Examining bacteria growing on toddlers' teeth, a team from the University of Pennsylvania and Georgia Tech found that the microbes' spatial organization is crucial to how they cause tooth decay
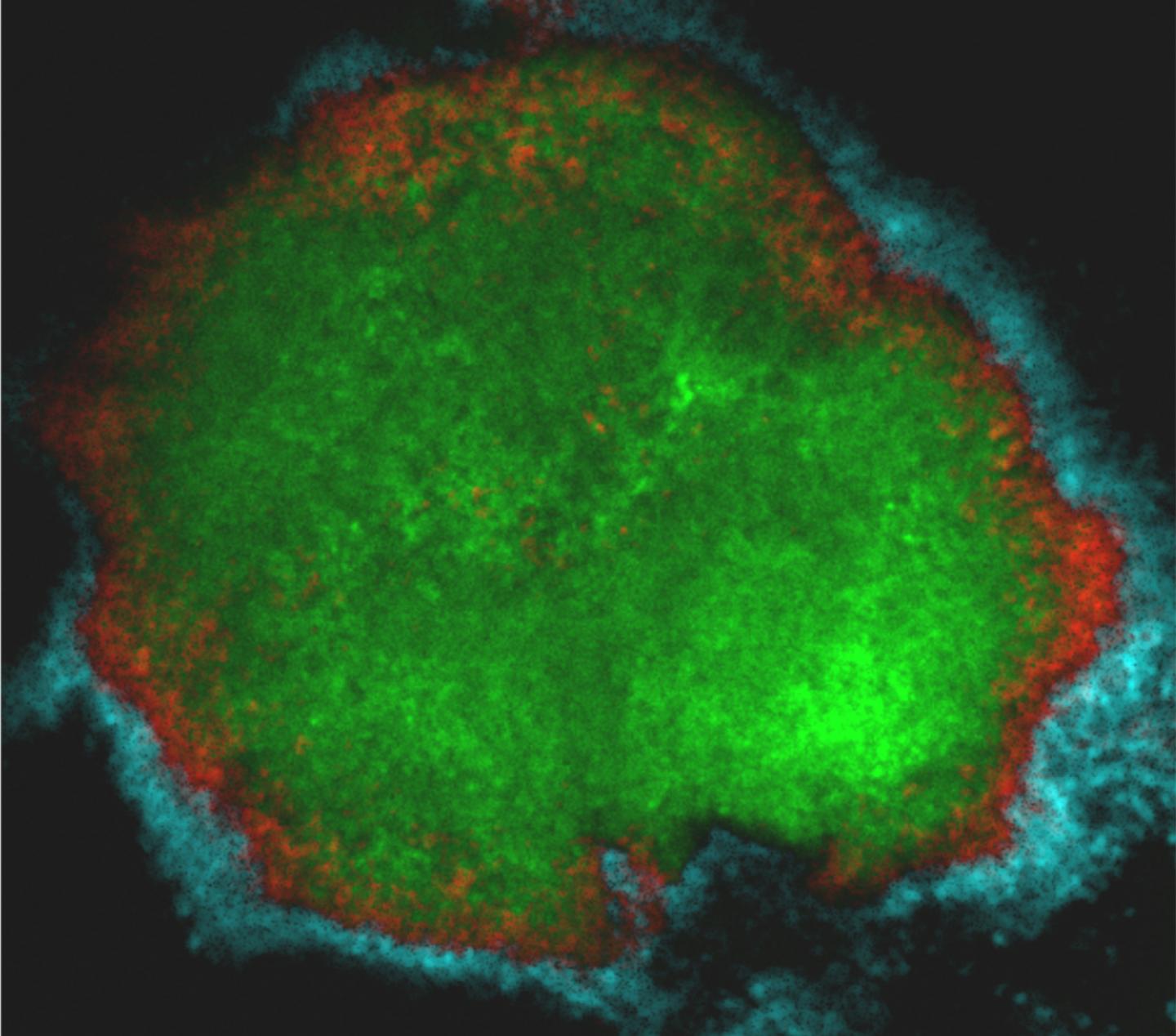
Taking a translational approach, researchers at the University of Pennsylvania School of Dental Medicine and the Georgia Institute of Technology imaged the bacteria that cause tooth decay in three dimensions in their natural environment, the sticky biofilm known as dental plaque formed on toddlers' teeth that were affected by cavities.
The work, published in the journal Proceedings of the National Academy of Sciences, found that Streptococcus mutans, a major bacterial species responsible for tooth decay, is encased in a protective multilayered community of other bacteria and polymers forming a unique spatial organization associated with the location of the disease onset.
"We started with these clinical samples, extracted teeth from children with severe tooth decay," says Hyun (Michel) Koo of Penn Dental Medicine, a co-senior author on the work. "The question that popped in our minds was, how these bacteria are organized and whether their specific architecture can tell us about the disease they cause?"
To address this question, the researchers, including lead author Dongyeop Kim of Penn Dental Medicine and co-senior author Marvin Whiteley of Georgia Tech, used a combination of super-resolution confocal and scanning electron microscopy with computational analysis to dissect the arrangement of S. mutans and other microbes of the intact biofilm on the teeth. These techniques allowed the team to examine the biofilm layer by layer, gaining a three-dimensional picture of the specific architectures.
This approach, of understanding the locations and patterns of bacteria, is one that Whiteley has pursued in other diseases.
"It's clear that identifying the constituents of the human microbiome is not enough to understand their impact on human health," Whiteley says. "We also have to know how they are spatially organized. This is largely under studied as obtaining intact samples that maintain spatial structure is difficult."
In the current work, the researchers discovered that S. mutans in dental plaque most often appeared in a particular fashion: arranged in a mound against the tooth's surface. But it wasn't alone. While S. mutans formed the inner core of the rotund architecture, other commensal bacteria, such as S. oralis, formed additional outer layers precisely arranged in a crownlike structure. Supporting and separating these layers was an extracellular scaffold made of sugars produced by S. mutans, effectively encasing and protecting the disease-causing bacteria.
"We found this highly ordered community with a dense accumulation of S. mutans in the middle surrounded by these 'halos' of different bacteria, and wondered how this could cause tooth decay," Koo says."
To learn more about how structure impacted the function of the biofilm, the research team attempted to recreate the natural plaque formations on a toothlike surface in the lab using S. mutans, S. oralis, and a sugar solution. They successfully grew rotund-shaped architecture and then measured levels of acid and demineralization associated with them.
"What we discovered, and what was exciting for us, is that the rotund areas perfectly matched with the demineralized and high acid levels on the enamel surface," says Koo. "This mirrors what clinicians see when they find dental caries: punctuated areas of decalcification known as 'white spots.' The domelike structure could explain how cavities get their start."
In a final set of experiments, the team put the rotund community to the test, applying an antimicrobial treatment and observing how the bacteria fared. When the rotund structures were intact, the S. mutans in the inner core largely avoided dying from the antimicrobial treatment. Only breaking up the scaffolding material holding the outer layers together enabled the antimicrobial to penetrate and effectively kill the cavity-causing bacteria.
The study's findings may help researcher more effectively target the pathogenic core of dental biofilms but also have implications for other fields.
"It demonstrates that the spatial structure of the microbiome may mediate function and the disease outcome, which could be applicable to other medical fields dealing with polymicrobial infections," says Koo.
"It's not just which pathogens are there but how they're structured that tells you about the disease that they cause," adds Whiteley. "Bacteria are highly social creatures and have friends and enemies that dictate their behaviors."
The field of microbial biogeography is young, the researchers say, but extending this demonstration that links community structure with disease onset opens up a vast array of possibilities for future medically relevant insights.
Dongyeop Kim was a research associate at Penn's School of Dental Medicine's Department of Orthodontics and is now an assistant professor at the Jeonbuk National University (Korea).
Hyun (Michel) Koo is a professor in Penn's School of Dental Medicine's Department of Orthodontics in the divisions of Community Oral Health and Pediatric Dentistry.
Marvin Whiteley is a professor of biological sciences, the Georgia Tech Bennie H. and Nelson D. Abell Chair in Molecular and Cellular Biology, and the Georgia Research Alliance Eminent Scholar co-director in Emory-Children's CF Center at the Georgia Institute of Technology.
Koo, Kim, and Whiteley's coauthors were Penn Dental Medicine's Rodrigo A. Arthur, Yuan Liu, Elizabeth L. Scisci, and Evlambia Hajishengallis; Georgia Tech's Juan P. Barraza; and Indiana University's Anderson Hara and Karl Lewis.
The work was supported in part by the National Institute for Dental and Craniofacial Research (grants DE025220, DE018023, DE020100, and DE023193).




