Enamel Matrix Derivative for Hard- and Soft-Tissue Regeneration: Case Reports
Bradley Weinstein, DMD, MSD
Abstract: A valuable adjunct to both hard- and soft-tissue grafting, enamel matrix derivative (EMD) has been shown to regenerate the periodontal attachment, including cementum, Sharpey's fibers, and new bone. For hard-tissue regeneration, EMD has been applied to intrabony defects with and without bone substitutes. In soft-tissue regeneration applications, EMD has been utilized with a coronally advanced flap for gingival recession treatment. This article describes cases in which a protein-based gel containing EMD was used for hard- and soft-tissue regeneration.
Enamel matrix derivative (EMD) has been used in dentistry for more than 20 years,1 during which time research has shown it to have beneficial effects on both hard- and soft-tissue regeneration.2-6 Human histology has demonstrated that EMD has the ability to encourage regeneration of cementum with Sharpey's fibers insertion perpendicularly, a key requisite of periodontal regeneration.7 Periodontal regeneration, as opposed to repair with a long junctional epithelium, may be the reason for positive long-term outcomes with this material.8
For hard-tissue regeneration, EMD has been applied to intrabony defects with and without bone substitutes. When used without bone substitutes, EMD has been shown to reduce probing depths and encourage more bone fill as compared to controls.2,3 When used with bone substitutes, the evidence is mixed as to the benefits of EMD, with some studies showing benefits, and others showing no additional benefit to its use.9 A recent systematic review showed slight additional benefit to using EMD with bone grafts for the treatment of intrabony defects.10
For soft-tissue regeneration, EMD has been utilized with a coronally advanced flap (CAF) for gingival recession treatment. EMD used with CAF has similar outcomes in resolving recession long term when compared to the use of autogenous connective tissue (CT).8 For single-rooted tooth recession defects, CAF plus EMD has been recommended as a preferred procedure along with CAF plus CT.11 Histologically, recession treatment with EMD has been shown to regenerate cementum in recession defects; without EMD application, a long junctional epithelium is observed.12,13
This article describes cases where Emdogain® (Straumann, straumann.com), a unique protein-based gel containing EMD, was used for hard- and soft-tissue regeneration.
Hard-Tissue Regeneration
Case 1
An American Society of Anesthesiologists (ASA) class I, 28-year-old African American female patient, who was on Celexa and oral contraceptives, presented on referral from her general dentist to have teeth Nos. 24 and 25 evaluated for deep pocketing and purulent exudate. The general dentist had completed scaling and root planing on the site 6 months prior with minimal improvements observed.
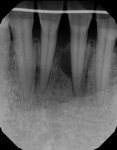
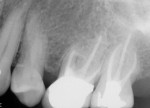
On the patient's initial examination, tooth No. 24 had an 8 mm probing depth on the mesiolingual line angle with heavy suppuration. Her initial periapical radiograph of the site showed a radiolucency on the mesial aspect of tooth No. 24 (Figure 1). The patient reported wearing a tongue ring for 2 years, which, combined with heavy calculus, appeared to have caused the bone defect.
Guided tissue regeneration (GTR) under a surgical microscope was planned for the site, and written informed consent was obtained. A sulcular incision was made from teeth Nos. 23 through 26 on the lingual surfaces, and a full-thickness flap was reflected. Tooth No. 24 was debrided with an ultrasonic scaler and hand instruments, and the bone defect was decorticated with a small round bur. The root surface was then treated with PrefGel® (Straumann) for 30 seconds, Emdogain was applied, and freeze-dried bone allograft (FDBA) was placed into the bone defect. The flap was closed with three simple interrupted 7-0 Vicryl® sutures (Ethicon, jnjmedicaldevices.com), and the patient was given prescriptions for postoperative amoxicillin 500 mg, chlorhexidine 0.12% topical rinse, and nonsteroidal anti-inflammatory drugs (NSAIDs) for pain control.
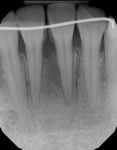
The patient was seen for a 1-year follow-up, at which point the probings had improved from 8 mm to 3 mm, and the periapical radiograph showed good bone fill at the bone defect (Figure 2).
Case 2
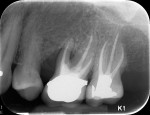
An ASA class I, 54-year-old Caucasian woman presented on referral from her general dentist to evaluate tooth No. 14, which had a 9 mm probing depth with suppuration present. The periapical radiograph showed a deep vertical bone defect and mesial furcation involvement (Figure 3). A CBCT revealed an unfilled second mesiobuccal (MB2) canal, thus the patient was referred to an endodontist for endodontic retreatment.
After endodontic retreatment was performed, the probing depth at tooth No. 14 remained deep at 10 mm with suppuration. Therefore, the patient was presented with several treatment options: GTR, root amputation, or extraction. The patient wished to retain the tooth, so GTR was planned, and written informed consent was obtained.
A sulcular incision was made from teeth Nos. 13 to 14 buccally and palatally, and a full-thickness flap was reflected. Heavy granulation tissue was removed from the mesiopalatal aspect of No. 14 as well as the furcation area. Heavy calculus was removed with an ultrasonic scaler and hand scalers. The root surface was treated with PrefGel and Emdogain, and FDBA was placed into the furcation and bone defect. The flap was closed with a single Gore-Tex® modified vertical mattress suture (Gore, goremedical.com) between Nos. 13 and 14, and the patient was given prescriptions for postoperative amoxicillin 500 mg, chlorhexidine 0.12% topical rinse, and NSAIDs for pain control.
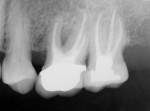
At the 4-month follow-up visit, probings had improved from 10 mm to 5 mm at tooth No. 14 (mesial), and the mesial class II furcation was now a class I furcation. The radiograph showed good bone fill of the prior defect (Figure 4). At the 1.5-year follow-up another radiograph was exposed that showed stability of the regenerated bone (Figure 5), and the tooth probed 4 mm with no inflammation present.
Soft-Tissue Regeneration
Case 3
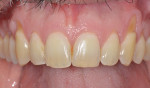
An ASA class I, 40-year-old Caucasian man, who had a 7 pack-year smoking history but was now quit, presented with a complaint of thermal sensitivity emanating from teeth Nos. 6 and 11. He reported a history of aggressive brushing. Examination revealed 4 mm of recession on tooth No. 6 and 6 mm of recession on tooth No. 11 (Figure 6). Tooth No. 6 had 2 mm of keratinized gingiva and a 2 mm facial probing depth, and tooth No. 11 had 1 mm of keratinized gingiva and a 3 mm probing depth. Both sites were treatment planned for coronally positioned flaps with CT grafting and root treatment with Emdogain, to be completed under a surgical microscope. Written informed consent was obtained.
A sulcular incision was made at both Nos. 6 and 11, as well as at teeth Nos. 4 and 5in order to address slight recession on these teeth. Vertical incisions were made at No. 11 mesial and distal, and a single vertical incision was made at No. 6 mesial. Full-thickness flaps were reflected at both sites and advanced via periosteal releasing incisions. The roots were conditioned with PrefGel and treated with Emdogain, and 2-mm thick subepithelial CT grafts were stabilized over the Nos. 6 and 11 roots with 6-0 Vicryl sling sutures. The overlying flaps were positioned with 6-0 and 7-0 Vicryl sutures as well as 7-0 polypropylene sutures.
At the 8-week follow-up visit, complete root coverage was observed, as was a gain of 2 mm to 3 mm of keratinized gingiva compared to baseline and increased tissue thickness (Figure 7).
Case 4
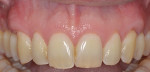
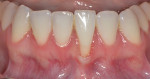
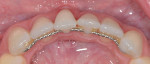
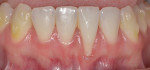
An ASA class I, 33-year-old Caucasian female patient presented with the chief complaint of progressing recession at tooth No. 24. On examination, tooth No. 24 had 4 mm of recession and no keratinized gingiva present (Figure 8). Additionally, the tooth was observed to be facially prominent, which appeared to be due to having been moved out of alignment by a bend in the lingual archwire, which had originally been placed for orthodontic retention (Figure 9).
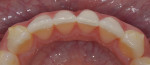
The patient was referred to an orthodontist to align the tooth, which was completed using clear aligners (Figure 10). After alignment, however, tooth No. 24 still had 4 mm of recession (Figure 11), so a coronally positioned flap with CT grafting and EMD application was planned, and written informed consent was obtained.
A sulcular incision was made at tooth No. 24 with vertical releasing incisions mesial and distal, then the root surface was treated with PrefGel and Emdogain. The graft and flap were secured with 6-0 Vicryl sutures, and the patient was given prescriptions for postoperative amoxicillin 500 mg, chlorhexidine 0.12% topical rinse, and NSAIDs for pain control.
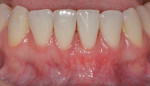
At the 8-week follow-up visit, complete root coverage was observed along with a gain of 3 mm of keratinized gingiva (Figure 12).
Discussion
In each of the presented cases, EMD (Emdogain) was utilized to enhance the surgical outcomes. In the hard-tissue regeneration cases (Cases 1 and 2), the rationale for using EMD was its proven ability to stimulate regeneration of the periodontal apparatus.1,2 Regeneration of clinical attachment allows for pocket reduction without significant gingival recession and its associated side effects such as dentin hypersensitivity. Also, in sites with challenging access, such as the lingual surfaces of mandibular teeth or the palatal surfaces of maxillary molars, the use of traditional GTR approaches with barrier membranes can be difficult. The handling properties of syringable EMD make it useful for these types of cases, and the less-invasive flap reflection leads to less swelling and greater patient comfort during healing.
In the soft-tissue cases (Cases 3 and 4), the patients presented with exposed root surfaces lacking cementum. EMD was used to stimulate cementum regeneration for a long-lasting attachment of the grafts to the previously exposed dentin surfaces. Histologically, it is possible that EMD also encouraged regeneration of bone and Sharpey's fibers in addition to cementum, leading to true periodontal regeneration and potentially a longer-lasting outcome. Because EMD reduces inflammation and swelling, there was also less tension on the flap, which is an important factor in patient comfort and amount of root coverage attained.
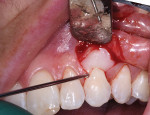
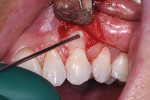
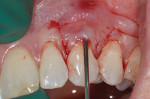
The author favors the use of EMD for soft-tissue grafting when recession is more than 4 mm. PrefGel (24% ethylenediaminetetraacetic acid [EDTA]) is applied to the root surface after flap reflection (Figure 13) and thoroughly rinsed, at which point the root is reinspected for any evidence of calculus. Hemostasis is achieved at the site prior to application of Emdogain to ensure that the material adsorbs to the root surface before any blood by-products materialize (Figure 14).14 Finally, after the flap is sutured, additional Emdogain is applied beneath the flap and along the incision lines (Figure 15). With this approach, the author has found that patients report minimal pain and swelling postoperatively.
Patients typically are eager to receive treatment that will yield them the best outcome with the least amount of pain, thus the use of a material such as EMD can help drive case acceptance when described in these terms. The author helps patients understand EMD by explaining it as a type of tissue "glue" that, by regenerating cementum, Sharpey's fibers, and bone, helps to restore the tissue types that have been lost. Because evidence supports the use of EMD instead of autogenous connective tissue for gingival recession,8 EMD is also a viable option for patients who wish to avoid a second surgical site for harvesting autogenous tissue.
Conclusion
As an adjunct to both hard- and soft-tissue grafting, EMD has been shown to regenerate the periodontal attachment, including cementum, Sharpey's fibers, and new bone. It also has beneficial effects on patient comfort. These features make it a valuable adjunct in challenging cases of hard- and soft-tissue defect treatment and useful in potentially increasing case acceptance.
Disclosure
This article was commercially supported by Straumann.
About the Author
Bradley Weinstein, DMD, MSD
Affiliate Faculty, University of Washington School of Dentistry, Seattle, Washington; Private Practice in Periodontics and Dental Implant Surgery, Seattle, Washington
References
1. Miron RJ, Sculean A, Cochran DL, et al. Twenty years of enamel matrix derivative: the past, the present and the future. J Clin Periodontol. 2016;43(8):668-683.
2. Heijl L, Heden G, Svärdström G, Ostgren A. Enamel matrix derivative (EMDOGAIN) in the treatment of intrabony periodontal defects. J Clin Periodontol. 1997;24(9 Pt 2):705-714.
3. Froum SJ, Weinberg MA, Rosenberg E, Tarnow D. A comparative study utilizing open flap debridement with and without enamel matrix derivative in the treatment of periodontal intrabony defects: a 12-month re-entry study. J Periodontol. 2001;72(1):25-34.
4. Spahr A, Haegewald S, Tsoulfidou F, et al. Coverage of Miller class I and II recession defects using enamel matrix proteins versus coronally advanced flap technique: a 2-year report. J Periodontol. 2005;76(11):1871-1880.
5. Cueva MA, Boltchi FE, Hallmon WW, et al. A comparative study of coronally advanced flaps with and without the addition of enamel matrix derivative in the treatment of marginal tissue recession. J Periodontol. 2004;75(7):949-956.
6. Cairo F, Pagliaro U, Nieri M. Treatment of gingival recession with coronally advanced flap procedures: a systematic review. J Clin Periodontol. 2008;35(8 suppl):136-162.
7. Sculean A, Alessandri R, Miron R, et al. Enamel matrix proteins and periodontal wound healing and regeneration. Clin Adv Periodontics. 2011;1(2):101-117.
8. McGuire MK, Scheyer ET, Nunn M. Evaluation of human recession defects treated with coronally advanced flaps and either enamel matrix derivative or connective tissue: comparison of clinical parameters at 10 years. J Periodontol. 2012;83(11):1353-1362.
9. Miron RJ, Guillemette V, Zhang Y, et al. Enamel matrix derivative in combination with bone grafts: a review of the literature. Quintessence Int. 2014;45(6):475-487.
10. Matarasso M, Iorio-Siciliano V, Blasi A, et al. Enamel matrix derivative and bone grafts for periodontal regeneration of intrabony defects. A systematic review and meta-analysis. Clin Oral Investig. 2015;19(7):1581-1593.
11. Tonetti MS, Jepsen S. Clinical efficacy of periodontal plastic surgery procedures: consensus report of Group 2 of the 10th European Workshop on Periodontology. J Clin Periodontol. 2014;41 suppl 15:S36-S43.
12. McGuire MK, Cochran DL. Evaluation of human recession defects treated with coronally advanced flaps and either enamel matrix derivative or connective tissue. Part 2: Histological evaluation. J Periodontol. 2003;74(8):1126-1135.
13. Rasperini G, Silvestri M, Schenk RK, Nevins ML. Clinical and histologic evaluation of human gingival recession treated with a subepithelial connective tissue graft and enamel matrix derivative (Emdogain): a case report. Int J Periodontics Restorative Dent. 2000;20(3):269-275.
14. Miron RJ, Bosshardt DD, Laugisch O, et al. Enamel matrix protein adsorption to root surfaces in the presence or absence of human blood. J Periodontol. 2012;83(7):885-892.