Relative Performance of Antisensitivity Dentifrice, Rinse, and Oxalate Strips: An In Vitro Comparison of Common Global Over-the-Counter Products
Tiffany C. Hare, BS; Marianne Zsiska, PhD; Ying Boissy, BS; Matthew L. Barker, PhD; and Phillip A. Drake, PhD
Abstract
Objective: The objective of this investigation was to compare the effectiveness of several popular over-the-counter antisensitivity product technologies using a novel in vitro model.
Methods: Hydraulic conductance was measured before and after product application to human dentin specimens to quantitatively compare the magnitude and durability of tubule occlusion. Scanning electron microscopy (SEM) of the fractured cross-sections of treated dentin discs was also employed to compare morphology and extent of deposition within the tubuli. The following products were tested: a flexible strip coated with 1.5% oxalate gel; a rinse containing 1% oxalate; a rinse containing 0.8% arginine; a 0.454% stannous fluoride dentifrice; a dentifrice containing 8% strontium acetate; a dentifrice containing 8% arginine and calcium carbonate; a dentifrice containing 5% calcium sodium phosphosilicate; and an oxalate-free (placebo) strip.
Results: The oxalate-coated strip demonstrated markedly enhanced tubule occlusion relative to the dentifrice and arginine rinse products tested. In addition, the oxalate strip was shown to reduce hydraulic conductance much more efficiently—ie, with fewer applications—than an oxalate rinse. Of the other products tested, the stannous fluoride dentifrice showed the highest occlusion under conditions simulating daily treatment and challenge conditions.
Conclusion: The oxalate strip was significantly more effective at reducing fluid movement through human dentin than the other rinse and dentifrice products studied. In addition, flow resistance in strip-treated dentin was better maintained over a range of mechanical and chemical challenges.
The pain mechanism widely believed to be primarily responsible for dentin hypersensitivity involves a pulse of fluid movement within patent dentin tubuli, which directly or indirectly activates pulpal nerve fibers.1,2 The fluid pulse is a result of a momentary pressure differential, which may be induced by osmotic, thermal, or tactile stimuli. Tubuli are normally covered by enamel or cementum, but are often exposed by gingival recession or enamel erosion. Historically, the most popular over-the-counter (OTC) therapeutic options—eg, potassium nitrate—were designed to relieve sensitivity via direct action on the nerve. In recent years, development in the industry has focused more on occlusion of dentin tubuli to eliminate fluid movement. Some of the more popular OTC occluding agents reported to show clinical efficacy include stannous fluoride,3-9 strontium salts,10-15 calcium sodium phosphosilicate,16-19 and arginine/calcium carbonate.20-26 These OTC therapies generally rely on amorphous smear layers spread across the tubule orifice in order to reduce dentin transmission of pulpal fluid.
Clearly ascertaining the relative clinical efficacy of the plethora of available antisensitivity products is important, but it presents significant challenges to investigators. Controlled laboratory studies may yield valuable comparative information, but only if the in vitro model is physiologically relevant and represents a fair and unweighted comparison. An experimentally unbiased measure is especially important when the comparison spans different product delivery systems, such as rinses, dentifrices, prophy pastes, strips, sealants, etc. More specifically, the experimental configuration and measurement protocol for any meaningful comparison must: (1) enable product application as per labeled usage instruction; and (2) create a physiologically relevant microenvironment to ensure realistic interaction between the product and the dentin substrate.
Oxalate salts are somewhat unique among antisensitivity agents in that they occlude tubuli through classical crystal deposition. Although studied for decades,27-32 oxalate products have produced mixed results in reducing hypersensitivity clinically,33 arguably as a result of the product delivery system and application. To date, little emphasis has been given to optimization of oxalate deposition, specifically via exploitation of product contact time.
Purpose/Objective
The purpose of this study was to compare a new oxalate strip to several of the most popular OTC antisensitivity product technologies marketed globally. A novel in vitro model was developed in order to incorporate essential elements of the oral microenvironment and to enable direct product application according to labeled instructions.
Materials/Methods/Design
Preparation of Dentin Samples
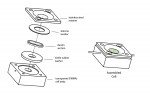
Coronal dentin samples approximately 0.8 mm in thickness were prepared by sectioning mandibular and maxillary third molars parallel to the occlusal surface, avoiding pulpal horns and coronal enamel. Sections were acid-etched and conditioned as previously described.34 Dentin samples were mounted between a liquid cell and tapered bushing, which allows pressurization with liquid from the cervical face of the dentin disc, while permitting access to the coronal surface for treatment (Figure 1). This configuration also enables controlled pressurization of the cell during treatment. The protocol to establish a pellicle on inner surfaces of tubuli and the quantitative flow measurement procedure have been previously described.34
Measurement of Hydraulic Conductance
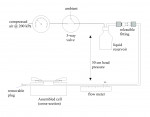
Quantitative flow measurements were made using the apparatus shown in Figure 2 during application of 200 kPa of liquid pressure using Hartmann’s solution (30 mM lactic acid, 2 mM CaCl2, 5 mM KCl, 100 mM NaCl, adjusted to pH 7 with NaOH) and a custom bubble-based flowmeter to obtain a volumetric flow rate.34 Flow was again measured after treatment, and flow reduction was expressed as a percentage of baseline flow, where the baseline flow rate equals 0% reduction and 100% reduction represents complete cessation of flow.
Treatment Protocol
A freshly prepared aliquot of 500 µl of a 25% wt:wt slurry of each dentifrice product was pipetted onto the dentin surface. A manual toothbrush, pre-rinsed with the dentifrice slurry, was used to brush the slurry into the surface for 10 seconds using circular strokes with approximately 100 N of force. The slurry concentration and application time for dentifrices were chosen to approximate salivary dilution and brush contact time experienced by an individual tooth in vivo. Rinses and the oxalate strip were applied according to the instructions on the product label. Prior to the treatment period, the dentin was flushed with a synthetic pulpal fluid (SPF) consisting of Hartmann’s solution to which bovine serum albumin (Sigma-Aldrich p/n A2153) was added to achieve a protein concentration of 1.2%. The SPF solution was allowed to flow through the dentin section under a 30-cm head (3 kPa) during the treatment period to simulate the outward flow of pulpal fluid that occurs in vivo.35-37
Challenge Protocol
Unless otherwise indicated, each application was followed by a mechanical challenge consisting of contact for 15 seconds with a sonic power toothbrush operated in the “Sensitive” brushing mode labeled for gentle cleaning. Under this protocol, each sample was subjected to three application/challenge cycles. During the 15-second challenge period, 100 kPa of liquid pressure was applied to simulate the osmotic pressures that might regularly be experienced in vivo in conjunction with diet and oral hygiene.
Separately, a second, more intensive treatment protocol was executed with the following alterations: (1) dentifrices were applied neat, using a pea-sized quantity brushed into the surface for 60 seconds, rotating the section every 15 seconds; and (2) application cycles were uninterrupted by a mechanical or acid challenge procedure until 14 cycles were complete. After the 14 application cycles, one sample from each group was removed for evaluation by SEM, and the remainder were subjected to a single challenge procedure consisting of 10 mM citric acid applied with a manual toothbrush for 60 seconds. While product performance—particularly for dentifrice products—was likely significantly exaggerated under these conditions, this protocol was largely effective in producing substantial smear layers, which could be readily characterized by scanning electron microscopy (SEM).
Product technologies that were evaluated are shown in Table 1. Products were labeled with abbreviated names corresponding to principal antisensitivity ingredients.
Imaging
Samples were placed in Hartmann’s solution for storage and transport to the imaging facility. Samples were prepared by blot-drying and then fracturing the dentin section between forceps. Fractured samples were affixed to a mount positioned at 45°, and were then sputter-coated with Au/Pd prior to SEM imaging at 3 kV. Elemental mapping data was obtained with excitation at 10 kV. The 45° mounting angle was found to be particularly useful in enabling visualization of tubuli cross-sections as well as the unfractured dentin surface.
Statistical Analysis
Groups were compared pairwise for percent flow reduction using a non-parametric Wilcoxon rank-sum test. This non-parametric test was used due to the feasible sample size (n = 4 per group for Protocol No. 1, n = 6 per group for Protocol No. 2). The significance level used for each pairwise treatment comparison was .05, and P values were not adjusted for the overall experiment-wise error rate. Summary statistics of means and standard errors were also calculated and provided in the figures. To quantify the change in percent flow reduction over multiple treatments with Protocol No. 1, slopes for percent reduction/application cycle were compared between groups using a general linear mixed model with a common random intercept over all groups. A larger positive slope indicated a faster reduction in hydraulic conductance with treatment.
Results
Protocol No. 1: Three Application/Mechanical Challenge Cycles
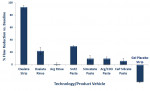
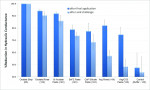
Effectiveness of the various occlusive therapies, as measured by in vitro flow reduction, varied by technology, protocol, and number of treatment cycles. Flow reduction at the completion of the experiment is shown in Figure 3. The oxalate strip demonstrated near-complete occlusion of the dentin to liquid flow after only three treatment/challenge cycles, which was markedly improved relative to the other OTC products after three treatment/challenge cycles. Mean flow reduction and statistical significance among products were as follows: 92.9% for oxalate strips (a), 29.7% SnF2 paste (b), 22.1% oxalate rinse (bc), 9.9% Arg/CO3 paste (cd), 9.9%
Sr acetate paste (cd), 5.9% CaP silicate paste (cd), 0.70% Arg rinse (d), and -28.7% for placebo strips (e). Groups with different letters (a-e) were significantly different (Wilcoxon exact test, P < .05).
Figure 4 shows flow reduction observed during each step of the protocol, ie, after each individual application and challenge step. As expected, flow was reduced during the treatment phase of each cycle. However, brief physical contact with the power toothbrush during the challenge phase was sufficient to significantly reverse the effects of treatment with some of the test products, increasing flow and reducing the net occlusive effect over the course of the experiment. The rate at which the oxalate strip occluded liquid flow, as measured by slope in a plot of percent flow reduction per application cycle, was significantly higher at 30.8%/cycle than response rates for all other treatments (P < .0001). The slope of the same response curve for the SnF2 paste was significantly higher at 12.2%/cycle (P < .03) than each of the products tested with the exception of the oxalate strip and oxalate rinse (12.2%/cycle). Slopes (percent flow reduction per cycle) were 5.7 for CaP silicate paste, 4.8 for Arg/CO3 paste, 4.6 for Sr acetate paste, 4.5 for Arg rinse, and -6.4 for placebo strips. For reference, the common estimated random intercept was -7.3 with a standard error of 2.7. Standard error for the slopes was 2.0.
Protocol No. 2: 14 Sequential Applications
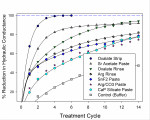
Figure 5 shows flow resistance observed in dentin sections after up to 14 back-to-back product applications, where no chemical or mechanical challenge was introduced between each treatment. While a reduction in hydraulic conductance was observed for all treatments, including the buffer control, the oxalate strip demonstrated statistically significant greater flow reduction after three treatments than all other products after 14 treatments (P <.05), with the exception of the oxalate rinse (P = .08). Following a single acid challenge at the completion of the treatment phase of the protocol (Figure 6), the flow resistance of the oxalate strip was significantly higher than for all other treatments without exception (P < .05).
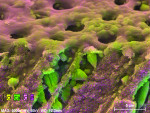
In most cases, no measurable liquid flow was detected in samples treated with more than four oxalate strips, indicating total occlusion of tubuli. Features of oxalate crystals deposited by the oxalate strip may be clearly seen in Figure 7, which was collected at higher magnification. Figure 7 also contains superimposed elemental maps generated with energy-dispersive x-ray spectrometry for additional contrast between dentin mineral and oxalate crystals. Figure 8 through Figure 15 show SEM images of fractured dentin surfaces following back-to-back product application of each of the products tested and an untreated control. Most of the tested products produced visible smear layers or other deposits under these conditions.
Discussion
A side-by-side in vitro comparison of occlusive antisensitivity products is an ambitious undertaking, particularly when the comparison spans different product delivery systems, eg, dentifrices, rinses, and strips. The design of the experimental apparatus was critical for such a comparison, because it enabled access to the dentin surface while it was mounted in the cell. This made possible: (1) direct application of products such as dentifrice and strips as per labeled usage instructions; and (2) application of biologically relevant fluid pressure during product application. In addition, an effort was made to replicate the in vivo biochemical environment through the use of simulated pulpal fluids and deposition of a protein pellicle on dentin surfaces. Given that dentifrice has historically been heavily represented in the OTC marketplace relative to other product forms, a special effort was made to span a range of dentifrice application conditions, eg, product dilution and duration of application. Consideration was also given to the nature and intensity of challenges that were designed to simulate daily activity that would lead to deterioration of therapeutic barriers.
Across the variety of experimental conditions described above, the performance of the oxalate gel–coated strip in vitro readily exceeded that of the common OTC antisensitivity therapies included in this evaluation. Specifically, the oxalate strip was significantly more effective in reducing fluid movement through dentin in terms of both magnitude and efficiency. In this context, efficiency was defined by the number of application cycles required to achieve complete occlusion.
Quantitative in vitro results also indicate that the oxalate gel–coated strip creates a flow barrier that is superior in resistance to many chemical and mechanical processes that may occur at the surface of exposed dentin in vivo. These natural processes may include mastication, exposure to dietary acids, abrasion from aggressive or improper toothbrushing, flexural forces associated with abfraction, and even periodic surges in outward pulpal flow associated with sensitivity pain. Although substantivity is challenging to study in vitro, it is also a critical property of the therapeutic layer, given that the post-application product benefit would be expected to deteriorate as rapidly as does the barrier.
The SEM images of dentin samples posttreatment (Figure 7 and Figure 9 through Figure 15) illustrate fundamental morphological differences between oxalate-based barriers and those created by other agents, eg, dentifrice smear layers. The oxalate barrier is uniquely composed of individual crystalline structures, many of which have dimensions that approach or match that of the tubule diameter. Importantly, most of the oxalate crystals are located just inside the lumen of the tubuli, approximately 5 µm to 25 µm from the orifice. Conversely, dentifrice smear layers appear primarily as a relatively superficial, amorphous composite. These differences in morphology are useful in explaining the observed differences in in vitro quantitative performance. Smear-layer aggregates maintained by adhesive forces between multiple particles might well be expected to be more susceptible to a mechanical or chemical challenge than would a proportionally large, homogeneous crystal. Additionally, the protected location of the oxalate crystals inside the tubuli should afford improved protection against abrasion and chemical attack.
Not surprisingly, significant differences in the extent of crystal deposition are evident after application of the oxalate rinse delivery system, which was applied for 60 seconds according to the product label, when compared with an equal number of applications of the oxalate strip, which was applied for 10 minutes per application. The strip as a delivery system protects the oxalate gel formulation from salivary dilution during the extended application time, maintaining oxalate concentration at the site of action and providing the necessary time to establish a robust crystalline barrier against liquid movement.
Conclusion
In an in vitro comparison of OTC antisensitivity products, an oxalate gel–coated strip was significantly more effective at reducing hydraulic conductance in human dentin under biologically relevant conditions than the marketed rinse and dentifrice products studied. In addition, the flow resistance induced by the oxalate strip was better maintained throughout a range of mechanical and chemical challenges.
Disclosure
This research was sponsored by Procter & Gamble.
About the Authors
Tiffany C. Hare, BS
Principal Researcher
Procter & Gamble
Mason, Ohio
Marianne Zsiska, PhD
Senior Scientist
Procter & Gamble
Mason, Ohio
Ying Boissy, BS
Principal Researcher
Procter & Gamble
Cincinnati, Ohio
Matthew L. Barker, PhD
Principal Statistician
Procter & Gamble
Mason, Ohio
Phillip A. Drake, PhD
Senior Scientist
Procter & Gamble
Mason, Ohio
References
1. Brännström M. Sensitivity of dentine. Oral Surg Oral Med Oral Pathol. 1966;21(4):517-526.
2. Ahlquist M, Franzen O, Coffey J, Pashley DH. Dental pain evoked by hydrostatic pressures applied to exposed dentin in man: A test of the hydrodynamic theory of dentin sensitivity. J Endod. 1994;20(3):130-134.
3. Absi EG, Addy M, Adams D. Dentine hypersensitivity: uptake of toothpastes onto dentine and effects of brushing, washing and dietary acid. J Oral Rehabil. 1995;22(3):175-182.
4. He T, Barker ML, Biesbrock A, Sharma N. A randomized controlled clinical trial to assess the desensitizing effect of a stannous fluoride dentifrice. Am J Dent. 2014;27(2):106-110.
5. Parkinson C, Hughes N, Jeffery P, et al. The efficacy of an experimental dentifrice containing 0.454% w/w stannous fluoride in providing relief from the pain of dentin hypersensitivity: an 8-week clinical study. Am J Dent. 2013;26(spec no A):25A-31A.
6. Schiff T, He T, Sagel L, Baker R. Efficacy and safety of a novel stabilized stannous fluoride and sodium hexametaphosphate dentifrice for dental hypersensitivity. J Contemp Dent Pract. 2006;7(2):1-8.
7. Schiff T, Saletta L, Baker RA, et al. Desensitizing effect of a stabilized stannous fluoride/sodium hexametaphosphate dentifrice. Compend Contin Educ Dent. 2005;26(9 suppl 1):35-40.
8. He T, Barker ML, Qaqish J, Sharma N. Fast onset sensitivity relief of a 0.454% stannous fluoride dentifrice. J Clin Dent. 2011;22(2):46-50.
9. He T, Cheng R, Biesbrock AR, et al. Rapid desensitizing efficacy of a stannous-containing sodium fluoride dentifrice. J Clin Dent. 2011;22(2):40-45.
10. Gedalia I, Brayer L, Kalter N, et al. The effect of fluoride and strontium application on dentine: in vivo and in vitro studies. J Periodontol. 1978;49(5):269-272.
11. Parkinson CR, Wilson RJ. A comparative in vitro study investigating the occlusion and mineralization properties of commercial toothpastes in a four-day dentin disc model. J Clin Dent. 2011; 22(3):74-81.
12. Mason S, Hughes N, Sufi F, et al. A comparative clinical study investigating the efficacy of a dentifrice containing 8% strontium acetate and 1040 ppm fluoride in a silica base and a control dentifrice containing 1450 ppm fluoride in a silica base to provide immediate relief of dentin hypersensitivity. J Clin Dent. 2010;21(2):42-48.
13. Karim BF, Gillam DG. The efficacy of strontium and potassium toothpastes in treating dentine hypersensitivity: a systematic review. Int J Dent. 2013;2013:573258. doi: 10.1155/2013/573258. Epub 2013 Apr 8.
14. West N, Newcombe RG, Hughes N, et al. A 3-day randomised clinical study investigating the efficacy of two toothpastes, designed to occlude dentine tubules, for the treatment of dentine hypersensitivity. J Dent. 2013;41(2):187-194.
15. Seong J, Macdonald E, Newcombe RG, et al. In situ randomised trial to investigate the occluding properties of two desensitising toothpastes on dentine after subsequent acid challenge. Clin Oral Investig. 2013;17(1):195-203.
16. Earl JS, Topping N, Elle J, et al. Physical and chemical characterization of surface layers formed on dentin following treatment with a fluoridated toothpaste containing NovaMin. J Clin Dent. 2011;22(3):68-73.
17. Earl JS, Leary RK, Muller KH, et al. Physical and chemical characterization of dentin surface following treatment with NovaMin technology. J Clin Dent. 2011;22(3):62-67.
18. Acharya AB, Surve SM, Thakur SL. A clinical study of the effect of calcium sodium phosphosilicate on dentin hypersensitivity. J Clin Exp Dent. 2013;5(1):e18-e22.
19. Surve SM, Acharya AB, Shetty A, Thakur SL. Efficacy of calcium sodium phosphosilicate in managing dentin hypersensitivity. Gen Dent. 2012;60(5):e308-e311.
20. Ayed F, Ayad N, Zhang YP, et al. Comparing the efficacy in reducing dentin hypersensitivity of a new toothpaste containing 8.0% arginine, calcium carbonate and 1450 ppm fluoride to a commercial sensitive toothpaste containing 2% potassium ion: an eight-week clinical study on Canadian adults. J Clin Dent. 2009;20(1):10-16.
21. Lavender SA, Petrou I, Heu R, et al. Mode of action studies on a new desensitizing dentifrice containing 8.0% arginine, a high cleaning calcium carbonate system and 1450 ppm fluoride. Am J Dent. 2010;23 spec no A:14A-19A.
22. Orsini G, Procaccini M, Manzoli L, et al. A 3-day randomized clinical trial to investigate the desensitizing properties of three dentifrices. J Periodontol. 2013;84(11):e65-e73.
23. Kapferer I, Pflug C, Kisielewsky I, et al. Instant dentin hypersensitivity relief of a single topical application of an in-office desensitizing paste containing 8% arginine and calcium carbonate: a split-mouth, randomized-controlled study. Acta Odontol Scand. 2013;71(3-4):994-999.
24. Kakar A1, Dibart S, Kakar K. Clinical assessment of a new dentifrice with 8% arginine and calcium carbonate on dentin hypersensitivity in an Indian population using a new measuring device: the Jay Sensitivity Sensor Probe. Am J Dent. 2013;26 spec no B:13B-20B.
25. Markowitz K. A new treatment alternative for sensitive teeth: a desensitizing oral rinse. J Dent. 2013;41 suppl 1:S1-S11.
26. Sharif MO, Iram S, Brunton PA. Effectiveness of arginine-containing toothpastes in treating dentine hypersensitivity: a systematic review. J Dent. 2013;41(6):483-492.
27. Greenhill JD, Pashley DH. The effects of densensitizing agents on the hydraulic conductance of human dentin in vitro. J Dent Res. 1981;60(3):686-698.
28. Pashley DH, Galloway SE. The effects of oxalate treatment on the smear layer of ground surfaces of human dentine. Arch Oral Biol. 1985;30(10):731-737.
29. Pereira JC, Segala AD, Gillam DG. Effect of densensitizing agents on the hydraulic conductance of human dentin subjected to different surface pre-treatments – an in vitro study. Dent Mater. 2005;21(2):129-138.
30. Sharma D, Hong C, Heipp P. A novel potassium oxalate-containing tooth-desensitising mouthrinse: a comparative in vitro study. J Dent. 2013;41(suppl 4):S18-S27.
31. Pashley DH, O’Meara JA, Kepler EE, et al. Dentin permeability. Effects of desensitizing dentifrices in vitro. J Periodontol. 1984;55(9):522-525.
32. Pashley DH. Dentin permeability, dentin sensitivity, and treatment through tubule occlusion. J Endod. 1986;12(10):465-474.
33. Cunha-Cruz J, Stout JR, Heaton LJ, Wataha, JC. Dentin hypersensitivity and oxalates: a systematic review. J Dent Res. 2011;90(3):304-310.
34. Hare TC, Zsiska M, Boissy Y, et al. Immediate and durable effects of an oxalate strip on human dentin in vitro. Compend Contin Educ Dent. 2016;37(spec iss 1):6-12.
35. Ciucchi B, Bouillaguet S, Holz S, Pashley D. Dentin fluid dynamics in human teeth, in vivo. J. Endod. 1995;21(4):191-194.
36. Andrews SA, Van Hassel HJ, Brown AC. A method for determining the physiologic basis of pulp sensory response. A preliminary report. J Hosp Dent Pract. 1972;6(2):49-53.
37. Beveridge EE, Brown AC. The measurement of human dental intrapulpal pressure and its response to clinical variables. Oral Surg Oral Med Oral Pathol. 1965;19:655-668.