Minimally Invasive Extraction Site Management With Dehydrated Amnion/Chorion Membrane (dHACM): Open-Socket Grafting
Daniel Cullum, DDS; and Mark Lucas, DDS, MS
Abstract: Site preservation grafting after tooth extraction offers improved predictability for alveolar volume maintenance and successful delayed implant treatment. Multiple techniques and materials are used for these procedures, and each possesses relative benefits and challenges for the clinician and patient. In the case reports presented, the patients were treated with minimally invasive open extraction site preservation with dehydrated amnion/chorion membrane (dHACM) at pontic sites and prior to delayed implant therapy. The biologic rationale for material selection will be reviewed.
Post-extraction site preservation grafting is often indicated to ensure adequate alveolar bone dimensions at pontic sites and effective delayed implant placement to prevent significant loss of ridge volume over time.1 Numerous graft materials and barrier membranes have been cited for use in such procedures.2 Open (non-primary closure)-site preservation with dense polytetrafluorethylene (dPTFE) is an established and well-documented site preservation technique.3 Upregulation of wound healing has been advocated for medically compromised patients and/or compromised wounds using materials that possess signaling molecules to enhance new tissue formation, as well as to modulate postoperative inflammation and patient discomfort.4 Autologous bone and adjuncts, including platelet-rich plasma, plasma rich in growth factors, and platelet-rich fibrin preparations, have been investigated for use in site preservation procedures to provide signaling molecules capable of accelerated wound healing.5-7
This continuum of biomaterial development includes recombinant human proteins such as rhBMP-2 and rhPDGF-BB.8,9 Dehydrated de-epithelialized human amnion/chorion also has been described as an adjunct containing a wide array of signaling proteins that can facilitate wound healing, as well as regulate inflammation and pain.10-12 The importance of patient-centered outcomes, using minimally invasive procedures with diminished postoperative pain and reduced patient visits, has been emphasized with regard to research outcomes.13 The following cases describe open-site preservation utilizing particulate bone grafting and dehydrated amnion/chorion membrane (dHACM).
(Editor's note: A second article from the present authors on minimally invasive extraction site management with dHACM is tentatively scheduled for an upcoming issue of Compendium.It will discuss immediate implant placement with dynamic navigation.)
Case 1
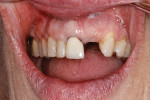
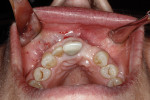
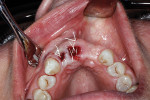
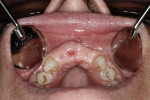
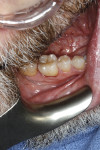
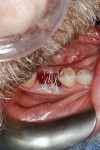
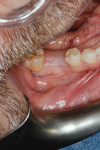
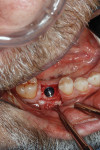
Application of the dHACM in the esthetic zone was demonstrated in a 59-year-old female patient with a failing anterior bridge who was requesting removal of the maxillary right central and lateral incisors (Figure 1 through Figure 3). The patient opted to have these teeth removed to allow for correction of a significant midline asymmetry and space inadequacy using a tooth-supported fixed partial bridge with pontics to replace the edentulous areas.
Atraumatic removal of the teeth was followed by site assessment with complete curettage and debridement. The defects were grafted with particulate mineralized xenograft (InterOss®, SigmaGraft, Inc, sigmagraft.com) blended 2:1 with dense hydroxylapatite (OsteoGraf/D300®, Dentsply Sirona, dentsplysirona.com) for optimal long-term volume maintenance. The graft particles were saturated in autologous venous blood and packed to the residual bone margins (Figure 4) using specialized grafting instruments (H & H Company, hhcompany.store).
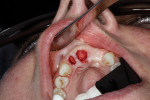
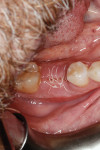
The sites were each covered with a single layer of 8-mm x 8-mm dHACM (BioXclude®, Snoasis Medical, snoasismedical.com) placed over the graft material (Figure 5). These membranes were positioned with saline rinse and tucked into the gingival sulcus. A 4-0 PTFE suture (Cytoplast™, Osteogenics Biomedical, osteogenics.com) was passed as an inverse "figure 8" starting on the mesial, from the buccal aspect through the palatal gingival margin, then through the palatal aspect of the buccal and repeated on the distal aspect and tied (Figure 6). This suture technique diminishes the possibility of the suture needle engaging or displacing the membrane and provides excellent stabilization.
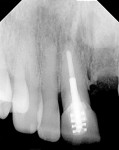
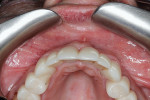
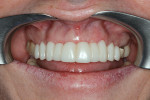
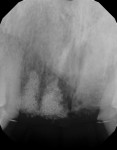
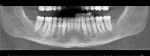
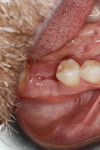
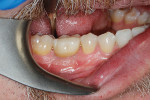
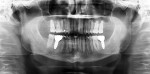
In Figure 7 ridge healing and bone volume can be seen after 25 days. Figure 8 through Figure 10 show the results at 11 months with the provisional restoration in position and a periapical radiograph of the site.
Case 2
Thissite preservation technique was further demonstrated in a 61-year-old male patient with a noncontributory medical history, who presented with a nonrestorable right mandibular first molar (Figure 11 and Figure 12). The patient chose treatment that utilized a delayed approach.
The tooth was removed in an atraumatic fashion using root sectioning and periotome-style elevators (X-Tool, H & H Company) for maximum site preservation. Site assessment with a periodontal probe showed intact bone margins and soft-tissue granulations that were excised with a micro-rongeur. Particulate cortico-cancellous allograft material (enCore®, Osteogenics Biomedical) was blended 1:1 with particulate xenograft (BioOss®, Geistlich Biomaterials, geistlich-na.com) saturated in autologous venous blood and packed with specialized grafting instruments to the surrounding bone margins (Figure 13).
A dHACM membrane (BioXclude) was trimmed, placed in the wound, saturated with sterile saline to seal over the defect, and stabilized with two inverse "figure 8" PTFE sutures (Cytoplast) (Figure 14). The suture was passed from the buccal aspect through the lingual marginal gingiva and then through the lingual aspect of the buccal tissue. This was repeated more distal (at about the mid-point of the socket) and tied. A second suture was then placed more distal, completing the stabilization of the dHACM in position.
The patient was instructed to discontinue chlorhexidine rinse and dentifrice usage for 7 days and to brush teeth with tap water only. He also was told to continue taking oral antibiotics for 5 days. The patient was seen at 10 days postoperative, where the grafted site demonstrated fibrin deposition with minimal inflammation (Figure 15). A second postoperative visit took place at 28 days, during which complete granulation and maturing epithelium was evident (Figure 16).
After 6 months at implant surgery, healing of the site demonstrated good soft-tissue and bone contours preoperatively (Figure 17), and significant buccal bone volume was demonstrated at single-stage implant placement (Figure 18). A wide-platform fixture, 5.5 mm diameter (NobelParallel™, Nobel Biocare, nobelbiocare.com), was used. A flared (7 mm x 8 mm) polyether ether ketone (PEEK) healing abutment was placed and the flap was secured with apical flap positioning.
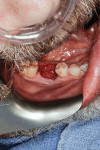
Six months after implant restoration the site showed excellent bone and soft-tissue contours (Figure 19 and Figure 20).
Perioperative Management
Perioperative patient management using exposed dHACM requires some deviation from the use of traditional materials. Chlorhexidine rinse is recommended starting 3 days preoperatively (rinsing for 2 minutes twice per day) and then discontinued after surgery. Due to the charged bioactive proteins present in dHACM, postoperative management requires avoidance of any surface-active agents or chemicals, including chlorhexidine, hydrogen peroxide, baking soda, or alcohol-containing mouthrinses. The larger the exposed surface area of the membrane, such as with a molar-sized defect compared to a premolar, the more important this issue becomes. Similarly, while the use of PTFE sutures typically are well-tolerated, the preservative chemicals of chromic gut sutures and periodontal dressings can be detrimental to healing with dHACM.
Patients are instructed to rinse gently and toothbrush with tap water for 7 days while the initial fibrin clot and epithelium are forming. The wounds typically close over quickly, especially in single-rooted tooth sites.
Discussion
Open-socket grafting techniques have the benefit of avoiding flap elevation and preserving the periosteal blood supply, allowing for a minimally invasive approach with maintenance of vestibular depth and increase in the amount of keratinized tissue. The selection of various bone grafting materials can influence the turnover time and volume maintenance at grafted sites to allow rapid re-entry for implantation or enhance volume maintenance at a pontic site as demonstrated in case 1. Typically, mineralized cancellous allografts heal in 3 to 4 months, mineralized xenografts heal in 6 months or more, and dense hydroxylapatite displays no significant resorption.
The rationale for using PURION® processed (MiMedx, mimedx.com) dehydrated de-epithelialized human amnion/chorion membrane is based on placental tissues possessing both a collagen-based extra-cellular matrix as well as soluble signaling molecules, which provides for accelerated healing, antimicrobial action, and anti-inflammatory components.14 Although dHACM contains cellular material such as cell membrane-associated proteins and intracellular proteins, the immune-privileged status of this material does not elicit a foreign-body reaction.15 Additionally, the intra- and extracellular proteins contained in the membrane have been shown to downregulate the inflammatory response in the applied area and serve as a chemotactic source for hematopoetic and mesenchymal stem cells.16-18 Due to the reduced immune response, patients treated with these tissues generally experience less postoperative pain.12,19 A comparative study examining open-socket grafting outcomes using dHACM and dPTFE membranes revealed significantly less postoperative pain with the use of dHACM in the early postoperative healing days 1 through 3.12 Additionally, the dHACM-treated sites demonstrated significantly more osteoid formation with higher bone-volume density than the dPTFE-treated sites at the implant placement visits that occurred approximately 3.5 months after grafting.12
Studies employing placental membranes have shown increased levels of bone induction compared to controls during vestibuloplasty procedures as well as higher levels of bone formation in critical size bone defects when compared to control sites treated with no membrane or a traditional xenograft collagen membrane.20,21
Placental tissues contain natural antimicrobial agents, such as human beta defensins, that guard against microbial proliferation.22 dHACM also contains extracellular matrix proteins, which facilitate cellular migration.23 Use of the PTFE inverse "figure 8" suturing technique, passing from inside the wound from the periosteal side through the epithelium, minimizes potential displacement of the barrier. Postoperative care management varies from traditional open-socket grafting methods in that any surface-active chemicals that can damage the dHACM or inhibit epithelial migration or wound healing are avoided.
The thin and pliable nature of the hydrated dHACM precludes the need for custom trimming the membrane to fit the grafted defect site as is required for traditional collagen membranes. The dHACM can fold onto itself, become corrugated, and approximate the incision margins without affecting its ability to close the wound with reduced intraoperative time. These same qualities of the dHACM, however, reduce its capacity to be sutured and tacked into place. Additionally, the relative decrease in total fibrous content of the dHACM prohibits its ability to maintain space independent of graft material. The dHACM can serve as an adjunct to promote healing when other biologically inert membranes are used.24
Conclusion
Site preservation grafting methodology continues to progress with new materials and techniques. The use of dHACM with signaling molecules and a minimally invasive surgical approach reduces inflammation and pain and enhances open-socket grafting healing. The relatively economical availability of dHACM without the need for harvesting autologous tissues or venipuncture and processing is a significant advance for socket grafting procedures. Patient outcomes with pontic site maintenance and delayed implant reconstruction demonstrated excellent bone and soft-tissue contours in the case reports presented here. Clinicians should consider selecting materials and techniques for patient-centered outcomes, including minimally invasive protocols that enhance healing, reduce the number of visits, and diminish postoperative pain. Further clinical investigation into this exciting area of dentistry is indicated.
Disclosure
Dr. Cullum is a consultant with Snoasis Medical, and Dr. Lucas is Vice President of Research & Development with Snoasis Medical.
About the Authors
Daniel Cullum, DDS
President, Implants Northwest LIVE Learning Center, Coeur d'Alene, Idaho; Visiting Lecturer, University of California Los Angeles and Loma Linda University Departments of Oral and Maxillofacial Surgery; Consultant with Snoasis Medical, Golden, Colorado; Private Practice specializing in Oral and Maxillofacial Surgery, Coeur d'Alene, Idaho
Mark Lucas, DDS, MS
Clinical Assistant Professor, University of Colorado School of Dental Medicine, Aurora, Colorado; Vice President of Research & Development, Snoasis Medical, Golden, Colorado
References
1. Van der Weijden F, Dell'Acqua F, Slot DE. Alveolar bone dimensional changes of post-extraction sockets in humans: a systematic review. J Clin Periodontol. 2009;36(12):1048-1058.
2. Troiano G, Zhurakivska K, Lo Muzio L, et al. Combination of bone graft and resorbable membrane for alveolar ridge preservation: a systematic review, meta-analysis, and trial sequential analysis. J Periodontol. 2017;1-17. doi: 10.1902/jop.2017.170241.
3. Hoffman O, Bartee BK, Beaumont C, et al. Alveolar bone preservation in extraction sockets using non-resorbable dPTFE membranes: a retrospective non-randomized study. J Periodontol. 2008;79(8):1355-1369.
4. Kou X, Xu X, Chen C, et al. The Fas/Fap-1/Cav-1 complex regulates IL-1RA secretion in mesenchymal stem cells to accelerate wound healing. Sci Transl Med. 2018;10(432). doi: 10.1126/scitranslmed.aai8524.
5. Marx RE. Platelet-rich plasma (PRP): what is PRP and what is not PRP? Implant Dent. 2001;10(4):225-228.
6. Anitua E. Plasma rich in growth factors: preliminary results of use in the preparation of future sites for implants. Int J Oral Maxillofac Implants. 1999;14(4):529-535.
7. Choukroun J, Adda F, Schoeffler C, Vervelle A. An opportunity in perio-implantology: the PRF. Implantodontie. 2001;42:55-62.
8. Misch CM. The use of recombinant human bone morphogenetic protein-2 for the repair of extraction socket defects: a technical modification and case series report. Int J Oral Maxillofac Implants. 2010;25(6):
1246-1252.
9. Nevins ML, Reynolds MA. Tissue engineering with recombinant human platelet-derived growth factor BB for implant site development. Compend Contin Educ Dent. 2011;32(2):18-27.
10. Holtzclaw D, Toscano N. BioXclude placental allograft tissue membrane used in combination with bone allograft for site preservation: a case series. J Implant Adv Clin Dent. 2011;3(3):35-50.
11. Wallace S, Cobb C. Histological and computed tomography analysis of amnion-chorion membrane in guided bone regeneration socket augmentation. J Implant Adv Clin Dent. 2011;3(6):61-72.
12. Hassan M, Prakasam S, Bain C, et al. A randomized split-mouth clinical trial on effectiveness of amnion-chorion membranes in alveolar ridge preservation: a clinical, radiologic, and morphometric study. Int J Oral Maxillofac Implants. 2017;32(6):1389-1398.
13. Fleming PS, Koletsi D, O'Brien K, et al. Are dental researchers asking patient-important questions? A scoping review. J Dent. 2016;49:9-13.
14. Chen EH, Tofe AJ. A literature review of the safety and biocompatibility of amnion tissue. J Implant Adv Clin Dent. 2009;2(3):67-75.
15. Warning JC, McCracken SA, Morris JM. A balancing act: mechanisms by which fetus avoids rejection by the maternal immune system. Reproduction. 2011;141(6):715-724.
16. Park CY, Kohanim S, Zhu L, et al. Immunosuppressive property of dried human amniotic membrane. Opthalmic Res. 2009;41(2):112-113.
17. Koob TJ, Lim JJ, Massee M, et al. Angiogenic properties of dehydrated human amnion/chorion allografts: therapeutic potential for soft tissue repair and regeneration. Vasc Cell. 2014;6:10.
18. Maan ZN, Rennert RC, Koob TJ, et al. Cell recruitment by amnion chorion grafts promotes neovascularization. J Surg Res. 2015;193(2):953-962.
19. Velez I, Parker WB, Siegel MA, Hernandez M. Cryopreserved amniotic membrane for modulation of periodontal soft tissue healing: a pilot study. J Periodontol. 2010;81(12):1797-1804.
20. Samandari MH, Adibi S, Khoshzaban A, et al. Human amniotic membrane, best healing accelerator, and the choice of bone induction for vestibuloplasty technique (an animal study). Trans Res Risk Man. 2011;3:1-8.
21. Koushaei S, Samandari MH, Razavi SM, et al. Histological comparison of new bone formation using amnion membrane graft versus resorbable collagen membrane: an animal study. J Oral Implantol. 2018;44(5):335-340.
22. King AE, Paltoo A, Kelly RW, et al. Expression of natural antimicrobials by human placenta and fetal membranes. Placenta. 2007;28(2-3):161-169.
23. Niknejad H, Peirovi H, Jorjani M, et al. Properties of the amniotic membrane for potential use in tissue engineering. Eur Cell Mater. 2008;15:88-99.
24. Holtzclaw D. Dehydrated human amnion-chorion barrier used to assist mucogingival coverage of titanium mesh and rhBMP-2 augmentation of severe maxillary alveolar ridge defect: a case report. J Imp Adv Clin Dent. 2016;8(4):6-16.