Implant Dentistry: Evolution and Current Trends—The Times They are A-Changin’
Michael Sonick, DMD
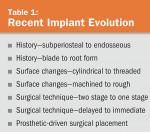
No longer considered experimental or exotic, implant dentistry has now entered mainstream dentistry. It is part and parcel of routine therapy and should be considered an option in every plan that requires tooth replacement. In fact, not offering implant treatment to restore edentulous areas may be considered substandard. The evolution of implant dentistry and some of the current implant trends will be explored in this article (Table 1 and Table 2).
Implant Evolution
A Brief History
The history of implant dentistry spans not only decades, but millennia. The ancient cultures of the world—in Egypt, Honduras, China, and Turkey, among others—substituted missing dentition with shells, stones, ivory, and other human or animal teeth.1,2 The establishment of metal replacements for teeth is a relatively recent development. In the 1800s, surgeons used gold, silver, lead, and platinum molded into various forms with varying degrees of success. By the early- to mid-20th century, more familiar implant morphologies developed. Dahl in 1940 created the subperiosteal implant, a structure that rested on, not in, the jaw.2 These implants frequently were met with complications including infection and bone resorption. Leonard Linkow developed the blade fixture for areas of deficient bone.3 Attached directly to the flat blade, an abutment protruded from the tissue. With time, the blade design fell out of favor as its complication rate precluded its use. In its place came the root form implant, which is the current standard shape. Thanks to the significant research by Brånemark, it was realized that osseointegration occurred between bone and titanium.4 Endosseous implants made from titanium have now become the standard.
Implant Surface Changes
Surface area augmentation often drives alterations in implant design. Over the past 15 years, practitioners have gradually switched from press-fit, cylindrical fixtures to threaded ones. A threaded implant leads to more immediate stability and hence greater success of osseointegration. A threaded form additionally facilitates self-tapping, which eases placement. Surface alterations could also be made microscopically. Surface roughness of the implant can be created through grit blasting, plasma spraying, etching, or coating. This raises the percentage of bone-to-implant contact and also accelerates wound healing.5-7 During the last two decades, a shift from a smooth to roughened design has transpired.
Surgical Technique,Timing, & Loading
The original Brånemark protocol published in 1977 influenced implant surgical technique and timing for years.4 It required that implants be submerged under the soft tissue for at least 4 months and discouraged load during the healing period. As more research accumulated, so did the evidence for unsubmerged implantation (ie, placement of a transmucosal healing abutment).8 Barring the need for grafting, one-stage fixture placement is not only acceptable clinically but also easily accepted by patients, as it prevents the need for an uncovering surgery. This decreases the surgical experiences for the patient and minimizes chair time.
Another time-saving surgical change is immediate temporization and immediate loading of dental implants within 48 hours of surgical implant placement. Case selection can be a thorny issue for these cases. Typically, immediate loading is non-occlusal, that is, non-functional temporization. The interim restoration placed should not occlude with the existing dentition. These cases usually involve single teeth and short-span fixed bridges. While not voluminous, the initial data seems quite promising with the exception of single posterior teeth.9
Immediate occlusal loading of dental implants shows significant promise when full-arch treatment is considered. Studies on full-arch immediate loading show success rates equal to conventional dental implant treatment.9 Contraindications for an immediate occlusal and non-occlusal load protocol consist of regions that experience undue mechanical stress, require grafting at the time of surgery or exhibit low bone density.10
Prosthetic-driven Surgical Placement
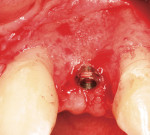
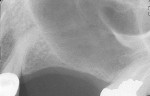
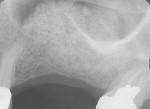
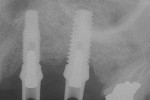
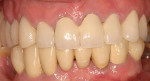
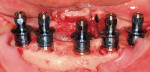
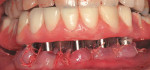
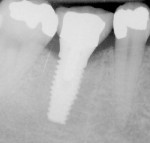
The most critical advancement for implant dentistry concerned a philosophical reversal. All too often, restorative dentists were left frustrated by non-favorably placed implants. “Well, that is where the bone was,” was a frequent refrain. Surgeons based implant positioning on the location or availability of the bone, for osseous grafting techniques were in their infancy. Today, this is not the case. The high predictability of current augmentation methods (ie, block grafting, guided bone regeneration (GBR), sinus elevation) allows for implantation based on prosthetic desires instead of biologic limitations (Figure 1; Figure 2; Figure 3; Figure 4; Figure 5).11-15 Thus the treatment goal targets the ideal reconstruction of the dentition. For the vast majority of cases, no compromises founded on anatomy need be made. Significant amounts of horizontal and vertical bony dimension is able to be achieved with GBR. GBR has been shown to equal intraoral onlay grafting with respect to the amount of bone regenerated.16 Additionally, investigations do not demonstrate resorption of or lower implant survival in regenerated bone over time, when comparing GBR to onlay grafting.11,13 Block grafts are not by and large superior to GBR; the evidence simply fails to support that statement.
Current Implant Trends
As modern implantology is prosthetic-driven, recent innovations affect the design, ease, esthetics and, in particular, the rapidity of fixture restoration (Table 2). Professionally and publicly, there appears to be a trend to speeding the process of implant treatment. Faster treatment has both risks and benefits. An examination of the process follows.
Immediate Implantation with Immediate Temporization
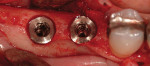
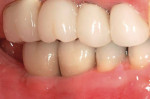
Immediate loading of an edentulous mandible is not a new concept. However, full-arch or full-mouth edentulation followed by immediate implantation and temporization is, both to the literature and the clinic (Figure 6; Figure 7; Figure 8).17,18 Because of its cursory appeal, a number of implant distributors offer their own versions of the immediate surgery-and-load protocol and advertise full-mouth reconstruction executed in one appointment to the public. It must be noted that hours of judicious multidisciplinary diagnosis, planning, discussion, and laboratory work prior to the surgical appointment are needed, regardless of the corporate protocol used. In the end, case selection becomes the limiting factor. The patient requires a favorable occlusal scheme, sufficient bone to ensure primary stability, good health and a lack of parafunctional habits, among other traits. There is a trend in the direction of this treatment. While early results are promising, this protocol is currently not the standard of care.
Abutments
Abutment fabrication has and continues to undergo significant metamorphosis. Many abutment options exist: standard machined titanium, standard machined gold, standard ceramic, custom made gold abutments (eg, UCLA) and computer-aided design/computer-aided manufacturing (CAD/CAM) titanium abutments. From a practical stance, implant success criteria include not only stability and function but also esthetic harmony. Depending on the tissue thickness, implant location, and bone level, a standard titanium abutment may appear gray through the mucosa. One solution involves use of abutments constructed from gold or ceramic. In certain cases the soft tissue will appear healthier and more esthetic.19-22 Improved appearance in the presence of a thin mucosa is the goal.
If the implant angulation falls short of ideal, a prefabricated straight or angled abutment may not compensate for off-axis orientation. Custom-made abutments, whether processed by the clinician or industrially, ease the restorative procedure and perfect results. Technology exists that reduces clinical work but still generates an abutment tailored to the individual. Some manufacturers offer CAD/CAM implant prosthetics (Figure 9; Figure 10; Figure 11).23,24 In this process, the company typically requires only a fixture-level index or a healing abutment or implant-level impression. The surgeon or restorative dentist sends that and the appropriate casts to a company laboratory, which fabricates a custom abutment.
Platform Switching
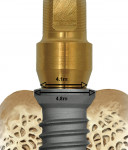
The interface between the abutment and implant, or the microgap, is subject to micromovement and bacterial seeding, and if it lies at or below the crest of the bone, prompts osseous resorption for those reasons.25 An alternate design for the two-stage implant is platform switching, which is achieved by aligning a relatively wide implant platform to a comparatively narrow abutment and medializes the microgap, thus removing the interface from direct contact with the bone (Figure 12; Figure 13; Figure 14; Figure 15). With possible movement and infection compartmentalized more or less to only the soft tissue, less crestal resorption results. Clinical studies that employ this implant–abutment configuration observe reduced vertical bone loss, even after function.26 Although a concept that garnered investigation only recently, platform switching data accumulates and shows potential.
Thread Modification
To enhance the fixture surface area, one may increase thread pitch, alter thread morphology, or augment surface roughness. At present, manufacturers fabricate implants that vary in at least one of the above characteristics. For example, integration of fluoride onto the implant surface boosts mineralization and attracts bone-forming cells.27 Rather than exhibiting threads of similar size spaced evenly along the body, some implants possess microthreads at their coronal segments (body or collar) to amplify surface area—and, in turn, potential BIC—at the crestal bone. Correspondingly, other designs increase the area covered by a roughed surface, including even the collar. Another construct exhibits a scalloped body that abuts the curve of the interdental bone and in doing so, keeps it from resorbing. All of these implant morphologies have little scientific evidence to substantiate claims of greater BIC, stability, or long-term survival but in some instances, previous versions of implants without these attributes are no longer available.
Conclusion
The usefulness of the implant trends discussed above is yet to be determined. With time, some of these innovations may become conventions; others will end up historical sidenotes. As always, we rely on biologic principles as well as longer-term clinical investigation to guide our judgment. Is a new design logical? Is it practical? Does it resolve or merely mask surgical or restorative problems? Most importantly, does it surpass significantly what is available? Clinicians, as individuals and as a collective, must be able to discriminate between a fad and a breakthrough, to separate the wheat from the chaff.
References
1. Ring ME. A thousand years of dental implants: a definitive history–part 1. Compend Contin Educ Dent. 1995;16(10):1060, 1062, 1064.
2. Ring ME. A thousand years of dental implants: a definitive history–part 2. Compend Contin Educ Dent. 1995;16(11):1132, 1134, 1136.
3. Linkow LI. The blade ventæa new dimension in endosseous implantology. Dent Concepts. 1968;11(2):3-12.
4. Brånemark PI, Hansson BO, Adell R, et al. Osseointegrated implants in the treatment of the edentulous jaw. Experience from a 10-year period. Scand J Plast Reconstr Surg Suppl. 1977;16:1-132.
5. Carlsson L, Rostlund T, Albrektsson B, et al. Removal torques for polished and rough titanium implants. Int J Oral Maxillofac Implants. 1988;3(1):21-24.
6. Gotfredsen K, Berglundh T, Lindhe J. Anchorage of titanium implants with different surface characteristics: an experimental study in rabbits. Clin Implant Dent Relat Res. 2000;2(3):120-128.
7. Ivanoff CJ, Hallgren C, Widmark G, et al. Histologic evaluation of the bone integration of TiO(2) blasted and turned titanium microimplants in humans. Clin Oral Implants Res. 2001;12(2):128-134.
8. Collaert B, De Bruyn H. Comparison of Brånemark fixture integration and short-term survival using one-stage or two-stage surgery in completely and partially edentulous mandibles. Clin Oral Implants Res. 1998;9(2):131-5.
9. Del Fabbro M, Testori T, Francetti et al. Systematic review of survival rates for immediately loaded dental implants. Int J Periodontics Restorative Dent. 2006;26(3): 249-263.
10. Esposito M, Worthington HV, Thomsen P, Coulthard P. Interventions for replacing missing teeth: different times for loading dental implants. Cochrane Database Syst Rev. 2004;(3):CD003878.
11. Fiorellini JP, Nevins ML. Localized ridge augmentation/preservation. A systematic review. Ann Periodontol. 2003;8(1): 321-327.
12. Wallace SS, Froum SJ. Effect of maxillary sinus augmentation on the survival of endosseous dental implants. A systematic review. Ann Periodontol. 2003;8(1): 328-343.
13. Hammerle CH, Jung RE, Feloutzis A. A systematic review of the survival of implants in bone sites augmented with barrier membranes (guided bone regeneration) in partially edentulous patients. J Clin Periodontol. 2002;29(Suppl 3):226-231.
14. Simion M, Jovanovic SA, Tinti C, et al. Long-term evaluation of osseointegrated implants inserted at the time or after vertical ridge augmentation. A retrospective study on 123 implants with 1-5 year follow-up. Clin Oral Implants Res. 2001;12(1):35-45.
15. Cordaro L, Amade DS, Cordaro M. Clinical results of alveolar ridge augmentation with mandibular block bone grafts in partially edentulous patients prior to implant placement. Clin Oral Implants Res. 2002;13(1):103-111.
16. Coulthard P, Esposito M, Jokstad A, Worthington HV. Interventions for replacing missing teeth: bone augmentation techniques for dental implant treatment. Cochrane Database Syst Rev. Database 2006;(1):CD003607.
17. van Steenberghe D, Glauser R, Blomback U, et al. A computed tomographic scan-derived customized surgical template and fixed prosthesis for flapless surgery and immediate loading of implants in fully edentulous maxillae: a prospective multicenter study. Clin Implant Dent Relat Res. 2005;7(Suppl 1): S111-S120.
18. Lazzara RJ, Testori T, Meltzer A, Misch et al. Immediate Occlusal Loading (IOL) of dental implants: predictable results through DIEM guidelines. Pract Proced Aesthet Dent. 2004;16(4):3-15.
19. Andersson B, Glauser R, Maglione M, et al. Ceramic implant abutments for short-span FPDs: a prospective 5-year multicenter study. Int J Prosthodont. 2003;16(6):640-646.
20. Gehrke P, Dhom G, Brunner J, et al. Zirconium implant abutments: fracture strength and influence of cyclic loading on retaining-screw loosening. Quintessence Int. 2006;37(1): 19-26.
21. Glauser R, Sailer I, Wohlwend A, et al. Experimental zirconia abutments for implant-supported single-tooth restorations in esthetically demanding regions: 4-year results of a prospective clinical study. Int J Prosthodont. 2004;17(3):285-290.
22. Tan PL, Dunne JT Jr. An esthetic comparison of a metal ceramic crown and cast metal abutment with an all-ceramic crown and zirconia abutment: a clinical report. J Prosthet Dent. 2004;91(3):215-218.
23. Grossmann Y, Pasciuta M, Finger IM. A novel technique using a coded healing abutment for the fabrication of a CAD/CAM titanium abutment for an implant-supported restoration. J Prosthet Dent. 2006;95(3):258-261.
34. Tselios N, Parel SM, Jones JD. Immediate placement and immediate provisional abutment modeling in anterior single-tooth implant restorations using a CAD/CAM application: a clinical report. J Prosthet Dent. 2006;95(3):181-185.
35. Hermann JS, Buser D, Schenk RK, et al. Crestal bone changes around titanium implants. A histometric evaluation of unloaded non-submerged and submerged implants in the canine mandible. J Periodontol. 2000;71(9): 1412-1424.
36. Chou CT, Morris HF, Ochi S, Walker L, et al. AICRG, Part II: Crestal bone loss associated with the Ankylos implant: loading to 36 months. J Oral Implantol. 2004;30(3):134-143.
37. Ellingsen JE, Thomsen P, Lyngstadaas SP. Advances in dental implant materials and tissue regeneration. Periodontol 2000.2006;41:136-156.
About the Author
Michael Sonick, DMD
Private Practice
Fairfield, Connecticut