root coverage: a systematic review. J Periodontol. 2006;77(10):1625-1634.
11. Miller PD Jr. Root coverage grafting for regeneration and aesthetics. Periodontol 2000. 1993;1(1):118-127.
12. Bergstrom J. Tobacco smoking and chronic destructive periodontal disease. Odontology. 2004;92(1):1-8.
13. Bahat O, Handelsman M. Periodontal reconstructive flaps—classification and surgical considerations. Int J Periodontics Restorative Dent. 1991;11(6):480-487.
14. Allen EP, Miller PD Jr. Coronal positioning of existing gingiva: short term results in the treatment of shallow marginal tissue recession. J Periodontol. 1989;60(6): 316-319.
15. Bernimoulin JP, Luscher B, Muhlemann HR. Coronally repositioned periodontal flap. Clinical evaluation after one year. J Clin Periodontol. 1975;2(1):1-13.
16. Prato GP, Clauser C, Cortellini P. Periodontal plastic and mucogingival surgery. Periodontol 2000. 1995;9:90-105.
17. Sullivan HC, Atkins JH. Free autogenous gingival grafts. I. Principles of successful grafting. Periodontics. 1968;6(3):121-129.
18. Langer B, Langer L. Subepithelial connective tissue graft technique for root coverage. J Periodontol. 1985;56(12):715-720.
19 Gapski R, Parks CA, Wang HL. Acellular dermal matrix for mucogingival surgery: a meta-analysis. J Periodontol. 2005;76(11): 1814-1822.
20. Harris RJ. A short-term and long-term comparison of root coverage with an acellular dermal matrix and a subepithelial graft. J Periodontol. 2004;75(5): 734-743.
21. Gottlow J, Nyman S, Lindhe J, et al. New attachment formation in the human periodontium by guided tissue regeneration. Case reports. J Clin Periodontol. 1986;13(6):604-616.
22. Trombelli L, Minenna L, Farina R, et al. Guided tissue regeneration in human gingival recessions. A 10-year follow-up study. J Clin Periodontol. 2005;32(1):16-20.
23. Harris RJ. GTR for root coverage: a long-term follow-up. Int J Periodontics Restorative Dent. 2002;22(1):55-61.
24. Al-Hamdan K, Eber R, Sarment D, et al. Guided tissue regeneration-based root coverage: meta-analysis. J Periodontol. 2003(10);74: 1520-1533.
25. Oates TW, Robinson M, Gunsolley JC. Surgical therapies for the treatment of gingival recession. A systematic review. Ann Periodontol. 2003;8(1):303-320.
26. Harris RJ. Histologic evaluation of root coverage obtained with GTR in humans: a case report. Int J Periodontics Restorative Dent. 2001;21(3):240-251.
27. Huang LH, Neiva RE, Soehren SE, et al. The effect of platelet-rich plasma on the coronally advanced flap root coverage procedure: a pilot human trial. J Periodontol. 2005;76(10):1768-1777.
28. Wilson TG Jr, McGuire MK, Nunn ME. Evaluation of the safety and efficacy of periodontal applications of a living tissue-engineered human fibroblast-derived dermal substitute. II. Comparison to the subepithelial connective tissue graft: a randomized controlled feasibility study. J Periodontol. 2005;76(6):881-889.
29. McGuire MK, Scheyer ET. Comparison of recombinant human platelet-derived growth factor-BB plus beta tricalcium phosphate and a collagen membrane to subepithelial connective tissue grafting for the treatment of recession defects: a case series. Int J Periodontics Restorative Dent. 2006;26(2): 127-133.
30. McGuire MK, Nunn M. Evaluation of human recession defects treated with coronally advanced flaps and either enamel matrix derivative or connective tissue. Part 1: Comparison of clinical parameters. J Periodontol. 2003;74(8):1110-1125.
31. McGuire MK, Cochran DL. Evaluation of human recession defects treated with coronally advanced flaps and either enamel matrix derivative or connective tissue. Part 2: Histological evaluation. J Periodontol. 2003;74(8):1126-1135.
32. Petrungaro PS. Using platelet-rich plasma to accelerate soft tissue maturation in esthetic periodontal surgery. Compend Contin Educ Dent. 2001;22(9): 729-746.
33. Clauser C, Nieri M, Franceschi D, et al. Evidence-based mucogingival therapy. Part 2: Ordinary and individual patient data meta-analyses of surgical treatment of recession using complete root coverage as the outcome variable. J Periodontol. 2003;74(5):741-756.
34. Pini Prato GP, Baldi C, Nieri M, et al. Coronally advanced flap: the post-surgical position of the gingival margin is an important factor for achieving complete root coverage. J Periodontol. 2005;76(5):713-722.
35. Harris RJ. Creeping attachment associated with the connective tissue with partial-thickness double pedicle graft. J Periodontol. 1997;68(9):890-899.
36. Dorfman HS, Kennedy JE, Bird WC. Longitudinal evaluation of free autogenous gingival grafts. A four-year report. J Periodontol. 1982;53(6):349-352.
37. Matter J. Creeping attachment of free gingival grafts. A five-year follow-up study. J Periodontol. 1980;51(12):681-685.
38. Matter J, Cimasoni G. Creeping attachment after free gingival grafts. J Periodontol. 1976;47(10): 574-579.
About the Authors
Michael Sonick, DMD
Director, Sonick Seminars
Fairfield, Connecticut
Debby Hwang, DMD
Private Practice
Fairfield, Connecticut