When Guided Surgery Fails: A Routine to Recover the Implant Original Treatment Plan
Robert J. Stanley, DDS, PhD, MS
ABSTRACT
Guided dental implant surgeries, their associated stackable surgical guides, and respective prosthetic techniques facilitate control and precision during implant placement and simultaneously streamline immediate provisionalization. Although implant treatments have demonstrated documented success, their failures both immediately and over time have traditionally required an additional surgery, a completely new prosthetic work-up, and subsequent fabrication of associated surgical placement components. This article describes a routine to recover the original implant placement treatment plan, without the added time, expense, or inconvenience involved in redeveloping the implant placement surgical guide. A case is presented to document the protocol for this routine.
Guided implant surgery has enhanced the accuracy and efficiency of providing dental implant treatments,1,2 affording patients with hopeless dentition the benefits of prosthetically driven, life-altering rehabilitations. Based on CAD/CAM and 3D printing, digitally planned implant treatments, corresponding stackable surgical guides, and prosthetic techniques facilitate control and precision during implant placement. They also streamline immediate provisionalization with a prefabricated long-term provisional. This approach to "all-on-x" treatment is certainly gaining in popularity because of the aforementioned benefits and as indicated by an ever increasing number of manufacturers entering the market. This process, however, can have a potential costly liability.
Although dental implants demonstrate documented success, their failures both immediately and over time are equally well reported.3,4 Failures may result from improper implant position, peri-implant diseases, bone loss, or other factors.3,4 When implants fail, dentists and their patients invest considerable time and resources to determine if the implants are salvageable, require explantation, and/or can be re-implanted.3-5
Unfortunately, from the moment an implant surgical guide is removed from the patient's mouth after the placement procedure, it and all its stackable components become obsolete. Traditionally, if one or more implants fails during or after surgical placement, the future reimplantation surgery requires a completely new prosthetic work-up and subsequent fabrication of the surgical guide components. The time, cost, and patient inconvenience associated with reimplantation are all undesirable consequences of implant placement failures and the now-useless original guided placement armamentarium.
Fortunately, it is possible to prepare for potential problems, develop a contingency plan for strategically handling an implant failure if it occurs, and otherwise "recover" the original implant placement treatment plan.6 Doing so can be accomplished without the added time, expense, and inconvenience associated with redeveloping the implant placement surgical guide plan.
By converting the original bone-level surgical guide to a grounded (ie, rigid fixation) implant-supported surgical guide immediately following the primary implant placement surgery, the need to perform a completely new prosthetic work-up and subsequently fabricate new surgical placement components is eliminated. This approach facilitates a simple recovery of the original surgical treatment plan for both the practicing clinician and the patient.
This article discusses the routine to recover the original implant placement treatment plan, and a case is presented to document its respective protocol.
Case Presentation
A 45-year-old man presented with the chief complaint of missing teeth, an unesthetic smile, sleep issues, and failing implants. Medical history revealed chronic lower back pain (for which he was taking hydrocodone/acetaminophen [Vicodin®]), sleep apnea, and no known medication allergies. The patient denied tobacco and alcohol use. Thorough intraoral, photographic, periodontal, and radiographic examinations were performed.
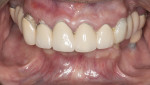
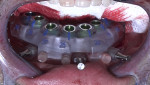
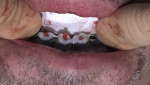
In addition to failing implants at Nos. 19 and 30, which had been treatment-planned for single-unit crowns by another dentist, the patient exhibited a class 2 malocclusion and pathological habitual deep bite exacerbated by inadequate posterior support (Figure 1). The combination of reported sleep apnea, severe attrition, and failing restorations rendered the risk factors for this case high. Risk mitigation techniques, including placing additional implants, fabricating the final prosthesis from zirconia, and providing sleep appliances, would be utilized.
Treatment options discussed with the patient included removable partial dentures, dental implants to replace select missing teeth, and/or full-mouth implant-supported reconstruction executed using a fully guided implant placement with a bone-level surgical guide and immediate provisionalization. After the risks and benefits of these options were outlined and the clinician answered all of the patient's questions, the treatment plan agreed upon would incorporate maxillary and mandibular full-arch reconstruction according to a prosthetically driven protocol, fully guided implant placement using a bone-level surgical guide, and immediate provisionalization with a prefabricated milled titanium framework wrapped in milled monolithic polymethyl methacrylate (PMMA) long-term provisionals.
Prosthetic Planning
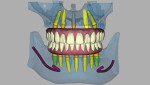
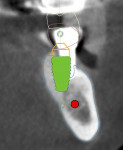
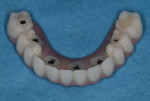
Five maxillary and seven mandibular implants were digitally planned to maximize anterior-posterior implant spread and minimize prosthetic cantilevers. Implant depth was based on hiding the soft-tissue-to-prosthesis transition line apical to the high smile line. All implant placements were planned according to the five-thread guideline for predicting immediate stability7; this would ensure the implants would be surrounded by 2 mm of bone circumferentially, maximizing implant diameter and length while maintaining a 2 mm safety zone from all vital structures (Figure 2).
Implant Placement
A certified registered nurse anesthetist assisted with administering intravenous sedation to the patient, after which local anesthetic was administered. The remaining teeth were extracted, and two failing mandibular implants removed.
The implant at No. 19 was surrounded by purulence and was removed with minimal effort using 151 forceps. An autogenous bone graft harvested from the patient was placed at this site. The implant at No. 30 had 60% cupping bone loss and was removed with a piezotome (Piezotome Cube, Acteon North America, acteongroup.com) and 151 forceps.
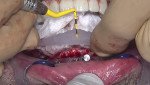
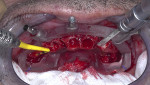
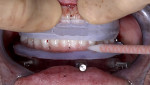
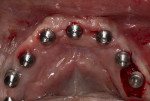
Full-thickness flap elevation enabled secure seating of the bone foundation guide (BFG) (nSequence® CT Surgical Guides, NDX nSequence, nsequence.com) directly onto the bony mandible. This fixed coordinate guide system was the foundation for driving the entire prosthetic process while simultaneously guiding bone reduction. To ensure the guide was accurately seated prior to rigid fixation, the bone coronal to the BFG was visually compared to the laboratory-fabricated acrylic bone reduction model (Figure 3). Upon confirmation, the guide was fixed to the bone using three fixation pins via bicortical stabilization through the buccal and lingual cortical plates. The bone was reduced utilizing a combination of reciprocating saw, bone burs, and rongeurs and copious sterile saline solution (Figure 4).
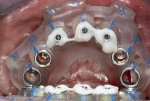
The implant surgical guide was stacked on top of the BFG and secured with the supplied plastic pins (Figure 5). Fully guided implant placement was performed, with ≥70 Ncm insertion torque, as measured by the surgical handpiece (Bien-Air, bienair.com) on the five maxillary and seven mandibular implants, except for the implant planned for the No. 19 site. The implant placed in the No. 19 site was deemed unstable and removed due to the extent of localized infection; the risk associated with this site was anticipated during the digital planning phase (Figure 6).
The implant surgical guide was removed from the BFG, and individual multi-unit abutments were placed. Titanium copings (Ti-copings) were lightly screwed onto their respective abutments, after which the first of three chairside pick-up procedures was performed.
First pick-up procedure: The first chairside pick-up was used for creating the long-term provisional. A silicone block-out gasket was secured to the BFG via three dado grooves on lingual aspect of the BFG. The provisional was stacked securely on top of the block-out gasket and supported apically by the BFG. A laboratory-fabricated bite splint was then placed, allowing the provisional to be properly seated prior to luting. With the patient closed into maxillary mandibular relations, thereby securing the provisional, luting agent (Luxatemp, DMG, dmg-america.com) was injected into the facial luting holes on the provisional (Figure 7). Once the luting agent set, the Ti-coping screws were loosened, and the provisional was removed from the patient's mouth. Any luting flash was removed and the provisional polished.
Second pick-up procedure: The second chairside pick-up created an acrylic clear duplicate of the long-term provisional. This process was identical to that previously described. The Ti-copings were placed, the acrylic clear duplicate secured via bite splint, and the luting agent injected into the duplicate for securing to the Ti-copings (Figure 8). The clear duplicate was removed, cleaned, and safely stored for the final restorative phase, which was planned for 6 months after the initial surgery.
Third pick-up procedure: To attempt to recover the original treatment plan after the first reimplantation failure at No. 19, a third chairside pick-up was undertaken using the BFG still secured to bone to preserve the fixed coordinate system. Ti-copings were placed on the three anterior mandibular implants, and the implant surgical guide was then placed over them and secured onto the BFG via the three plastic pins (Figure 9). Luting agent was injected into the space between the Ti-copings and the master cylinders inside the surgical guide. Extreme care was taken to only inject sufficient luting agent to capture the Ti-coping and not express into the unprotected and exposed bone (Figure 10).8,9 Once the luting agent was set, the Ti-coping screws were loosened, and the surgical guide removed.
This pick-up technique successfully converted the previously bone-supported surgical guide into an implant-supported surgical guide. The new implant-supported surgical guide (ie, implant treatment plan recovery surgical guide) was set aside for later use when replacing the No. 19 implant in the exact same location as the original prosthetic plan.
Surgical Completion
The BFG was then removed, and the flap was lowered and sutured with 4-0 chromic gut. Because the cantilever at the No. 19 location on the long-term provisional was sufficiently small to pose little concern during healing, the hole at that area of the provisional was filled with provisional material (Luxatemp) and polished (Figure 11). The long-term provisionals were placed, the occlusion refined, and the patient dismissed.
Treatment Plan Recovery
Three months after the primary surgery, the patient returned for the second surgical attempt at placing an implant at the No. 19 site utilizing the implant-supported treatment plan recovery surgical guide. The patient was anesthetized via an inferior alveolar nerve block with lidocaine 2% plus epinephrine 1:100,000. The provisional was removed, and a #15 blade was used to create a mid-crestal incision. Keratinized gingival tissue was displaced buccally and inferior to the No. 19 site to ensure that at least 3 mm of keratinized gingiva remained on the buccal aspect (Figure 12).10
The treatment plan recovery surgical guide was secured to implants Nos. 22, 25, and 27 via standard prosthetic screws. Guide stability was confirmed, and the implant was placed in the No. 19 location using routine fully guided surgical protocol.
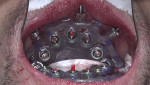
A healing abutment was placed on the No. 19 implant, after which suture caps were placed on the six other healed mandibular implants (Figure 13). Because the patient had sustained fractures to the long-term polymethyl methacrylate (PMMA) provisional prosthesis due to his bruxing, he was dismissed without it to enable laboratory repair. The patient returned 3 days later, at which time the repaired long-term provisional was placed, and the implant in the No. 19 site was captured according to the previously described Ti-coping and luting protocol.
Final Prosthesis
Three months after the implant treatment plan recovery surgery (ie, 6 months after the initial surgery), the implant in the No. 19 position had healed successfully, and the successful final prosthetic work-up was completed according to the original prosthetic plan (Figure 14).
Conclusion
When planning fully guided implant placement, many intricate components are designed, fabricated, and utilized. If one or more planned implants fails, the prospect of re-implantation at the failed site can be costly for the doctor while potentially subjecting the patient to additional appointments, radiographs, and invasive surgery. The proposed solution of converting the bone-level surgical guide into an implant-supported surgical guide at the time of primary surgery eliminates these concerns.
Of course, some implants establish primary stability and later fail to osseointegrate, and it may be difficult to anticipate which implants might fail. Therefore, a prudent approach may be to capture all implants when converting the surgical guide. If three implants remain stable, the fixed coordinate system remains intact.
The protocol described in this article facilitates a simple recovery routine for the practicing clinician and could become a standard operating protocol for all prosthetic-driven, fully guided multiple implant placement procedures. It enables reinserting an implant at the failed site while preserving the original prosthetic plan.
About the Author
Robert J. Stanley, DDS, PhD, MS
Adjunct Professor in Prosthodontics, University of North Carolina School of Dentistry, Chapel Hill, North Carolina; Co-founder and Senior Instructor, Stanley Institute for Comprehensive Dentistry, Cary, North Carolina; Private Practice, Cary, North Carolina
References
1. Umapathy T, Jayam C, Anila BS, Ashwini CP. Overview of surgical guides for implant therapy. J Dent Implants. 2015;5(1):48-52.
2. D'Souza KM, Aras MA. Types of implant surgical guides in dentistry: a review. J Oral Implantol. 2012;38(5):643-652.
3. Gomes GH, Misawa MYO, Fernandes C, et al. A systematic review and meta-analysis of the survival rate of implants placed in previously failed sites. Braz Oral Res. 2018;32:e27.
4. Greenstein G, Cavallaro J. Failed dental implants: diagnosis, removal and survival of reimplantations. J Am Dent Assoc. 2014;145(8):835-842.
5. Machtei EE. What do we do after an implant fails? A review of treatment alternatives for failed implants. Int J Periodontics Restorative Dent. 2013;33(4):e111-e1119.
6. Mitrani R. Contingency, priority, and risk (CPR) protocol: a roadmap for designing successful and long-lasting implant therapy. Compend Contin Educ Dent. 2014;35(3 suppl):18-24.
7. Stanley R. The five thread guideline: a new guideline for predicting primary stability with dental implants. J Oral Implantol. 2020;46(1):81-86.
8. Wilson TG Jr. The positive relationship between excess cement and peri-implant disease: a prospective clinical endoscopic study. J Periodontol. 2009;80(9):1388-1392.
9. Linkevicius T, Puisys A, Vindasiute E, et al. Does residual cement around implant-supported restorations cause peri-implant disease? A retrospective case analysis. Clin Oral Implants Res. 2013;24(11):1179-1184.
10. Lin GH, Chan HL, Wang HL. The significance of keratinized mucosa on implant health: a systematic review. J Periodontol. 2013;84(12):1755-1767.