Long-term Case Reports Demonstrating Use of Porcine Collagen Matrix to Augment Width and Thickness of Keratinized Gingiva
Robert A. Levine, DDS; Jeffrey Ganeles, DMD; Ron Wang, DMD; and P.D. Miller, DDS
The presence of an adequate zone of attached keratinized tissue is generally believed to be conducive to the maintenance of gingival health.1 The immobile and dense gingival tissue is presumed to act as a barrier that provides better resistance to bacterial and mechanical insults than unattached alveolar mucosa.2 Exactly how much attached tissue is necessary for the maintenance of gingival health has been a controversial subject in the periodontal literature due to the paucity of well-controlled clinical trials.
Lang and Löe demonstrated that in subjects with effective oral hygiene, areas with less than 2 mm of keratinized gingiva experienced persistent inflammation.3 Stetler and Bissada found more inflammation and gingival recession in sites with subgingival restorative margins and less than 2 mm of keratinized gingiva.4 Although numerous studies have shown that periodontal health can be maintained around teeth with minimal keratinized gingiva,5-9 the consensus from the 2014 American Academy of Periodontology Regeneration Workshop suggested that when plaque control is suboptimal, or when potentially traumatic etiologies like subgingival restorative margins or removable prosthesis clasps are present, at least 2 mm of keratinized tissue (1 mm of which is attached gingiva) is necessary to prevent progressive attachment loss and inflammation.10 Absence of this tissue dimension in these circumstances may indicate gingival augmentation therapy. Additionally, emerging evidence on peri-implant health also indicates that the absence of sufficient keratinized tissue around implant-borne prostheses may lead to higher plaque and gingival indices as well as the development of mucosal recession.11-13
The chief goal of gingival augmentation therapy is to create a healthy band of attached keratinized tissue to facilitate plaque control, improve patient comfort, and prevent further gingival recession.1 Since its introduction in the 1960s, the autogenous epithelialized palatal graft (ie, free gingival graft) has been considered the "gold standard" treatment for gingival augmentation procedures.14-18 Transplantation of the graft from either the hard palate or maxillary tuberosity with its keratinized epithelium to the recipient site allows the clinician to increase the width of attached gingiva, deepen the vestibule for hygiene access, eliminate tension from the frenal attachment, relieve brushing discomfort, and treat gingival recession.18 A systematic review by Thoma and colleagues that assessed different gingival augmentation techniques concluded that the use of autogenous tissue (epithelialized palatal graft or subepithelial connective tissue graft [CTG]) with an apically positioned flap procedure significantly increases the width of attached gingiva compared to untreated controls and leads to less graft shrinkage compared to the use of allogeneic dermal tissue.19 A recent long-term study (18 to 35 years) showed that 83% of sites treated with epithelialized palatal graft showed a reduction in gingival recession, whereas 48% of untreated contralateral sites experienced increase in recession.20 The use of epithelialized palatal graft also has been shown to prevent the development or progression of non-carious cervical lesions over a 25- to 30-year observation period.21 Soft-tissue augmentation with autogenous tissue grafts around dental implants has also gained recognition in recent years as an important adjunctive therapy to maintain and improve peri-implant tissue health13,22-24 and marginal bone stability.
Despite its predictability and long-term efficacy, the epithelialized palatal graft has some disadvantages. Harvesting the graft requires a second surgical site, which increases chairtime and postoperative discomfort.25 Anatomical limitations like the shape of the palatal vault and location of blood vessels and nerves dictate and potentially restrict the availability of donor tissue.26,27 Also, the quality of donor tissue may vary depending on the patient's age, gender, and phenotype. The risk of complications, such as damage to neurovascular tissue, uncontrolled hemorrhage, and poor healing of the donor site, should be strongly considered and weighed against potential benefits, especially in patients with clotting deficiencies or systemic conditions that may compromise healing.28 After healing, the grafted site often shows a striking color discrepancy and thickness and texture differences compared to adjacent areas,29-31 possibly leading to an unesthetic result.
Many patients who would benefit from an autotransplantation procedure may be psychologically averse to it because of negative anecdotal reports from friends or relatives, previous pain experienced from the palatal donor site, or the mere thought of the potential discomfort. As a result, they may not seek treatment. In addition, patients who have had autogenous tissue graft treatment in the past and found it uncomfortable may be reluctant to undergo the procedure again.
Soft-Tissue Augmentation Alternatives
The limitations of autogenous tissue grafts have spurred interest in commercially available alternatives, such as allogeneic donor material like acellular dermal matrices, to augment soft tissue. Among newer US Food and Drug Administration-approved alternative materials is a resorbable xenogeneic collagen-based matrix (Geistlich Mucograft®, Geistlich Pharma AG, geistlich-pharma.com) that the authors (RAL, JG) have used extensively in practice. The matrix is made from purified porcine type I and type III collagen and processed without the addition of chemical cross-linkers.32 Its bilayer structure consists of a thin compact outer layer with cell occlusive and tissue adherence properties and a thick porous inner layer that stabilizes the blood clot and serves as a scaffold for host cellular integration.32 As an alternative to autologous soft-tissue grafts, the material has been investigated in the indications of augmenting keratinized tissue around teeth and implants,29-31,33-38 gingival recession coverage,39-43 and coverage of grafted extraction sockets.44-46
In a pilot study by Herford et al, the material's clinical efficacy to treat various types of intraoral mucosal deficiencies was evaluated, and satisfactory healing with minimal complications was achieved.33 The authors noted that the tissue of the grafted sites remodeled to match the phenotype (ie, keratinized or non-keratinized) of the surrounding tissue. The apically positioned flap is a common procedure for mucogingival augmentation, and the collagen matrix may be used to cover the periosteal wound bed to improve healing and regeneration. Several clinical studies have demonstrated that the grafted areas heal with minimal inflammation and complications, and the procedure predictably leads to a functional band of attached keratinized tissue of sufficient width.29-31,33-38 Histologically, the regenerated tissue is indistinguishable from sites grafted with epithelialized palatal graft,29,30,35-37 presenting physiologically normal keratinized epithelium and connective tissue with minimal inflammatory infiltrate. From an esthetic standpoint, unlike with sites augmented with epithelialized palatal grafts where the augmented tissue may have a characteristic "tire-track" appearance, collagen matrix-regenerated tissue has excellent tissue blend in color and texture with the surrounding mucosa.29-31 Benefits of the porcine collagen matrix (PCM) are outlined in Table 1.29,31,33,34
In a prospective clinical study of long-term outcomes by Schmitt and colleagues, mandibular sites augmented with the collagen matrix experienced 52.89% loss of width of keratinized mucosa, compared to 40.65% in the sites grafted with epithelialized palatal graft.31 The investigators observed that the greatest loss of vestibular depth occurred during the first 90 days postoperatively in both the collagen matrix and the epithelialized palatal graft groups, with minimal graft contraction occurring between 1 and 5 years. Despite the statistically significantly greater loss of keratinized mucosa in the collagen matrix group, a clinically sufficient width of keratinized tissue remained after 5 years (mean: 6.15 mm ± 1.23 mm). Based on extensive clinical experience, the present authors estimate a long-term 50% shrinkage of augmented tissue using the collagen matrix, compared to 35% to 40% when using epithelialized palatal grafts.
Two case reports are presented herein in which the collagen-based matrix material was used for soft-tissue augmentation.
Clinical Case 1
The first case involved a healthy, nonsmoking 60-year-old woman whose medical history included American Society of Anesthesiologists (ASA) II classification, penicillin allergy, gastroesophageal reflux disease, and asthma. She presented by referral from her restorative dentist to the primary author (RAL) with concerns of progressive facial gingival recession of her remaining mandibular teeth (Nos. 20 through 29). She had been wearing a maxillary complete denture for many years and was highly concerned about losing her remaining lower dentition. She presented with fair plaque control and no root sensitivity. Her immediate desire was to complete treatment of her maxillary implant-retained removable overdenture with partial palatal coverage.
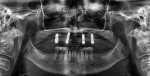
Taking the patient's financial concerns into consideration, her case was treatment-planned in three phases. The first phase entailed completion of a maxillary prosthesis using a restorative-driven team approach that would include placement of four dental implants (Straumann® Tissue Level, Straumann, straumann.com) using a surgical guide for a locator-retained overdenture; the implants were placed on January 26, 2011. The second phase involved periodontal plastic surgery to increase the width and thickness of attached keratinized gingiva at Nos. 20 through 22 and 27 through 29 with no attempt at root coverage since esthetics were not a concern to her and complete root coverage was unattainable because the recession defects were classified as Miller class III or recession type 2.47,48 Even though esthetics were not an issue, increasing the zones and thickness of the attached keratinized gingiva was indicated becausethere was documented increased gingival recession noted in periodontal maintenance on several of the teeth that were to be treated. Presurgery, the amount of remaining keratinized gingiva present on the teeth to be treated was 0 mm to 2 mm. Lastly, the third phase of treatment included restorative-driven single implants to replace missing teeth Nos. 19 and 30 to create first-molar occlusion against the maxillary overdenture.
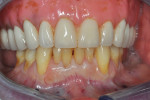
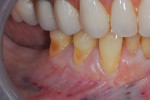
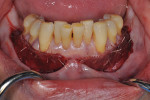
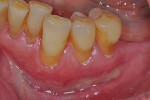
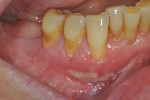
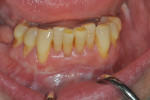
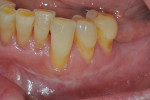
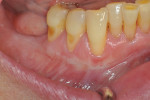
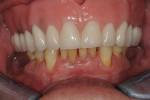
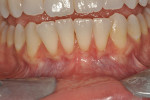
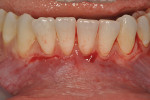
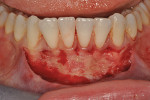
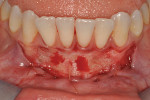
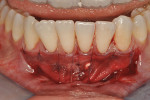
Once the patient was ready to commence phase 2 of her treatment plan (February 7, 2013), two surgical options were discussed with her: (1) augmentation of the tissue using her palate bilaterally as donor sites in one to two surgical visits, which the authors considered a viable option given her new removable partial palatal coverage overdenture; or (2) use of what was at the time a recently introduced PCM (Geistlich Mucograft) to complete treatment in one surgical appointment. Given her low esthetic demands, with root coverage not being a treatment objective, along with her history of compliance of home care and periodontal maintenance every 3 months (alternating with her restorative dentist), she was a candidate for both surgical approaches. On teeth Nos. 20 through 22 and 27 through 29, facial recessions as measured from the cementoenamel junction (mid-buccal) of the teeth to the apical extent of the sulci ranged from 5 mm to 6 mm (Figure 1 through Figure 4).
The patient decided that she wanted to avoid the morbidity involved with the donor surgical site and that completing the procedure in two visits was undesirable to her given her busy work schedule. She opted to have the procedure completed in one visit using the PCM. Based on a discussion with the clinician of the available literature at the time in 2012 she was pleased that the PCM could be incorporated and totally replaced by her own tissue in 4 weeks along with less operative time and morbidity (Table 1).
In one surgical appointment, bilateral inferior alveolar nerve blocks and long buccal infiltrations were administered. Beveled incisions were performed just coronal to the mucogingival junction at the level of the depth of the recessions. Partial-thickness reflection was completed using sharp dissection with a #15c blade from site Nos. 19 through 23 and 25 through 30 so as to incorporate at least one additional tooth on either side of the most anterior and posterior tooth being treated. It should be noted that the surgeon must be fully aware of the location of the mental foramen with regard to incision design coronal to the mental foramen. For both surgical and medico-legal purposes, a full-mouth x-ray examination, panoramic x-ray (as in the present case), or site-specific cone-beam computed tomography scan is necessary presurgically to locate the mental foramina.
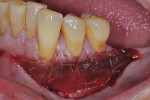
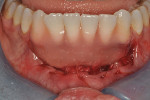
Upon completion of partial flap reflection, a new #15c blade is used to scribe the apical border of the reflection through the periosteum to the osseous cortex. The PCM comes in two sizes with the larger being 20 mm width x 30 mm length. Trimming the material length-wise in half, as was done in this case, typically allows one full piece of the material to treat most patients from teeth Nos. 20 through 29. The PCM was sutured easily to the existing interproximal papillae with resorbable 6-0 plain gut suture. No attempt was made to stabilize the PCM to the periosteum with apical or lateral suturing, as it was deemed unnecessary in this case. Nor was the apical extent of the flap sutured, as this is not needed in most cases. The PCM acts as a postsurgical bandage, therefore the use of periodontal dressing may be a case-by-case decision; in this case no periodontal dressing was needed (Figure 5 through Figure 7). Because the objective of the case was to create a barrier of at least 2 mm to 3 mm of keratinized gingiva to prevent further facial attachment loss, and not root coverage, treatment of sites Nos. 24 and 25 was not necessary as adequate zones and thickness of keratinized gingiva were present.
Postoperative care included rinsing with chlorhexidine gluconate (CHG) twice per day for 2 weeks and use of a cotton tip to clean the buccal cervical areas, with no toothbrushing or flossing for 2 weeks. At 2 weeks, toothbrushing and flossing could be reinstated, which is approximately 4 to 6 weeks sooner than when a CTG is used for root coverage. A water irrigation device can also be reinstated at this time.
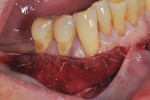
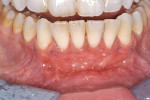
The patient was seen for her first postoperative visit at 2 weeks, at which she received a full-mouth prophylaxis. She was introduced to the use of a soft two-row toothbrush, flossing was reinstated, and the CHG rinse was discontinued (Figure 8 through Figure 10). Another postoperative visit took place at 2 months to review healing and plaque control (Figure 11 through Figure 13). The patient has been in an alternating 3-month periodontal maintenance frequency with her restorative dentist.
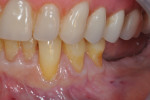
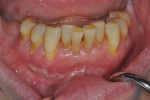
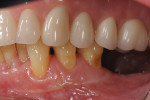
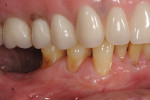
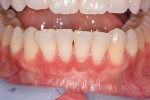
Photographs taken during her 6-year postoperative follow-up, which was her most recent alternating 3-month maintenance visit under the authors' care, demonstrated significant increase in zone and thickness of attached keratinized gingiva (Figure 14 through Figure 16). At 6 years, the attached gingiva gained was as follows: 2 mm gain at No. 20, 3 mm gain at No. 21, 2 mm gain at No. 22, 3 mm gain at No. 27, 2 mm gain at No. 28, and 3 mm gain at No. 29. The authors no longer use CHG in their practice and over the past 3 years have incorporated an oral antioxidant, antibacterial, antifungal non-alcohol mouthwash that the patient uses twice daily and is given routinely to patients presurgically.
Clinical Case 2
A healthy, nonsmoking 28-year-old woman (ASA I) presented for evaluation to the periodontal private practice of one of the authors (JG). She was concerned that her dentist had advised that a frenectomy and gingival graft from her palate were necessary to maintain her mandibular anterior teeth. The patient had a previous history of orthodontic treatment as an adolescent. She had no specific chief complaint other than anxiety about gingival surgery, and she also was highly averse to palatal harvesting for the autogenous gingival graft that was recommended.
Clinical examination showed excellent oral hygiene and virtually no inflammation. It also confirmed the observation of a near total lack of attached keratinized tissue and thin scalloped biotype on teeth Nos. 22 through 27. Incipient recession was observed on these teeth and was classified as Miller class I or recession type 1.47,48 Additionally, a prominent labial frenum pull was noted between teeth Nos. 24 and 25 (Figure 17). Because the patient was adamantly opposed to consenting to autogenous graft harvesting, the PCM was proposed and accepted.
Preoperatively the patient was given 2 g amoxicillin and 1 mg lorazepam orally. Local anesthesia was obtained by infiltration using articaine and bupivacaine with epinephrine 1:200,000. A partial-thickness incision was created just coronal to the mucogingival junction from teeth Nos. 22 through 27 using a #15 scalpel blade and continued to the depth of the vestibule (Figure 18 and Figure 19). Remnants of the labial frenum and fibers from the mentalis muscle were removed to bone. The periosteum was scored to bone near the base of the incision. Mucosal margins of the incision were sutured apically to the periosteum using 5-0 chromic gut sutures (Figure 20).
A piece of collagen matrix was shaped to cover the periosteal bed. It was placed on the bed dry and allowed to rehydrate from tissue fluid and blood before being stabilized into the bases of the papillae with interrupted 6-0 polyglactin sutures (Ethicon, ethicon.com). Additional circumferential sutures anchoring apically to the periosteum, looping around teeth Nos. 24 and 25, were placed to adapt the collagen matrix material against the bed (Figure 21). Periodontal surgical dressing (Barricaid® VLC, Dentsply Sirona, dentsplysirona.com) was then adapted against the teeth and material and cured with a bonding light to further stabilize and isolate the surgical site from the rest of the mouth (Figure 22). Although as noted earlier the PCM acts as a postsurgical bandage, periodontal dressing, though not essential, may be used if the clinician believes it will enhance patient comfort and further stabilize the collagen matrix.
The patient was maintained on amoxicillin 500 mg four times per day for 7 days and instructed to rinse with CHG twice daily. Nonsteroidal anti-inflammatory drugs were prescribed for management of pain.
One week after surgery, the patient returned for postoperative assessment. She was concerned that the surgical area was still quite tender; therefore, the wound dressing was removed and the site gently cleaned with chlorhexidine. A new dressing was placed and light-cured. Eleven days later (18 days after surgery), the patient returned for removal of the second dressing. The area was again cleaned with chlorhexidine and observed (Figure 23). Most of the sutures were loose, and all of them were then removed. Healthy pink tissue proliferation and ingrowth was observed throughout the graft. Maintenance of the vestibuloplasty was confirmed with no indication of the pre-existing frenum pull. The patient was given appropriate instructions to gradually increase intensity of oral hygiene procedures over the next month.
She returned for semiannual routine periodontal supportive care for the next 5 years. At her 5-year examination, a new photograph was taken showing continued health of the site with approximately 50% loss of the vestibular extension depth (Figure 24). A denser, whitish band of scar tissue was noted apically, which was analogous to a new mucogingival junction. There was 3 mm to 5 mm of attached tissue, which appeared to be immobile, keratinized, and healthy. This tissue was somewhat more reddish in color compared to the paler gingiva around the more posterior teeth but had no inflammation. Creeping attachment occurred on teeth Nos. 22 through 27, eliminating the previously observed minor recession. All of the goals of the surgical procedure, including vestibular deepening, creation of an adequate band of attached tissue, and correction of recession, were achieved without harvesting palatal tissue. Color and texture match of the tissue were good, and the patient was very pleased with the result.
Conclusion
There is growing evidence of the importance of soft-tissue dimensions around teeth and dental implants in preventing long-term complications, as the absence of sufficient keratinized tissue and tissue thickness can contribute to persistent inflammation around teeth and implants3,4,11-13,49 and may eventually lead to recession and attachment loss. The use of evidence-based alternatives like porcine collagen matrix has many clinical advantages compared to autogenous palatal grafts when augmenting the width and thickness of attached keratinized gingiva. Benefits include decreased intraoperative discomfort and postoperative pain,36 high patient acceptance and favorable esthetic outcomes,30,31 significantly shortened operation time,31,36 and the ability for clinicians to augment large or multiple sites in one visit.31,35,36 These patient-centered attributes are especially important given that mucogingival defects oftentimes do not cause any symptoms and patients frequently are unaware of them. The option to treat these patients in fewer and shorter visits while minimizing discomfort and disruption to quality of life may be a valuable asset for clinicians to increase patient acceptance. Long-term clinical trials are needed to evaluate PCM stability over time.
Disclosure
Drs. Levine and Ganeles have received honoraria from Straumann and Geistlich. Dr. Wang is an employee of Geistlich.
About the Authors
Robert A. Levine, DDS
Clinical Professor, Kornberg School of Dentistry, Temple University, Philadelphia, Pennsylvania; Diplomate, American Board of Periodontology; Fellow, International Team for Implantology; Fellow, Academy of Osseointegration; Private Practice, Dental Implants and Periodontics, Philadelphia, Pennsylvania
Jeffrey Ganeles, DMD
Adjunct Associate Professor, Nova Southeastern University College of Dental Medicine, Fort Lauderdale, Florida; Adjunct Associate Professor, Boston University, Boston, Massachusetts; Diplomate, American Board of Periodontology; Fellow, International Team for Implantology; Fellow, Academy of Osseointegration; Private Practice, Periodontics and Dental Implant Surgery, Boca Raton, Florida
Ron Wang, DMD
Clinical Science and Education Manager, Geistlich Biomaterials; Private Practice, General Dentistry, Philadelphia, Pennsylvania
P.D. Miller, DDS
Clinical Professor (Ret.), Department of Periodontology, Medical University of South Carolina, Charleston, South Carolina
References
1. Kim DM, Neiva R. Periodontal soft tissue non-root coverage procedures: a systematic review from the AAP Regeneration Workshop. J Periodontol. 2015;86(2 suppl):S56-S72.
2. Nevins M. Attached gingiva-mucogingival therapy and restorative dentistry. Int J Periodontics Restorative Dent. 1986;6(4):9-27.
3. Lang NP, Löe H. The relationship between the width of keratinized gingiva and gingival health. J Periodontol. 1972;43(10):623-627.
4. Stetler KJ, Bissada NF. Significance of the width of keratinized gingiva on the periodontal status of teeth with submarginal restorations. J Periodontol. 1987;58(10):696-700.
5. Miyasato M, Crigger M, Egelberg J. Gingival condition in areas of minimal and appreciable width of keratinized gingiva. J Clin Periodontol. 1977;4(3):200-209.
6. Dorfman HS, Kennedy JE, Bird WC. Longitudinal evaluation of free autogenous gingival grafts. A four year report. J Periodontol. 1982;53(6):349-352.
7. Wennström J, Lindhe J. Plaque-induced gingival inflammation in the absence of attached gingiva in dogs. J Clin Periodontol. 1983;10(3):266-276.
8. Kennedy JE, Bird WC, Palcanis KG, Dorfman HS. A longitudinal evaluation of varying widths of attached gingiva. J Clin Periodontol. 1985;12(8):667-675.
9. Freedman AL, Green K, Salkin LM, et al. An 18-year longitudinal study of untreated mucogingival defects. J Periodontol. 1999;70(10):1174-1176.
10. Scheyer ET, Sanz M, Dibart S, et al. Periodontal soft tissue non-root coverage procedures: a consensus report from the AAP Regeneration Workshop. J Periodontol. 2015;86(2 suppl):S73-S76.
11. Chung DM, Oh TJ, Shotwell JL, et al. Significance of keratinized mucosa in maintenance of dental implants with different surfaces. J Periodontol. 2006;77(8):1410-1420.
12. Lin GH, Chan HL, Wang HL. The significance of keratinized mucosa on implant health: a systematic review. J Periodontol. 2013;84(12):1755-1767.
13. Roccuzzo M, Grasso G, Dalmasso P. Keratinized mucosa around implants in partially edentulous posterior mandible: 10-year results of a prospective comparative study. Clin Oral Implants Res. 2016;27(4):491-496.
14. Nabers JM. Free gingival grafts. Periodontics. 1966;4(5):243-245.
15. Gargiulo AW, Arrocha R. Histo-clinical evaluation of free gingival grafts. Periodontics. 1967;5(6):285-291.
16. Sullivan HC, Atkins JH. Free autogenous gingival grafts. I. Principles of successful grafting. Periodontics. 1968;6(3):121-129.
17. Sullivan HC, Atkins JH. The role of free gingival grafts in periodontal therapy. Dent Clin North Am. 1969;13(1):133-148.
18. Miller PD Jr, Allen EP. The development of periodontal plastic surgery. Periodontol 2000. 1996;11:7-17.
19. Thoma DS, Benić GI, Zwahlen M, et al. A systematic review assessing soft tissue augmentation techniques. Clin Oral Implants Res. 2009;20(suppl 4):146-165.
20. Agudio G, Cortellini P, Buti J, Pini Prato G. Periodontal conditions of sites treated with gingival augmentation surgery compared with untreated contralateral homologous sites: an 18- to 35-year long-term study. J Periodontol. 2016;87(12):1371-1378.
21. Agudio G, Chambrone L, Selvaggi F, Pini Prato GP. Effect of gingival augmentation procedure (free gingival graft) on reducing the risk of non-carious cervical lesions: a 25- to 30-year follow-up study. J Periodontol. 2019;90(11):1235-1243.
22. Thoma DS, Buranawat B, Hämmerle CH, et al. Efficacy of soft tissue augmentation around dental implants and in partially edentulous areas: a systematic review. J Clin Periodontol. 2014;(41 suppl 15):S77-S91.
23. Thoma DS, Naenni N, Figuero E, et al. Effects of soft tissue augmentation procedures on peri-implant health or disease: a systematic review and meta-analysis. Clin Oral Implants Res. 2018;(29 suppl 15):32-49.
24. Buyukozdemir Askin S, Berker E, Akincibay H, et al. Necessity of keratinized tissues for dental implants: a clinical, immunological, and radiographic study. Clin Implant Dent Relat Res. 2015;17(1):1-12.
25. Wessel JR, Tatakis DN. Patient outcomes following subepithelial connective tissue graft and free gingival graft procedures. J Periodontol. 2008;79(3):425-430.
26. Reiser GM, Bruno JF, Mahan PE, Larkin LH. The subepithelial connective tissue graft palatal donor site: anatomic considerations for surgeons. Int J Periodontics Restorative Dent. 1996;16(2):130-137.
27. Monnet-Corti V, Santini A, Glise JM, et al. Connective tissue graft for gingival recession treatment: assessment of the maximum graft dimensions at the palatal vault as a donor site. J Periodontol. 2006;77(5):899-902.
28. Griffin TJ, Cheung WS, Zavras AI, Damoulis PD. Postoperative complications following gingival augmentation procedures. J Periodontol. 2006;77(12):2070-2079.
29. Nevins M, Nevins ML, Kim SW, et al. The use of mucograft collagen matrix to augment the zone of keratinized tissue around teeth: a pilot study. Int J Periodontics Restorative Dent. 2011;31(4):367-373.
30. McGuire MK, Scheyer ET. Randomized, controlled clinical trial to evaluate a xenogeneic collagen matrix as an alternative to free gingival grafting for oral soft tissue augmentation. J Periodontol. 2014;85(10):1333-1341.
31. Schmitt CM, Moest T, Lutz R, et al. Long-term outcomes after vestibuloplasty with a porcine collagen matrix (Mucograft®) versus the free gingival graft: a comparative prospective clinical trial. Clin Oral Implants Res. 2016;27(11):e125-e133.
32. Geistlich Mucograft® [instructions for use]. Wolhusen, Switzerland: Geistlich Pharma AG; 2014.
33. Herford AS, Akin L, Cicciu M, et al. Use of a porcine collagen matrix as an alternative to autogenous tissue for grafting oral soft tissue defects. J Oral Maxillofac Surg. 2010;68(7):1463-1470.
34. Sanz M, Lorenzo R, Aranda JJ, et al. Clinical evaluation of a new collagen matrix (Mucograft prototype) to enhance the width of keratinized tissue in patients with fixed prosthetic restorations: a randomized prospective clinical trial. J Clin Periodontol. 2009;36(10):868-876.
35. Urban IA, Nagy K, Werner S, Meyer M. Evaluation of the combination of strip gingival grafts and a xenogeneic collagen matrix for the treatment of severe mucogingival defects: a human histologic study. Int J Periodontics Restorative Dent. 2019;39(1):9-14.
36. Schmitt CM, Tudor C, Kiener K, et al. Vestibuloplasty: porcine collagen matrix versus free gingival graft: a clinical and histologic study. J Periodontol. 2013;84(7):914-923.
37. Thoma DS, Alshihri A, Fontolliet A, et al. Clinical and histologic evaluation of different approaches to gain keratinized tissue prior to implant placement in fully edentulous patients. Clin Oral Investig. 2018;22(5):2111-2119.
38. Maiorana C, Pivetti L, Signorino F, et al. The efficacy of a porcine collagen matrix in keratinized tissue augmentation: a 5-year follow-up study. Int J Implant Dent. 2018;4(1):1.
39. McGuire MK, Scheyer ET. Xenogeneic collagen matrix with coronally advanced flap compared to connective tissue with coronally advanced flap for the treatment of dehiscence-type recession defects. J Periodontol. 2010;81(8):1108-1117.
40. McGuire MK, Scheyer ET. Long-term results comparing xenogeneic collagen matrix and autogenous connective tissue grafts with coronally advanced flaps for treatment of dehiscence-type recession defects. J Periodontol. 2016;87(3):221-227.
41. Cardaropoli D, Tamagnone L, Roffredo A, Gaveglio L. Treatment of gingival recession defects using coronally advanced flap with a porcine collagen matrix compared to coronally advanced flap with connective tissue graft: a randomized controlled clinical trial. J Periodontol. 2012;83(3):321-328.
42. Camelo M, Nevins M, Nevins ML, et al. Treatment of gingival recession defects with xenogenic collagen matrix: a histologic report. Int J Periodontics Restorative Dent. 2012;32(2):167-173.
43. Rotundo R, Pini-Prato G. Use of a new collagen matrix (mucograft) for the treatment of multiple gingival recessions: case reports. Int J Periodontics Restorative Dent. 2012;32(4):413-419.
44. Jung RE, Philipp A, Annen BM, et al. Radiographic evaluation of different techniques for ridge preservation after tooth extraction: a randomized controlled clinical trial. J Clin Periodontol. 2013;40(1):90-98.
45. Cardaropoli D, Tamagnone L, Roffredo A, et al. Alveolar ridge preservation using tridimensional collagen matrix and deproteinized bovine bone mineral in the esthetic area: a CBCT and histologic human pilot study. Int J Periodontics Restorative Dent. 2018;38(suppl):s29-s35.
46. Fickl S, Kauffmann F, Stappert CF, et al. Scar tissue formation following alveolar ridge preservation: a case control study. Int J Periodontics Restorative Dent. 2018;38(1):e1-e7.
47. Miller PD Jr. A classification of marginal tissue recession. Int J Periodontics Restorative Dent. 1985;5(2):8-13.
48. Cairo F, Nieri M, Cincinelli S, et al. The interproximal clinical attachment level to classify gingival recessions and predict root coverage outcomes: an explorative and reliability study. J Clin Periodontol. 2011;38(7):661-666.
49. Levine RA, Ganeles J, Kan J, Fava PL. 10 keys for successful esthetic-zone single implants: importance of biotype conversion for lasting success. Compend Contin Educ Dent. 2018;39(8):522-529.