What You Can’t See Can Hurt Your Patients
According to "Oral Health in America," a 332-page report issued in 2000 by the US Surgeon General at the time, David Satcher, 65% of oral cancer is diagnosed in the late stages.1 That figure may be attributed to the fact that the signs and symptoms of oral cancer lesions are very similar to benign conditions often seen in the mouth, such as cheek bites, sores, and irritation caused by dentures and other appliances. Many patients are reluctant to seek care and may take a "wait and see approach" to what they believe are simply minor oral lesions, thinking they will resolve on their own. Patients finally make an appointment to see their doctor several weeks after they first discovered the lesion because now pain has set in the area. Their doctor then refers them to their dentist for an oral examination. The significant issue with oral cancers is that by the time they become painful or begin displaying erythema or leukoplakia, they are often dangerously well developed. Differential diagnosis of oral cancer at the earliest stage of the disease is crucial—that is, before the lesion is even apparent to the unaided eye and before it becomes painful.2,3
Examination and Detection Systems
There are several mucosal examination systems available to dentists today. Transepithelial (brush) biopsy products consist of a plastic stick with a small brush at one end, made up of tiny plastic bristles. The brush is swabbed on the suspect area, picking up microscopic pieces and disaggregated cells of mucosal tissue. The sample is then sent to a laboratory, which tests for the presence of dysplastic cells. This works well, but the difficulty is determining which area to test. How can dentists know where to sample if areas of concern cannot be clearly visualized? Other detection systems use the combination of a chemiluminescent light, an acetic acid (vinegar) oral rinse solution, and, finally, a blue marking dye. The patient must first swish the acetic acid solution in the mouth for up to 60 seconds and spit it out. The rinse must be thoroughly dispersed throughout the entire oral cavity before elimination. Next, a chemiluminescent light, or glow stick, is activated to shine in the patient’s mouth. Areas of mucosal tissue abnormality reflect the light more markedly and thus appear whiter than the surrounding tissue. To mark these areas, a swab saturated with a patented blue dye is applied to the locations that appeared whiter with the chemiluminescent light shone upon them.
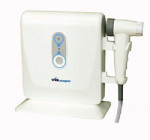
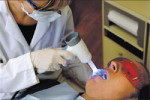
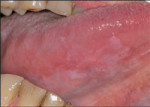
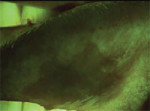
A new device in the quest for early identification of cancerous and precancerous squamous epithelium has recently been introduced: the VELscope™ Mucosal Examination System (LED Dental Inc, White Rock BC, Canada) (Figure 1 and Figure 2). Having received 510(k) approval from the US Food and Drug Administration in April 2006, VELscope uses direct optical fluorescence visualization (FV) technology. There are no stains, mouth rinses, or tissue sampling procedures involved. The handheld device emits a beam of blue light of a specific frequency band onto the oral mucosa and excites the various fluorescent molecules (ie, fluorophores) within the tissue. The clinician examines the oral mucosa by looking through the VELscope viewpiece. This noninvasive but robust field-of-view technology enables the direct visualization of fluorescence in the context of surrounding normal tissue. Under VELscope examination, normal, healthy tissue typically emits a bright apple-green glow, while potential early tumors and dysplastic or cancerous cells can appear as dark maroon to black (Figure 3 and Figure 4).
A pilot study demonstrated the efficacy of VELscope’s FV technology in detecting abnormal tissue by using histology as the gold standard to verify its findings. In this study, VELscope achieved a sensitivity rating of well over 95% when discriminating normal mucosa from severe dysplasia, carcinoma in situ, or invasive squamous cell carcinoma.4
Case Presentation
Following is the 12-year history of a patient who presented with an oral lesion that turned out to be squamous cell carcinoma. Unfortunately, the FV technology was not available before her initial diagnosis of oral cancer. If it had been, the lesion may have been visualized at its earliest stage (localized in situ), not after it presented as a painful sore.
In August 1994, 54-year-old Joyce presented with a 7-mm lesion on her left buccal mucosa. She had noticed it 2 months earlier, but held off having it checked, hoping it would heal and go away. Upon examination she was immediately referred to an oral surgeon who, based on his clinical experience, suspected a dangerous lesion and decided to excise the entire lesion immediately.
Histologic examination of the excised tissue returned the following diagnosis: "invasive, well-differentiated squamous cell carcinoma, left cheek, completely excised." A follow-up computed tomography (CT) scan was performed that indicated "no evidence of cervical adenopathy nor erosive change of the left mandible or maxilla." The location and size of the excision caused scarring and recurring discomfort to the patient. It was difficult to discern if the remaining tissue was normal but changed in appearance because of the surgery, or if it had changed due to a recurrence of cancer. The patient was re-examined every 6 months, but frankly the author was worried that he was missing something. The tissue was friable and tender, but the difficulty here was how to visually determine if this was a normal postsurgical mucosal appearance, or if neoplasm was the issue.
Throughout the following months and years, the site became progressively more uncomfortable with poor tissue integrity, and again, the author was not sure exactly what he was looking at. There were no significant changes in the outward appearance or size of the site over time. In January 2000, Joyce was experiencing increasing discomfort so the author sent her back to the oral surgeon for another biopsy of the area. This time the results from the histology report were "benign squamous cell mucosa with erosion, entrapped squamous epithelium and marked inflammation, with no neoplasm seen." This report explained the patient’s discomfort, but still, the author’s confidence was low—especially in the face of her continued soreness.
In 2001, Joyce still presented with oral soreness and poor tissue integrity in the area of the original excision. The oral surgeon felt it was just friable tissue at the surgical site, but the surgeon and the author together decided to send the patient to a dermatologist for further examination and diagnosis because she was now presenting with tiny ulcerative lesions in the area of the excision and on other areas of her body. The dermatologist ruled out lichen planus, oral pemphigus vulgaris/superficial pemphigoid, lupus, and molluscum contagiosum. Another biopsy was performed that was again negative.
The ulcerative lesions and discomfort seemed to subside during this treatment period, and the author continued to monitor the site every 6 months by means of a thorough visual (unaided) and palpation examination. In October 2002, the author used the OralCDx® Brush Biopsy (CDx Laboratories, Inc, Suffern, NY) to recheck the area because the site was so scarred that a visual examination made it impossible to differentiate and identify scar tissue and irritated epithelium from possible abnormal or neoplastic tissue. Using OralCDx to recheck the site was the first nonvisual test (short of a surgical biopsy) available at the time. The brush biopsy came back "negative for epithelial abnormality." Continued biopsies were problematic in this case because every time more tissue was taken, the less the area healed and the more discomfort the patient would endure.
In September 2003, tooth No. 17 lost all buccal attached gingiva and became increasingly sore and mobile, leading to its extraction. Eight months later, Joyce presented with difficulty opening her mouth and pain in the left temporomandibular joint. She was treated with nonsteroidal anti-inflammatory drugs (NSAIDs), as well as stretching exercises, a soft diet, and massage therapy. Two months later, during a recall exam, she was asymptomatic. Then, in August 2004, she presented with the chief complaint of increasing pain as she swallowed. There was erythema present in the left tonsilar fold area and left buccal mucosa. Again, the author and the oral surgeon were hesitant to perform another biopsy since the oral surgeon felt the pain was caused by poor tissue integrity, along with the fact that the patient had gone 10 years postoperatively with multiple negative tests.
Joyce was referred for consultation with a plastic surgeon in the hope that a plastic repair of the friable tissue might relieve her discomfort. The plastic surgeon first sent the patient to an ear-nose-throat (ENT) specialist for a comprehensive work-up and examination to rule out disease. A positron emission tomography (PET) scan was performed, which indicated a lesion in the left neck. The ENT specialist felt she must have a recurrence of the original squamous cell carcinoma or a new primary. To find the mucosal origin of this lesion, a biopsy was taken at the original surgical site on the left buccal mucosa. This site again returned a negative result. With the unaided eye, none of the doctors working on Joyce’s case could visualize abnormal mucosa anywhere else in her oral cavity, yet her ENT specialist felt there had to be a site of origin that would cause a lymph node to light up in her PET scan.
At this time, the author had just received a VELscope prototype unit, which he used on Joyce and found a positive area on her upper left maxillary tuberosity and adjacent soft palate. This mucosal abnormality was not apparent to the unaided eye and was easily associated with the positive node in the left neck that was identified by the PET scan. To be sure, the site was biopsied and histologic examination confirmed that it was in fact "squamous cell carcinoma."
After the diagnosis, Joyce underwent a 3-hour operation in which the intraoral area was excised and the neck node was treated with a neck dissection. The surgical treatment was then followed by chemotherapy and radiation treatments. Two years posttreatment, Joyce presents on VELscope exam and PET scan as cancer-free.
In this case, VELscope clearly indicated the position of the new cancerous site when it could not be visualized with the unaided eye. While no one involved in the case was sure if the new lesion was a recurrence or a new primary, the author feels that VELscope will give him a much higher level of confidence in discovering and identifying cancerous and precancerous oral tissues in all of his patients. It will also be indispensable when following up on patients after they are treated for oral cancer.
As dental professionals, there is a responsibility and obligation to the patients to examine and evaluate them for all oral diseases using the most clinically relevant diagnostic modalities available. There is no doubt in the author’s mind that routine and thorough oral mucosal examinations will save lives.
The author is in the process of incorporating VELscope into his practice routine as a part of the regular oral exam and cancer-screening protocol. He only wishes he had had the device in 1994 so he could have identified his patient’s oral cancer before it became invasive, and helped her to avoid a decade of surgeries and life-altering disease.
Disclosure
The author is a shareholder in LED Medical Diagnostics.
References
1. US Department of Health and Human Services. Oral Health in America: A Report of the Surgeon General. Rockville, Md: US Department of Health and Human Services, National Institute of Dental and Craniofacial Research, National Institutes of Health, 2000.
2. Pogrel MA. The dentist and oral cancer in the North East of Scotland. Br Dent J. 1974;137(1): 15-20.
3. Coffin F. Cancer and the dental surgeon. Br Dent J. 1964;161: 199-202.
4. Lane PM, Gilhuly T, Whitehead P, et al. Simple device for the direct visualization of oral cavity tissue fluorescence. J Biomed Opt. 2006:11(2).
About the Author
Anthony Palombaro, DDS
Binghamton, New York