Periodontal Classification Revisited: The Classification System in the Test of Time
Avigdor Klinger, DMD, PhD
At the EuroPerio conference held in Amsterdam in 2018, an update to the previous classification of periodontal diseases was presented. The update was a joint effort of the American Academy of Periodontology (AAP) and the European Federation of Periodontology (EFP). On the face of it, the new classification system for diagnosing periodontal diseases and conditions is merely an update of previous definitions from 1999. However, upon close examination, it is much more than another semantic change of old definitions. The new classification rocks the ground upon which generations of pre- and post-graduate students and specialists in the field have stood. It is a perceptual change in the way clinicians describe, diagnose, and ultimately treat periodontal patients.
A Historical Perspective
To properly assess this modification, it is worth looking back over the history of periodontics. In the modern era, it was Pierre Fauchard who in 1746 described a disease unique to the gums. Scurvy was a common diagnosis at the time, and it was frequently linked with acute devitaminosis, which manifests itself in soft tissues. In the 19th century, it became known as Riggs' disease, named after Dr. John Mankey Riggs who devoted his practice to treating gums, and did so by rather conservative means that included debridement and rinsing with various preparations.
Professor Bernhard Gottlieb was the one who in 1923 described the histopathological changes in the attachment apparatus surrounding teeth. He demonstrated loss of collagen fibers and alveolar bone, with widening of the periodontal ligament (PDL). He noted that in many studied specimens it appeared that the superficial gingival tissues were not involved in the pathological process that takes place. In histological detail, he identified damage to the cementum layer and destruction of the PDL fibers. He assumed that a degenerative process he called "deep cementopathy" was the explanation for the phenomenon.
The continued development of periodontal nomenclature took an unexpected turn at this point in time. Like many Jewish scientists in Vienna, Austria, Gottlieb was forced to flee the Nazis in the 1930s. He sailed to America where, in professional conferences, he presented slides he had collected. Many of the listeners, including Dr. Henry Goldman, were convinced. They proposed the term "periodontosis," meaning a degenerative vascular process.
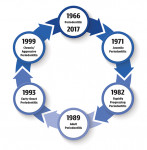
In 1966, at a world workshop held by the American Academy of Periodontics, a decision was made that due to lack of a scientific basis for degenerative processes taking place, the term "periodontosis" should be abandoned and substituted with "periodontitis." This change reflected the understanding that the pathological changes in the tissues are the result of inflammatory processes rather than degeneration. A few years later, two articles, one of them in German, described a series of cases of young patients.1 Juvenile periodontitis was characterized by rapid bone loss and manifested itself mainly in the first molars and incisors. Degree of destruction to the attachment apparatus was not correlated with the amount of local secondary factors.
In 1982, Page et al proposed the term "rapidly progressing periodontitis."2 In their view, many of the cases were characterized by a rapid destruction of the periodontal attachment, but it was not always possible to limit them to adolescence as Baer1 suggested. In contrast, the term "prepubertal periodontitis"3 was appropriate for diagnosis in children with mixed dentition, in whom loss of attachment begins as early as 11 years of age.
Definitions were updated in the late 1980s following yet another world workshop.4 It has been suggested that gum disease, which is found in the majority of the population, should be referred to as "adult periodontitis"; it was characterized by a gradual loss of attachment and manifested itself mainly after the age of 30. Those cases characterized by fast periodontal destruction were included in the early-onset periodontitis group. Cases in which a systemic background was identified in a patient were diagnosed as periodontitis associated with a systemic disease. The term "refractory periodontitis" was also suggested for patients who do not respond favorably to standard treatment despite a strict maintenance protocol.
Necrotizing ulcerative periodontitis is a diagnosis that, to date, has not changed and represents all cases that meet the criteria of an acute gum condition with characteristic clinical features.
The Concept of "Chronic" Periodontitis
A significant change in diagnoses occurred about 20 years ago when Armitage, of the University of California, authored the paper, "Developing a Classification System for Periodontal Diseases and Conditions," in which the concept of "adult" was abandoned in favor of "chronic" periodontitis.5 The rationale was clinical experience and the conclusions of researchers in epidemiological studies, which indicated that the disease that appears in most of the elderly population could also be seen in younger patients. Characteristics were slow progression and noticeable presence of local factors, such as plaque, calculus, faulty restorations, anatomical aberrations, or cigarette smoking. Its prevalence was about 35% of the western population,6 of which mild-moderate form accounted for 31% and severe cases for only 4%. It was also decided to abandon the historical concept of "juvenile periodontitis" and adapt the term "aggressive" form of disease. The rationale was observations that many of the diagnosed patients with destructive periodontitis were over the age of 20. The characteristics of aggressive periodontitis were: patients who were systemically healthy with rapid disease progression and a proven familial aggregation. Its prevalence was only about 0.1% to 0.5% of the studied population.
Quite prominent was the presence of Aggregatibacter actinomycetemcomitans (Aa) bacteria in the subepithelial layer in localized cases. The generalized form was characterized by the presence of Aa and Porphyromonas gingivalis. Studies have linked the ability to produce IgG2 antibody titer against Aa to the presence of local or widespread disease.7 Neutrophils are considered the first line of defense of the immune system in the gingival tissues. As early as the mid-1980s, a decrease in their function was observed in aggressive periodontitis.8 Another characteristic was family aggregation, as many of the patients were asked to refer siblings for examination and quite a few first-degree relatives were diagnosed with a similar disease, sometimes in even more destructive stages.9
Despite all of the aforementioned developments, leading experts assembled again in 2017 under the common umbrella of the AAP and EFP. Following the conclusion of this latest world workshop, the authors of the relevant consensus report published the reasons for abandoning the historical separation into two distinct diseases.10 Essentially, there was lack of scientific basis for the existence of two distinct pathophysiological processes to justify diagnosing a patient as suffering from either the chronic or the aggressive form of periodontitis. Hence, the inability to justify a different treatment protocol for each one.
Thus, virtually all of the alternative terms proposed for diagnosing periodontal diseases in the past 50 years or so have been substituted. We find ourselves today back to using the nomenclature decided upon as early as 1966. The disease is now simply called "periodontitis" (Figure 1).
Principles of the New Classification
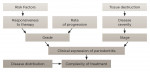
So what is new after all? The diagnosis of periodontitis is now based on the assessment that the patient has lost periodontal attachment in at least two sites not adjacent to one another. The loss is usually interdental but does not necessarily have to be. Loss of attachment of 1 mm to 2 mm is considered initial periodontitis, ie, stage 1 (Table 1). An increase of attachment loss will lead to an increase in disease stage. Periodontal pockets ≥6 mm deep, advanced bone resorption, furcation involvements, or tooth loss are characteristic of advanced periodontal disease in stages 3 or 4, depending on the number of teeth lost and the complexity of the rehabilitative care required (Table 1).
While stages reflect disease severity, degree reflects its rate of progression (Figure 2). This can be assessed directly by measuring the change that occurred at two different timepoints, or indirectly by calculating loss of bone support relative to the patient's age. Grade A reflects absence of disease progression, while grade C characterizes loss of attachment of 2 mm or more over the past 5 years (Table 2).
Another interesting modification introduced in the new classification is the taking into account of systemic factors that directly affect periodontal diagnosis. The link between periodontal disease and diabetes has been proven in many studies.11,12 For the first time, however, the patient's glycemic balance is said to directly affect the degree of disease in the mouth. Grade A represents a normoglycemic patient, while an unbalanced patient will be diagnosed with grade C periodontitis, regardless of its severity. This reflects the understanding that in unbalanced diabetes limited responsiveness to periodontal treatment and further loss of attachment are expected in the foreseeable future.
The same is true of patients with a smoking habit. Until now we had advised patients to stop smoking or at least reduce it; now we diagnose them accordingly. For example, a patient who is a heavy smoker will be diagnosed with grade C periodontitis.13-15
Summary in a Personal Tone
I started my post-graduate program in periodontology in 1998, about a year before the previous definitions came out. For the past 20 years my colleagues and I have treated our patients by diagnosing them with either a chronic or an aggressive disease. Today, this is all a thing of the past. Like my colleagues, I too question what the implications are of the new definitions for treating patients. If there are not two distinct diseases but one, is it justified to treat different patients differently? Does one disease also mean one treatment protocol for all?
For someone who has accompanied post-graduate students in periodontology programs for quite a few years now, I can attest that more than once the spirits in our department have been stormed on how to diagnose the patient. Not all cases fit the definition of a chronic or an aggressive type. The advantages of the new classification system are that it provides clinicians with well-defined parameters and gives them tools to measure and evaluate the situation accurately without getting into a controversy regarding the type of the patient's disease. Presumably, the new method will also be more suitable for conducting epidemiological studies that are more accurate and rely less on "gut feelings." This, of course, will have to stand the test of time.
About the Author
Avigdor Klinger, DMD, PhD
Faculty of Dental Medicine, Department of Periodontology, The Hebrew University-Hadassah
School of Dental Medicine, Jerusalem, Israel
REFERENCES
1. Baer PN. The case for periodontosis as a clinical entity. J Periodontol. 1971;42(8):516-520.
2. Page RC, Altman LC, Ebersole JL, et al. Rapidly progressive periodontitis. A distinct clinical condition. J Periodontol. 1983;54(4):197-209.
3. Page RC, Bowen T, Altman L, et al. Prepubertal periodontitis. I. Definition of a clinical disease entity. J Periodontol. 1983;54(5):257-271.
4. American Academy of Periodontology. Proceedings of the World Workshop in Clinical Periodontics. Chicago, IL: American Academy of Periodontology 1989:I/23-I/24.
5. Armitage GC. Development of a classification system for periodontal diseases and conditions. Ann Periodontol. 1999;4(1):1-6.
6. Albandar JM, Brunelle JA, Kingman A. Destructive periodontal disease in adults 30 years of age and older in the United States, 1988-1994. J Periodontol. 1999;70(1):13-29.
7. Tew JG, Zhang JB, Quinn S, et al. Antibody of the IgG2 subclass, Actinobacillus actinomycetemcomitans, and early-onset periodontitis. J Periodontol. 1996;67(suppl 3S):317-322.
8. Van Dyke TE, Schweinebraten M, Cianciola LJ, et al. Neutrophil chemotaxis in families with localized juvenile periodontitis. J Periodontal Res. 1985;20(5):503-514.
9. van Winkelhoff AJ, Rodenburg JP, Goene RJ, et al. Metronidazole plus amoxycillin in the treatment of Actinobacillus actinomycetemcomitans associated periodontitis. J Clin Periodontol. 1989;16(2):128-131.
10. Papapanou PN, Sanz M, Buduneli N, et al. Periodontitis: consensus report of workgroup 2 of the 2017 World Workshop on the Classification of Periodontal and Peri‐Implant Diseases and Conditions. J Clin Periodontol. 2018;45(suppl 20):S162-S170.
11. Grossi SG, Skrepcinski FB, DeCaro T, et al. Treatment of periodontal disease in diabetics reduces glycated hemoglobin. J Periodontol. 1997;68(8):713-719.
12. Soskolne WA, Klinger A. The relationship between periodontal diseases and diabetes: an overview. Ann Periodontol. 2001;6(1):91-98.
13. Kaldahl WB, Johnson GK, Patil KD, Kalkwarf KL. Levels of cigarette consumption and response to periodontal therapy. J Periodontol. 1996;67(7):675-681.
14. Rosen PS, Marks MH, Reynolds MA. Influence of smoking on long-term clinical results of intrabony defects treated with regenerative therapy. J Periodontol. 1996;67(11):1159-1163.
15. Haffajee AD, Socransky SS. Relationship of cigarette smoking to attachment level profiles. J Clin Periodontol. 2001;28(4):283-295.