In Vitro Interproximal Enamel Fluoride Uptake as a Function of Fluoride Delivery Vehicles
Abstract :
Background: Interproximal sites are often covered with a thin layer of saliva, which forms isocaps. These can restrict adequate delivery of therapeutic agents such as fluoride. Methods: Chemically demineralized enamel blocks were paired to simulate interproximal sites and exposed to human saliva to form isocaps. Enamel fluoride uptake (EFU) at these interfaces was studied using 220 ppm fluoride (F) rinse, 1.23% acidulated phosphate fluoride (Apf) gel, 5% sodium fluoride (NaF) varnish, and 2 novel vehicles – a pH nonacidic NaF disk (pHn disk), and an Apf disk, both containing 1.23% F and applied with or without NaF varnish. Enamel fluoride uptake was determined in peripheral and interior interproximal sites using the microdrill technique. Results: At the peripheral site, EFU was in the following order: pHn disk + varnish = Apf disk + varnish > pHn disk > Apf disk = Apf gel = NaF varnish > placebo disk = rinse. At the internal site, EFU was in the following order: pHn disk + varnish = Apf disk + varnish ≥ Apf disk ≥ pHn disk > Apf gel = NaF varnish = placebo disk = rinse. Conclusion: The present study, which simulated interproximal sites, showed that the novel disks used with or without NaF varnish significantly enhanced enamel fluoride uptake. Clinical Implications: The novel disks, when used alone or with NaF varnish, have the potential to effectively deliver fluoride to interproximal carious lesions.
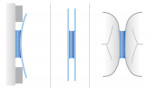
The contact area between two adjacent teeth has been erroneously referred to as a “contact point” resembling two marbles making contact. This is often not the case because interproximal attrition causes the contact to flatten and teeth to move, which results in a large contact area.1,2 These interproximal spaces, which also extend from the contact area, are filled with saliva. Saliva, which comprises about 10% of the oral cavity volume, forms various films over the hard and soft tissues and small, localized pools at different sites. These distributions usually depend on anatomic features, saliva flow rates, swallowing, mastication, gravitational forces, and capillary pressures. The latter occurs when saliva penetrates by capillary action around teeth at sites of caries susceptibility, such as at interproximal sites. If the amount of saliva is limited, isolated capillaries called isocaps form (Figure 1).3,4 These isocap micro-environments, which are similar to water or saliva between two glass slides, are under negative hydrostatic pressures and exert adhesive forces that can bend glass plates.3,4
Although adjacent teeth may be submerged in saliva, most interproximal sites are not soaked in saliva but form isocaps. It is hypothesized that these salivary isocap sites between adjacent enamel surfaces will restrict adequate access of therapeutic agents and even the penetration of the extremely active chemical ion, fluoride, which is the ninth smallest ion yet the most electro-negatively charged. Thus, the purpose of this in vitro study was to determine whether simulated isocaps restrict fluoride delivery from a homecare rinse and professionally applied topical fluorides in comparison with a novel series of topical fluoride disks designed to be placed interproximally, where they form a gel and then biodegrade after releasing fluoride into interproximal sites.
Materials and Methods
Specimen Preparation
Randomly selected enamel specimens were prepared from bovine teeth to form the hard-tissue enamel test substrate. The teeth were cleaned and then selected based on whether the particular tooth surface had sufficient size for obtaining a specimen large enough to meet the study requirements. The tooth sections were cut into specimens of 4×4 mm using a Buehler IsoMet® low-speed saw (Buehler, www.buehler.com) and stored in 0.01% thymol solution before and during the sample preparation process. The specimens were ground and polished to create flat surfaces; the bottom of the specimens was ground flat to a uniform thickness with 500-grit silicon carbide grinding paper, and the top of each specimen was ground using 1200-grit paper until most of the tooth surface was flattened. The specimens were serially polished using 4000-grit paper followed by 1-µm diamond polishing suspension. Specimens had at least 0.3 mm of enamel thickness. The specimens were sonicated in deionized water between grinding and polishing steps were performed. As a final cleaning step, the polished specimens were sonicated in 2% microliquid. The specimens were assessed under 10× magnification to ensure that they had no obvious cracks or flaws in the enamel surface, they had an evenly polished high-gloss enamel surface, and they had no contaminant such as sticky wax on the experimental surfaces. All specimen surfaces, except the polished enamel surfaces, were covered with a colored nail varnish. For each group, 12 specimens were used, and the prepared specimens were stored at 100% relative humidity until further use.
Creation of Subsurface Caries-like Lesion
An incipient, caries-like lesion was formed in each specimen by placement into a 0.1 M lactic acid/0.2% Carbopol 907/HAP solution for 24 hours at 37°C. After this demineralization, specimens were rinsed thoroughly with deionized water and again stored at 100% relative humidity.
Creation of Experimental Interproximal Space
Before any treatment phases were begun, the experimental interproximal sites were set up. Two enamel specimens, with the experimental enamel surfaces facing each other, were held adjacent and secured parallel to each other by positioning a resilient, compressible rubber block on the back surface of each enamel block (to stimulate the interproximal periodontal ligament pressure). The specimens and rubber blocks were also secured on an acrylic base with bordering acrylic units.
Treatment Phase
Details of the 6 test products can be found in Table 1. Acidulated phosphate fluoride (Apf) and pHn disks were used either alone or in combination with fluoride varnish, thus resulting in 8 treatment groups.
First, each enamel interproximal sample was coated with 0.25-mL fresh-pooled saliva (wax-stimulated, collected from at least 5 healthy adults) to create isocaps and then was treated at room temperature as described below. The fluoride rinse was applied without dilution at room temperature for 2 minutes under continuous shaking (by hand). During the treatment phase, specimens were fully immersed in the rinse. Specimens to be treated with the novel disks had a disk pushed between the adjacent enamel blocks performed according to the manufacturer’s instructions after drying with the use of an air syringe. The Apf gel was applied to the specimens for 4 minutes and then removed, with both steps conducted according to the manufacturer’s instructions. The fluoride varnish was applied with the supplied applicator brush. Specimens treated with both disks and varnish were treated by pushing the disks between the adjacent enamel blocks and then placing a dab of varnish on both sides of the disk and both enamel blocks.
After treatment had been performed, all interproximal sites were recoated with pooled human saliva, placed in pairs in a sealed container to ensure 100% humidity, and stored at 37°C for 4 hours. At 30-minute intervals, saliva was mixed by wrist-action swirling, and the specimens were recoated with saliva by gently tipping the container to coat each specimen’s interproximal site with approximately 0.25 mL of human saliva. After 4 hours, specimens were removed and rinsed with deionized water to remove any remnants of the disks that did not biodegrade. The fluoride varnish was removed by wiping the specimens with chloroform-soaked gauze until no visible varnish remained on the specimens. Specimens were rinsed again with deionized water and stored at 100% relative humidity until fluoride analysis could be performed.
Fluoride Analysis
The fluoride content of each enamel specimen was determined using the microdrill technique to a depth of 100 µm. Two sets of drill holes (Figure 2) were placed on each specimen – 4 along the peripheral interproximal margin (peripheral enamel) and 4 furthest from the experimental environment (interior enamel) – in order to also investigate fluoride migration into deeper parts of the interproximal spaces.
The diameters of the drill hole were determined using a microscope (average approximately 400 µm). The enamel powder from the drill holes was collected, dissolved (20 µL of HClO4, 40 µL of citrate/EDTA buffer, and 40 µL of deionized water), and analyzed for fluoride by comparison with a similarly prepared standard curve. Fluoride data were calculated as µg F/cm3: (µg F × dilution factor – volume of drilling).
Data Management and Analysis
The mean, standard deviation (SD), and standard error of mean (SEM) for each parameter of each group were calculated. Statistical analysis of the data was performed with a two-way analysis of variance model and a t test using SigmaPlotTM (11.0) software (Systat Software,
www.sigmaplot.com) with treatment and area of fluoride uptake as factors. In addition, all pairwise comparisons between the groups were conducted (Student-Newman-Keuls test). The selected level of significance was P < .05.
Results
The results of the enamel fluoride uptake and related statistical analyses are shown in Table 2 and Figure 2 through Figure 4. Regardless of site, the dual application of the novel disks and fluoride varnish proved most beneficial for enamel fluoride uptake (EFU), which was mainly driven by the disks and with no difference being observed between pHn and Apf disks except for interior enamel where the Apf disks with or without varnish application significantly improved EFU. The novel disks applied on their own delivered a greater amount of fluoride to early carious lesions than any of the other tested products, including two professionally applied fluoride products – an Apf gel and a sodium fluoride (NaF) varnish. At the peripheral site, the pHn disk delivered more fluoride than the Apf disk. The fluoride rinse failed to deliver a significant amount of fluoride regardless of the site under the chosen conditions. Likewise, fluoride delivery to interior enamel by Apf gel and NaF varnish was not significantly different to that from the placebo disk or fluoride rinse.
Regarding the comparison of fluoride uptake at different enamel areas (peripheral and internal) of the same treatment regimen, no significant difference within fluoride rinse, placebo disk, Apf gel, NaF varnish, pHn disk, and Apf disk with the varnish groups could be observed. However, the Apf disk alone and pHn disk with varnish both promoted significantly more fluoride uptake in the interior enamel than in the peripheral sites.
Discussion
The varnish and Apf tray gel were significantly more efficacious than the placebo and rinse; however, they were not significantly different. The in vitro results seemed to mimic what has been shown clinically as the interproximal decayed, missing, and filled tooth surface (DMFS) results5,6 of a varnish, and Apf tray gel applications were reported to show no significant differences. The DMFS increment results showed that the higher increase in caries was at the interproximal surfaces even though the study focused on 12- to 13-year-olds over 3 years, when eruption patterns probably allowed for significant exposure of mesial and distal surfaces to topical fluoride applications. The isocaps had probably not formed at all intervals and interfaces during the study.
The fluoride Apf and pHn disks used in this study have been produced for the prevention and remineralization of incipient interproximal caries within clinical settings. Dental professionals can insert the disks into interproximal sites of anterior teeth and where spacing allows. In cases in which spacing is restrictive even with tooth separation, these disks can be inserted in a folded format.
The Apf disks were less effective at the peripheral sites, as the hydrogen fluoride (HF) within them is more soluble and probably readily dissolved; however, the varnishes significantly improved their peripheral performance. Conversely, the Apf disks were significantly more effective at the interior sites. Based on the results from previous studies,7,8 it is reasonable to hypothesize that the Apf disks could also have an advantageous effect deeper within the enamel. Therefore, it may be prudent in the future to investigate fluoride differences and remineralization patterns at different surface layers by comparing nonacidic and acidic topical fluoride vehicles in the presence of salivary macromolecules, which seem to play an important role with fluoride in the continuation of deeper remineralization by reducing mineral gains at enamel surfaces.9
Although pooled human saliva was used, further research into the anticaries efficacy of the tested novel disks is necessary, as laboratory models are surrogates for the clinical situation. In vitro studies can overestimate or underestimate potential clinical outcomes, which are influenced by a wide range of human variables. This often makes comprehensive clinical assessments difficult.10 For example, the first varnish, Duraphat® (Colgate-Palmolive Company, www.colgateprofessional.com), which was introduced more than 45 years ago and remained popular for at least 30 years, lacked conclusive and comprehensive clinical data 30 years after having being marketed.11,12
Conclusion
Novel topical fluoride disks may significantly enhance the uptake of fluoride at interproximal sites. This preliminary evidence based on an in vitro model supports the use of topical fluoride disks for prevention and treatment of interproximal caries lesions.
Acknowledgments
The authors would like to acknowledge Ms. Jennifer Eder and Mr. Rolando Torres for their invaluable contributions to the study design and conduct of the study and Mrs. Tamar Zemel for the graphics.
Disclosures
Dr. Jodaikin patented and developed the Apf and pHn Phocal™ disks and is the founder and a director of Colldent Ltd. Dr. Cooley is an independent consultant in the oral care industry. Dr. Lippert and Dr. Martinez-Mier were provided support for their research.
References
1. Gilmore HW, Lund MR, Bales DJ, et al . Operative Dentistry. 3rd ed. St. Louis, MO: Mosby; 1977:25-26.
2. Kieser, JA, Groeneveld HT, Preston CB. Patterns of dental wear in the Lengua Indians of Paraguay . Am J Phys Anthropol. 1985;66(1):21-29.
3. O’Brien WJ. Surface phenomena and adhesion. In: O’Brien WJ, Ryge G . An Outline of Dental Materials and Their Selection. 1st ed. Philadelphia, PA: Saunders; 978:49-52.
4. Craig RG . Restorative Dental Materials. 6th ed. St. Louis, MO: Mosby; 1980:38-41.
5. Seppä L, Leppänen T, Hausen H. Fluoride varnish versus acidulated phosphate fluoride gel: a 3-year clinical trial . Caries Res. 1995;29(5):327-330.
6. Seppä L. Efficacy and safety of fluoride varnishes . Compend Contin Educ Dent. 1999;20(1 Suppl):18-35.
7. Clarkson BH, Wefel JS, Silverstone LM. Redistribution of enamel fluoride during white spot lesion formation: an in vitro study on human dental enamel . Caries Res. 1981;15(2):158-165.
8. Yamazaki H, Margolis HC. Enhanced enamel remineralization under acidic conditions in vitro . J Dent Res. 2008;87(6):569-574.
9. Fujikama H, Matsuyama K, Uchiyama A, Nakashima S, Ujiie T. Influence of salivary macromolecules and fluoride on enamel remineralization in vitro . Caries Res. 2008;42(1):37-45.
10. White DJ. The application of in vitro models to research on demineralization and remineralization of the teeth . Adv Dent Res. 1995;9(3):175-193.
11. Bader JD, Shugars DA, Bonita AJ. Systematic reviews of selected dental caries diagnostic and management methods . J Dent Educ. 2001;65(10):960-968.
12. Petersson LG, Twetman S, Dahlgren H, et al. Professional fluoride varnish treatment for caries control: a systematic review of clinical trials . Acta Odontol Scand. 2004;62(3):170-176.