Improving Endodontic Success Through Coronal Leakage Prevention
Gregori M. Kurtzman, DDS, MAGD, FACD, DICOI
Endodontic failure has been associated with coronal leakage within the canal system following obturation. The literature suggests that coronal leakage is more likely a determinant of clinical success or failure than apical leakage.1 Recent advances in resin obturation materials have been shown to provide superior sealing of the canal system, but without addressing the coronal aspect of the tooth, endodontic failure may occur. Studies confirm that a sound coronal seal is of paramount importance to the overall success of root canal treatment.2,3 Regardless of the obturation method, the best rule is that a properly cleaned, shaped, and obturated tooth should be permanently restored as soon as possible.4
No matter what our intentions following obturation of the canal system, patients may delay restoration of the treated tooth. Financial and time constraints often influence when the final restoration is completed. Between visits, an adhesive material prevents leakage and contamination of the canal.
Coronal leakage
No matter what clinicians place in the canal, if the coronal portion of the tooth is not sealed with materials that bond to tooth structure and are resistant to dissolution by oral fluids, then, over time, endodontic failure may be inevitable.
It is not unusual for a patient to present with decay at the margin of a crown of a tooth that had prior endodontic therapy. Because the tooth was treated endodontically, sensitivity that might indicate a problem under the crown will not be present to alert the patient to seek dental care. Coronal leakage for even a minimal amount of time may quickly lead to apical migration of bacteria. When such a patient does present, coronal leakage may have been ongoing for an extended period of time, thereby complicating treatment or rendering the tooth non-restorable and in need of extraction.
Studies indicate that significant coronal dye and bacterial leakage following exposure of sealed root canals to artificial and natural saliva, leading to complete bacterial leakage, may occur within 2 days.5 Supported by an in vitro study, it has been shown that dye leakage can occur in as few as 3 days.6 It has been suggested that gutta percha does not offer an effective barrier to crown-down leakage when exposed to the oral environment.7 Additional studies indicate that gutta percha will allow bacterial leakage, but the use of an adhesive sealer can significantly slow or stop coronal-apical bacterial migration.8
The predominant bacteria found in root-filled teeth with coronal leakage and persistent apical periodontitis is the Gram-positive facultative anaerobe, Staphylococcus. This is followed by the groups Streptococcus and Enterococcus, all normal salivary flora.9 Coronal leakage provides a constant source of microorganisms and nutrients that initiate and maintain periradicular inflammation and may well be the largest cause of failure in endodontic therapy.10
Endodontic obturation materials do not prevent coronal microleakage for an indefinite period of time.11 In a sample of 937 root-filled teeth that had not received restorative treatment during the previous year, the data show that the technical standard of coronal restoration and root filling were essential to periapical health.12 It is not uncommon for coronal leakage to occur after root canal treatment as a result of the presence of deficient composite resin fillings and secondary caries under the restorations.13
Yet, the endodontic materials that have been utilized over the past 50 years have shown that they do not prevent coronal leakage when challenged. In another investigation, 45 root canals were cleaned, shaped, and then obturated with gutta percha and root canal sealer using a lateral condensation technique. The coronal portions of the root-filling materials were placed in contact with Staphylococcus epidermidis and Proteus vulgaris. The number of days required for these bacteria to penetrate the entire root canals was determined. More than 50% of the root canals were completely contaminated after a 19-day exposure to S. epidermidis. Fifty percent of the root canals were also totally contaminated when the coronal surfaces of their fillings were exposed to P. vulgaris for 42 days.14 When comparing an epoxy resin sealer (e.g., AH 26) and other commonly used sealers after a 45-day exposure to the oral cavity, none of the sealers was capable of preventing leakage and coronal dye penetration.15 Therefore, the qualities of both the coronal restoration and obturation material are essential to periapical health because none of the present-day root canal sealers may hermetically seal “the root canal wall/ gutta percha filling interface”. In this respect, the importance of perfectly sealing both temporary and permanent coronal restorations must be emphasized.16
Pre-Endodontic Therapy Build-ups (i.e., Canal Projection)
Coronal leakage is a major contributor to endodontic failure.17 A bonded core placed prior to disinfection and obturation of the canal system of the tooth can greatly diminish the potential for leakage during and after endodontic therapy.
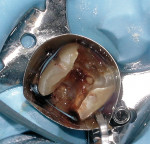
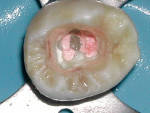
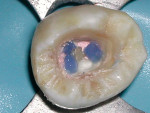
Isolation of the pulp chamber can be a challenging task when minimal coronal structure remains and endodontic therapy is required as part of the oral rehabilitation (Figure 1). Coronal reinforcement has traditionally been addressed after the endodontic phase. However, a coronal bonded build-up can simplify the endodontic phase and strengthen the tooth, decreasing the possibility of further damage to the tooth as a result of the dam clamp or mastication before a full-coverage restoration can be placed. Use of a canal Projector® cone (CJM Engineering, Santa Barbara, CA) allows isolation of the individual canals by surrounding them with a resin build-up (Figure 2). Sealing the pulpal floor and area surrounding the canal orifices will also decrease potential coronal leakage during and after endodontic treatment.
After identifying the canal orifices and removing caries, a canal Projector cone is placed on a hand file and inserted into each canal. A dentin adhesive is placed on all exposed surfaces and light-cured. Then, a dual-cure build-up material is injected around the Projector cones. When the build-up material has been completely set, the hand files and Projectors can be removed, leaving straight-line access into each individual canal. Visualization of the orifice is elevated to the occlusal plane, rather than deep within the tooth, and a bonded seal coronally around each orifice is achieved. Should the restoring dentist wish to place posts into the tooth, post space preparation is simplified, and misdirection of the post preparation is minimized.
Coronal Restoration (i.e., Access Sealing)
Microorganisms can penetrate through different temporary restorative materials and, supposedly, well-obturated root canals. The use of adhesive sealers may play an important role by minimizing coronal leakage. In addition, the importance of an immediate, definitive coronal seal should be emphasized after obturation of the canal system.18-20
Seventy extracted single-rooted mandibular premolars were studied to determine the length of time needed for bacteria present in natural human saliva to penetrate through 3 commonly used temporary restorative materials, as well as the entire root canal system obturated with the lateral condensation technique. The average time for broth contamination of access cavities closed with gutta percha was 7.85 days; with Intermediate Restorative Material (IRM), 12.95 days; and Cavit™-G (3M™ ESPE™, St. Paul, MN), 9.80 days. These results indicated that even in the short periods of time normally permitted between visits, complete leakage may result. IRM, long a common temporary material, was shown to leak to a significantly higher degree than glass ionomers.21 Because of its adhesive nature, glass ionomer cement may prevent bacterial penetration to the periapex of root-filled teeth over a 1-month period, as compared to IRM or Cavit temporary restorations.22
Another important consideration regarding the temporary restoration’s ability to prevent coronal leakage is how the material responds under mechanical load and thermocycling. Non-adhesive temporaries demonstrate an increased percentage of marginal breakdown and increased microleakage after thermocycling and loading. No significant improvement has been shown with increased thickness of the temporary material.23-25 When crowns were sealed with IRM, recontamination was detected within 13.5 days in the canals medicated with chlorhexidine, after 17.2 days in the group medicated with calcium hydroxide (CaOH2), and after 11.9 days in the group medicated with both chlorhexidine and CaOH2. The group with no medication but sealed with IRM showed recontamination after 8.7 days. There were statistically significant differences between the teeth with and without coronal seal. The coronal seal delayed—but did not prevent—leakage of microorganisms.26 Other studies confirm that IRM began to leak after 10 days, whereas Cavit and Dyract® (Dentsply Caulk, Milford, DE) leaked after 2 weeks.27
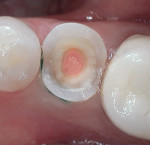
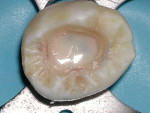
The use of a resin-based temporary restorative material or glass ionomer over partially removed resin composite restorations could be beneficial in achieving better resistance to marginal leakage (Figure 3). Maintaining partially removed permanent restorations does not seem to cause a problem in achieving a marginal seal.28 Glass ionomers provided a statistically better coronal seal than bonded composite or a bonded amalgam for preventing bacterial apical migration.29 This may be the result of the glass ionomers’ ability to adhere to the sclerotic dentin found on the pulpal floor better than adhesive resins. The key seems to be that if the coronal bacteria are locked out, the apical area will heal (Figure 4 and Figure 5).
Since its introduction a few years ago, Mineral Trioxide Aggregate (MTA) has been advocated as a sealing material, especially when perforation has occurred. However, an investigation found that mild inflammation was observed in 17% of the roots with an orifice plug and 39% without a plug. Without the development of severe inflammation, the sealing efficacy of MTA orifice plugs could not be determined.30
Should amalgam be the material of choice for the dentist, a bonded amalgam has been shown to produce significantly less leakage than non-bonded amalgams. To prevent the reinfection of the endodontically treated molar, it may be preferable to restore the tooth immediately after obturation by employing a bonded amalgam, coronal-radicular technique.31 Core build-up or access closure with adhesive materials has shown good long-term leakage resistance. The sandwich technique (i.e., a glass ionomer base with overlaying composite) and the composite resin restorations allowed significantly less coronal leakage than glass ionomer cement restorations, possibly because the composite resin prevents salivary dissolution of the glass ionomer long term.32
Results indicate that the sealing ability of adhesive and flowable materials can decrease coronal leakage potential.33,34 It is more prudent to use a permanent restorative material for provisional restorations in order to prevent inadequate canal sealing and the resulting risk of fluid penetration.35 To minimize the potential of perforation when re-entering the tooth to either place a post or re-treat endodontically, placement of a contrasting colored resin over each orifice may be beneficial, after which the entire pulpal floor is covered with a tooth-colored flowable resin (Figures 6, Figure 7 and Figure 8). These are available in a multitude of easily identifiable colors, such as pink (PermaFlo® Pink, Ultradent Products, Inc., South Jordan, UT), purple (PermaFlo® Purple), dark red (Flow-It™ ALC™/gingival dark, Pentron Clinical Technologies, LLC, Wallingford, CT), or dark blue (Den-Mat Corp., Santa Maria, CA).
Much emphasis is placed on the quality of the final restoration when dealing with coronal microleakage, and intracanal posts are frequently used for the retention of coronal restorations. Many authors have examined coronal microleakage in terms of gutta percha root fillings and coronal restorations, but few have investigated the coronal seal provided by various post systems. The seal provided by a cemented post depends upon the seal of the cement used. It appears that the dentin bonding cements (i.e., adhesive resins and glass ionomers) demonstrate less microleakage than the traditional, non-dentin bonding cements (i.e., zinc phosphates and polycarboxolates).36 Resin-supported polyethylene fiber and glass fiber dowels showed the lowest coronal leakage when compared to stainless steel and zirconia dowels. This may be the result of better adhesion of the luting agent to these resin-impregnated posts than to metal or ceramic posts, which do not allow adhesive penetration into the post surface. There were no significant differences between resin-supported polyethylene fiber and glass fiber dowels at any time period. The leakage measurements in zirconia dowel and stainless steel dowels were similar initially, but became significantly different at 3 and 6 months. The resin-supported polyethylene fiber dowels and glass fiber dowels tested exhibited less microleakage compared to zirconia dowel systems.37
Cleansing the Canal (Smear Layers)
Coronal sealing ability is not the only factor that influences the seal of the canal and prevents apical leakage. How well the sealer adheres to the canal walls is also important. The smear layer may prevent sealer penetration into the dentinal tubules. The frequency of bacterial penetration through teeth obturated with an intact smear layer (70%) was significantly greater than that of teeth from which the smear layer had been removed (30%). Removal of the smear layer enhanced sealability, as evidenced by increased resistance to bacterial penetration.38 The incidence of apical leakage was reduced in the absence of the smear layer, and the adaptation of gutta percha was improved regardless of which obturation method was used later.39-41 However, regardless of the obturation technique (e.g., thermoplastized, lateral or vertical condensation, or single cone), when a non-adhesive sealer was used, leakage increased after 30 days.42
While the material used is important, the manner in which the canal is prepared prior to obturation also determines how well the canal will be sealed when therapy is completed. Rotary instrumentation with nickel-titanium (NiTi) files has demonstrated less microleakage than hand instrument prepared canals, irrespective of what was used to obturate the canal.43 The machining of the canal walls with NiTi rotary instruments provides smoother canal walls and shapes that are easier to obturate than can be achieved with stainless steel files. The better the adaptation of the obturation material to the instrumented dentinal walls, the less leakage is expected along the entire root length. The better the canal walls are prepared, the more smear layer and organic debris is removed, which is beneficial to root canal sealing.
Smear layer removal is best achieved by irrigating the canals with sodium hypochlorite (NaOCL), followed by 17% ethylenediamine-tetraacetic acid (EDTA) solution.44 Whereas the NaOCL dissolves the organic component of the smear layer and exposes the dentinal tubules lining the canal walls, EDTA, a chelating agent, dissolves the inorganic portion of the dentin and opens the dentinal tubules. Alternating between the 2 irrigants as the instrumentation is being performed will enable removal of more organic debris further into the tubules, increasing resistance to bacterial penetration once the canal is obturated.45, 46
Obturation
The purpose of the obturation phase of an endodontic therapy is twofold: to prevent microorganisms from re-entering the root canal system and to isolate any microorganisms that may remain within the tooth from nutrients in tissue fluids. Regardless of how well clinicians seal the canal, if the coronal portion of the tooth is not thoroughly sealed, bacterial leakage may be inevitable. Accessory canals leading to the furcation area may be present in the pulp chamber. These may be additional sources of leakage that often go unaddressed either after obturation of the canals or during the restorative phase. Placement of a layer of resin-modified glass ionomer cement or adhesive resin immediately following obturation to seal this area can prevent leakage prior to the final restoration of the tooth.47 However, always remember that success will only be achieved if the root canal system has been as thoroughly debrided of infected material as possible. Irrigation is key to removing the smear layer that lines the canal walls.
The obturation material is a 2-pronged sword. Which sealer is used is as important as which core material is placed within the canal. Gutta percha demonstrates limitations in resistance to coronal leakage that have been overcome with newer resin alternatives. Although sealers can form close adhesion to the root canal wall, none is able to bond to the gutta percha core material. Upon setting, shrinkage of the sealer allows it to pull away from the gutta percha core, leaving a microgap through which bacteria may pass.48 Several alternatives are available for core material selection.
A resin gutta percha alternative (Resilon™: Pentron Clinical Technologies, LLC; and SybronEndo, Orange, CA) that is bondable with methacrylic sealers (e.g., Epiphany™, Pentron Clinical Technologies, LLC; and RealSeal™, SybronEndo) was introduced 3 years ago following extensive studies. The resin core material (Resilon) is available in ISO sized .02, .04, or .06 taper cones and as sized apical plugs (LightSpeed Technology, Inc., San Antonio, TX).49, 50
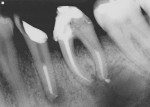
In studies performed at the University of North Carolina, the gutta percha group demonstrated leakage in 80% of specimens; the results were not dependant upon the obturation technique or sealer used.51 Based on these limitations with gutta percha, the seal of a coronal restoration may be as important as the gutta percha fill to prevent reinfection of the root canal. Studies have shown that bacterial leakage with Resilon is significantly reduced compared to gutta percha. The significance of this finding is that should the coronal aspect break down, the adhesive obturation material may slow or prevent apical migration of bacteria, allowing healing to occur (Figure 9 and Figure 10). An additional benefit when filling the canals with the new resin-based obturation material is an increase in fracture resistance. Endodontically treated single-canal extracted teeth were studied in vitro. Compared with standard gutta percha technique samples, Resilon demonstrated a 25% increase in root strength.52
Fiber obturators, an alternative core material, may be used when a post will be placed to strengthen the root and retain the coronal core. These allow obturation of the canal and placement of the post at the same time, ensuring a coronal seal.53,54 Microbial leakage occurred more quickly in lateral and vertical condensation techniques as compared with obturation with fiber obturation systems.55 Currently, 2 fiber obturator systems are commercially available: the FibreFill™ system (Pentron Clinical Technologies, LLC), which was introduced in 2001; and the recently available InnoEndo™ system (Heraeus Kulzer, Armonk, NY). Both systems use resin sealers, thereby allowing the formation of a monoblock across the root to strengthen and seal the canal system.
Sealer selection is also important for preventing microleakage and enabling a bond to the core material. Zinc oxide and eugenol (ZOE) sealers have been a mainstay in endodontic therapy for more than 100 years. When exposed to coronal leakage, ZOE sealers demonstrated complete leakage by the second day. Results indicate that none of the ZOE formulations tested could predictably produce a fluid-tight seal, even up to the fourth day.56
AH 26, an epoxy sealer originally introduced 40 years ago, was also unable to bond to gutta percha, thereby leading to coronal leakage issues. Leakage with AH 26 was not dependant upon obturation technique; gross leakage was shown to increase within the first 4 months after obturation when coronally challenged. Coronal leakage was significantly greater during the first 4 months.57 Complete bacterial leakage with AH 26 may be seen in as few as 8.5 weeks if the coronal restoration permits leakage.58
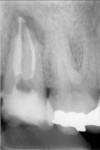
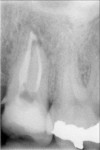
Additionally, in vitro studies found that gutta percha and AH 26 or AH Plus (Dentsply Maillefer, Tulsa, OK) permitted leakage of both bacteria and fungi. Leakage in experimental teeth occurred between 14 and 87 days, with 47% of the samples showing leakage. AH 26 sealer permitted bacterial leakage in 45% and fungi leakage in 60% of the samples. The samples with AH Plus demonstrated bacterial leakage in 50% and fungi in 55% of the samples. There was no statistically significant difference in penetration of bacteria and fungi between the 2 versions of the sealer.59 Comparative studies investigating periapical inflammation among teeth treated with gutta percha with AH 26 sealer and Resilon with methacrylic sealer found statistically less inflammatory response in the Resilon-treated teeth. Mild inflammation was observed in 82% of roots filled with gutta percha and AH 26 sealer, compared with 19% of the Resilon-treated teeth. The monoblock provided by the Resilon system was associated with less apical periodontitis, which may result from its superior resistance to coronal microleakage.60 Because AH 26 is unable to bond to gutta percha, polymerization shrinkage of the epoxy resin can cause a microgap, thereby leading to the leakage reported in the literature (Figure 11). Alternatively, the bond reported between the methacrylic sealer (Epiphany or RealSeal) and Resilon is sufficient to prevent microgap formation as the sealer polymerizes (Figure 12).
Recently completed electrophoresis leakage studies at the University of Maryland comparing gutta percha with AH 26 sealer to Resilon with Epiphany sealer found significant differences in leakage resistance. The gutta percha/AH 26 group demonstrated an average resistance of 404.6 micro amps, with 100% of the samples leaking, compared to an average resistance of 27.7 micro amps in the Resilon/ Epiphany group, with 60% showing some leakage. The lower the value of resistance in micro amps, the more resistant the specimen was to leakage.61 These results support other studies that indicate that, when challenged, gutta percha and AH 26 do not offer resistance to coronal leakage. Should the practitioner wish to continue using these materials, a permanent restoration should be placed at the appointment when endodontic therapy is completed.
Conclusion
Of 41 articles published between 1969 and 1999 (the majority of which are from the 1990s), the literature suggests that the prognosis of root canal treated teeth can be improved by sealing the canal and minimizing the leakage of oral fluids and bacteria into the periradicular areas as soon as possible after the completion of root canal therapy.62 Endodontic success is a multifactoral issue. Like a jigsaw puzzle, the full picture can only be seen when all the pieces are fit together. How the canals are instrumented is as important as what is used to obturate the canal system. This is also influenced by what is placed coronally and when the coronal aspect is sealed. NiTi rotary instruments and an irrigation protocol that includes NaOCL and EDTA will maximize the sealing ability of glass ionomers or the newer methacrylic resin sealers. The last piece of the puzzle (i.e., sealing coronally) should be performed with permanent adhesive restorative materials immediately at the conclusion of the first endodontic appointment to prevent apical migration of bacteria and ensure sealability of the canals.
Disclosure
The author has received an honorarium from Pentron Clinical Technologies, LLC.
References
1. Sritharan A. Discuss that the coronal seal is more important than the apical seal for endodontic success. Aust Endod J. 2002; 28(3):112-5.
2. Begotka BA, Hartwell GR. The importance of the coronal seal following root canal treatment. Va Dent J. 1996;73(4):8-10.
3. Siqueira JF Jr, Rocas IN, Favieri A, et al. Bacterial leakage in coronally unsealed root canals obturated with 3 different techniques. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;90(5):647-50.
4. Pommel L, Camps J. In vitro apical leakage of system B compared with other filling techniques. J Endod. 2001;27(7): 449-51.
5. Khayat A, Lee SJ, Torabinejad M. Human saliva penetration of coronally unsealed obturated root canals. J Endod. 1993;19(9):458-61.
6. Swanson K, Madison S. An evaluation of coronal microleakage in endodontically treated teeth. Part I. Time periods. J Endod. 1987; 13(2):56-9.
7. Cohen S, Burns R. Pathways to the Pulp. 8th ed. New York: CV Mosby; 2001.
8. Britto LR, Grimaudo NJ, Vertucci FJ. Coronal microleakage assessed by polymicrobial markers. J Contemp Dent Pract. 2003; 4(3):1-10.
9. Adib V, Spratt D, Ng YL, et al. Cultivable microbial flora associated with persistent periapical disease and coronal leakage after root canal treatment: a preliminary study. Int Endod J. 2004;37(8):542-51.
10. Leonard JE, Gutmann JL, Guo IY. Apical and coronal seal of roots obturated with a dentine bonding agent and resin. Int Endod J. 1996;29(2):76-83.
11. Pisano D, DiFiore P, McClanahan S, et al. Intraorific sealing of gutta-percha obturated root canal to prevent coronal microleakage. J Endod. 1998;24(10):609-62.
12. De Moor R, Coppens C, Hommez G. Coronal leakage reconsidered. Rev Belge Med Dent. 2002;57(3):161-85.
13. Chong BS. Coronal leakage and treatment failure. J Endod. 1995;21(3):159-60.
14. Torabinejad M, Ung B, Kettering JD. In vitro bacterial penetration of coronally unsealed endodontically treated teeth. J Endod. 1990;16(12):566-9.
15. Kopper PM, Figueiredo JA, Della Bona A, et al. Comparative in vivo analysis of the sealing ability of three endodontic sealers in post-prepared root canals. Int Endod J. 2003;36(12):857-63.
16. De Moor R, Hommez G. The importance of apical and coronal leakage in the success or failure of endodontic treatment. Rev Belge Med Dent. 2000;55(4):334-44.
17. Kurtzman GM. Restoring teeth with severe coronal breakdown as a prelude to endodontic therapy. Endodontic Therapy. 2004;4(1): 21-2.
18. Imura N, Otani SM, Campos MJA, et al. Bacterial penetration through temporary restorative materials in root-canal-treated teeth in vitro. Int Endod J. 1997;30(6): 381-5.
19. Uranga A, Blum JY, Esber S, et al. A comparative study of four coronal obturation materials in endodontic treatment. J Endod. 1999;25(3):178-80.
20. Fox K, Gutteridge DL. An in vitro study of coronal microleakage in root-canal-treated teeth restored by the post and core technique. Int Endod J. 1997;30(6):361-8.
21. Barthel CR, Zimmer S, Wussogk R, et al. Long-term bacterial leakage along obturated roots restored with temporary and adhesive fillings. J Endod. 2001;27(9):559-62.
22. Barthel CR, Strobach A, Briedigkeit H, et al. Leakage in roots coronally sealed with different temporary fillings. J Endod. 1999; 25(11):731-4.
23. Mayer T, Eickholz P. Microleakage of temporary restorations after thermocycling and mechanical loading. J Endod. 1997;23(5): 320-2.
24. Deveaux E, Hildelbert P, Neut C, et al. Bacterial microleakage of Cavit, IRM, and TERM. Oral Surg Oral Med Oral Pathol. 1992;74(5):634-43.
25. Deveaux E, Hildelbert P, Neut C, et al. Bacterial microleakage of Cavit, IRM, TERM, and Fermit: a 21-day in vitro study. J Endod. 1999;25(10):653-9.
26. Gomes BP, Sato E, Ferraz CC, et al. Evaluation of time required for recontamination of coronally sealed canals medicated with calcium hydroxide and chlorhexidine. Int Endod J. 2003;36(9):604-9.
27. Balto H. An assessment of microbial coronal leakage of temporary filling materials in endodontically treated teeth. J Endod. 2002;28(11):762-4.
28. Tulunoglu O, Uctasli MB, Ozdemir S. Coronal microleakage of temporary restorations in previously restored teeth with amalgam and composite. Oper Dent. 2005;30(3):331-7.
29. Nup C, Boylan R, Bhagat R, et al. An evaluation of resin-ionomers to prevent coronal microleakage in endodontically treated teeth. J Clin Dent. 2000;11(1):16-9.
30. Mah T, Basrani B, Santos JM, et al. Periapical inflammation affecting coronally-inoculated dog teeth with root fillings augmented by white MTA orifice plugs. J Endod. 2003;29(7):442-6.
31. Howdle MD, Fox K, Youngson CC. An in vitro study of coronal microleakage around bonded amalgam coronal-radicular cores in endodontically treated molar teeth. Quintessence Int. 2002;33(1):22-9.
32. Kleitches AJ, Lemon RR, Jeansonne BG. Coronal microleakage in conservatively restored endodontic access preparations. J Tenn Dent Assoc. 1995;75(1):31-4.
33. Shindo K, Kakuma Y, Ishikawa H, et al. The influence of orifice sealing with various filling materials on coronal leakage. Dent Mater J. 2004;23(3):419-23.
34. de Souza FD, Pecora JD, Silva RG. The effect on coronal leakage of liquid adhesive application over root fillings after smear layer removal with EDTA or Er:YAG laser. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2005;99(1):125-8.
35. Uranga A, Blum JY, Esber S, et al. A comparative study of four coronal obturation materials in endodontic treatment. J Endod. 1999;25(3):178-80.
36. Ravanshad S, Ghoreeshi N. An in vitro study of coronal microleakage in endodontically-treated teeth restored with posts. Aust Endod J. 2003;29(3):128-33.
37. Usumez A, Cobankara FK, Ozturk N, et al. Microleakage of endodontically treated teeth with different dowel systems. J Prosthet Dent. 2004;92(2):163-9.
38. Behrend GD, Cutler CW, Gutmann JL. An in-vitro study of smear layer removal and microbial leakage along root-canal fillings. Int Endod J. 1996;29(2):99-107.
39. Karagoz-Kucukay I, Bayirli G. An apical leakage study in the presence and absence of the smear layer. Int Endod J. 1994;27(2):87-93.
40. Saunders WP, Saunders EM. Influence of smear layer on the coronal leakage of Thermafil and laterally condensed gutta-percha root fillings with a glass ionomer sealer. J Endod. 1994;20(4): 155-8.
41. Gencoglu N, Samani S, Gunday M. Dentinal wall adaptation of thermoplasticized gutta-percha in the absence or presence of smear layer: a scanning electron microscopic study. J Endod. 1993;19(11):558-62.
42. Pommel L, Camps J. In vitro apical leakage of system B compared with other filling techniques. J Endod. 2001;27(7):449-51.
43. von Fraunhofer JA, Fagundes DK, McDonald NJ, et al. The effect of root canal preparation on microleakage within endodontically treated teeth: an in vitro study. Int Endod J. 2000;33(4): 355-60.
44. Behrend GD, Cutler CW, Gutmann JL. An in-vitro study of smear layer removal and microbial leakage along root-canal fillings. Int Endod J. 1996;29(2):99-107.
45. Clark-Holke D, Drake D, Walton R, et al. Bacterial penetration through canals of endodontically treated teeth in the presence or absence of the smear layer. J Dent. 2003;31(4):275-81.
46. Vivacqua-Gomes N, Ferraz CC, Gomes BP, et al. Influence of irrigants on the coronal microleakage of laterally condensed gutta-percha root fillings. Int Endod J. 2002; 35(9):791-5.
47. Carrotte P. Endodontics: Part 8. Filling the root canal system. Br Dent J. 2004;197(11): 667-72.
48. Teixeira FB, Teixeira EC, Thompson J, et al. Dentinal bonding reaches the root canal system. J Esthet Restor Dent. 2004;16(6): 348-54.
49. Maggio JD. RealSeal—the real deal. Compend Contin Educ Dent. 2004;25(10A):834, 836.
50. Chivian N. Resilon—the missing link in sealing the root canal. Compend Contin Educ Dent. 2004;25(10A): 823-4, 826.
51. Shipper G, Orstavik D, Teixeira FB, et al. An evaluation of microbial leakage in roots filled with a thermoplastic synthetic polymer-based root canal filling material (Resilon). J Endod. 2004;30(5):342-7.
52. Teixeira FB, Teixeira EC, Thompson JY, et al. Fracture resistance of roots endodontically treated with a new resin filling material. J Am Dent Assoc. 2004;135(5):646-52.
53. Kurtzman GM, Jones OJ, Lopez L. Predictable endodontics: A fiber reinforced adhesively bonded endodontic obturator and post system. Endodontic Therapy. 2003;3(1):16-21.
54. Kurtzman GM, Jones OJ, Lopez L. Fiberfill: A fiber reinforced adhesively bonded endodontic obturator and post system. Oral Health Journal. 2003;1(1):6-10.
55. Shipper G, Trope M. In vitro microbial leakage of endodontically treated teeth using new and standard obturation techniques. J Endod. 2004;30(3):154-8.
56. Tewari S, Tewari S. Assessment of coronal microleakage in intermediately restored endodontic access cavities. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2002; 93(6):716-9.
57. De Moor RJ, Hommez GM. The long-term sealing ability of an epoxy resin root canal sealer used with five gutta percha obturation techniques. Int Endod J. 2002;35(3): 275-82.
58. Chailertvanitkul P, Saunders WP, MacKenzie D, et al. An in vitro study of the coronal leakage of two root canal sealers using an obligate anaerobe microbial marker. Int Endod J. 1996;29(4):249-55.
59. Miletic I, Prpic-Mehicic G, Marsan T, et al. Bacterial and fungal microleakage of AH26 and AH Plus root canal sealers. Int Endod J. 2002;35(5):428-32.
60. Shipper G, Teixeira FB, Arnold RR, et al. Periapical inflammation after coronal microbial inoculation of dog roots filled with gutta-percha or resilon. J Endod. 2005;31(2):91-6.
61. von Fraunhofer JA, Kurtzman GM, Norby CE. Resin-based sealing of root canals in endodontic therapy. Gen Dent. 2005. Submitted for publication.
62. Heling I, Gorfil C, Slutzky H, et al. Endodontic failure caused by inadequate restorative procedures: review and treatment recommendations. J Prosthet Dent. 2002;87(6):674-8.
About the Author
Gregori M. Kurtzman, DDS, MAGD, FACD, DICOI
Assistant Clinical Professor
Department of Endodontics, Prosthodontics, and Operative Dentistry
University of Maryland College of Dental Surgery
Baltimore, MD
Private Practice
Silver Spring, MD
www.maryland-implants.com