Improved Yale Total Tic Severity Score Due to Craniofacial Manipulation With an Oral Appliance
Timothy L. Hottel, DDS; Robin L. Jack, MD; Elliott Taynor, DDS; Martha Wells, DDS; Wainscott Hollis, DDS; Robert Brandt, DDS; Stefan A. Hottel; and William L. Balanoff, DDS
ABSTRACT
Background: This study evaluated a novel treatment approach using the concept of cranial facial manipulation with an oral appliance, the TicTocStop Tic Guard (TTSTG), also known as the Tic Guard, to determine its safety and efficacy in the treatment of Tourette syndrome (TS) and chronic tic disorders (CTDs). Methods: A two-center study was undertaken at the University of Tennessee Health Science Center and a facility on Long Island, New York. A total of 77 subjects enrolled between the centers upon referral from local neurologists and via Internet enrollment; 67 subjects started the study, and 58 completed the study. A licensed Cognitive Behavioral Intervention for Tics (CBIT) determined the subjects' Yale Total Tic Severity Score (YTTSS) for tics at enrollment, after 1-week sham appliance, after 1-week Tic Guard, and after 10-weeks Tic Guard. Results: The results showed statistically significant improvement in the YTTSS with both the sham appliance and the Tic Guard with more robust improvement with the Tic Guard. The sham appliance resulted in a 25% overall reduction in tic severity, while the Tic Guard resulted in a 39% overall reduction in tic severity. There were no serious adverse events reported with either device. Conclusions: This study demonstrated significant reduction in YTTSS using the Tic Guard with no reported serious adverse events. As a result, this device could be considered for inclusion in treatment modalities offered to patients with TS/CTD.
Once considered rare, Tourette syndrome (TS) and chronic tic disorders (CTDs) are increasingly recognized among school-aged children. These conditions are characterized by the presence of involuntary and repetitive vocalization and/or movement, known as tics, the onset of which can be spontaneous or may be preceded by premonitory urge. TS is defined by the presence of multiple motor tics and at least one vocal tic present for more than 1 year.1 CTDs include motor tics or vocal tics (but not both) present for more than 1 year. Common motor tics involve muscles of the face, neck, and shoulders, while all vocal tics involve the oral and pharyngeal structures. With poor or inadequate management of the symptoms of TS, patients often are socially isolated and emotionally distressed and may have difficulty in school or maintaining employment. Some experience physical pain from intense complex tics. This syndrome currently has no cure, and the etiology remains unclear. Current estimates are that one out of 160 children or 0.6% between the ages of 5 and 17 in the United States have TS.1
Methods
Participants
This study was conducted at two investigation sites: the University of Tennessee Health Science Center, Memphis, and a facility on Long Island, New York. Patients were assessed for eligibility at the two centers upon referral from local neurologists and via Internet enrollment.
Patient eligibility criteria was as follows: (1) aged 7-45 years old and previously diagnosed with TS or CTD by a licensed medical doctor; (2) experienced tic severity of moderate to severe as measured by Yale Total Tic Severity Score (YTTSS), total score greater than 19 for those diagnosed with TS, and greater than 12 for those diagnosed with CTD; children with motor or vocal tics only; (3) fluent in English to participate in consent and assessment procedures; (4) patients with associated behaviors such as obsessive compulsive disorder (OCD), attention deficit hyperactivity disorder, anxiety, and/or depression were eligible unless the condition required immediate medical treatment or change in current medication; (5) participants receiving medications for tics were eligible if the dose was stable with no planned changes; (6) able to tolerate the oral orthotic; (7) agreed to follow-up care as advised; (8) IQ estimate of 70 or higher; (9) agreed to being subject to videotaping and other recording devices; and (10) patients and guardians agreed to allow the patient's medical and dental records to be made available for review.
Exclusion criteria included: (1) significant other medical, psychological, or neurological disease or disorders that could interfere with study assessments and/or wearing the oral orthotic device; (2) oral hygiene, loose teeth, or dental health concerns/issues that would miss-fit the device; not cleared by a dentist; (3) planned change of medications; patient not stable on medications; (4) did not consent to videotaping; or (5) did not want his/her medical or dental records made available for review.
Patients were removed from the study due to: (1) failure to show for appointments; (2) failure to wear the appliance; (3) change in medication during the course of treatment; or (4) commencement of orthodontic treatment.
Standard Protocols, Approvals, Registrations, and Patient Consents
Approval was granted through the University of Tennessee Institutional Review Board, and the study was registered at ClinicalTrials.gov, ID NCT02599519. Patients who met all inclusion and exclusion criteria reviewed the study's written consent form. Parents or legal guardians reviewed and signed the informed consent for patients under 18 years of age. Patients enrolled were assigned subject identifiers.
Study Design and Procedures
The TicTocStop Tic Guard (TTSTG), or Tic Guard, was evaluated in a single pivotal 12-week clinical trial. The study was designed with a 12-week interval to avoid the disruption of skeletal growth and in anticipation of any tooth loss, which could require adjustments to be made to the Tic Guard. A placebo device (standard mouthguard orthotic) was worn for 1 week, followed by the wearing of the Tic Guard for 1 week. The 1-week period using each device was selected based on unpublished pilot study data that demonstrated the short amount of time required to observe improvement in the number and/or severity of the subjects' tics. The 1-week sham/placebo run-in period established the placebo effect or efficacy observed in patients wearing a standard mouthguard. An additional 10-week period of the active Tic Guard orthotic followed this to assess patient compliance; identify safety issues such as headaches, migraines, temporomandibular joint (TMJ) symptoms, oral sore spots, and appliance breakdown; and track the reduction of tics over an extended period of use. After the 10-week active appliance trial, the subjects attended their final tic assessment.
For each study subject, a baseline evaluation was completed in a private room that included the YTTSS administered by trained CBIT (licensed Cognitive Behavioral Intervention for Tics). The personnel recording the YTTSS observations remained unchanged at both clinical sites throughout the study. Additionally, sessions were videotaped for further objective documentation of the subjects' YTTSS scoring. Subjects underwent a cone-beam computed tomography (CBCT) radiograph to demonstrate and view the lower two-thirds of the head from infraorbital rim to inferior of the mandible. A boarded dental radiologist assessed the radiographs and established the skeletal relationship of each subject. Patients were then scheduled for their Tic Guard custom dental appliance.
Follow-up visits occurred at 1-week post sham insertion to evaluate the effect of the sham and place the active device, followed by an additional 1-week visit to check the active device for fit and possible oral irritation. Then a 10-week follow-up with the active appliance took place. At each visit, the following was performed: oral examination to assess oral and dental health with and without the orthotic; YTTSS to assess tics with the orthotic device; reporting of unanticipated problems; reporting of any change in current prescribed medications; and scheduling of the next visit. Each assessment was videotaped. At the final visit, as a benefit for participating in the study, the subject was allowed to keep the oral orthotic if he/she chose to do so.
At each visit in which the YTTSS was taken, patients discussed with the assessor how often they wore the sham comparator or Tic Guard device. Because the device is worn only when the patient wants to control the urge to tic and not 24 hours a day, patients were not required to maintain a log.
Outcome
The primary endpoint of the pivotal study was assessment of changes in the YTTSS. The YTTSS is a clinical rating instrument designed for use in studies of TS and other tic disorders.2 The questionnaire is designed to identify symptoms of motor and phonic tics, severity, and age of onset; assess OCD symptoms, severity, and age of onset; and identify environmental effects on symptoms. The reliability and validity of the Yale Global Tic Severity Scale, including the YTTSS portion of the scale, have been previously established in adults and children.3 The primary endpoint was measured at screening, end of the placebo 1-week run-in period, and after weeks 1 and 11 where the patient wore the Tic Guard. In addition to evaluating tic severity and frequency, the appliance was evaluated for safety, including subject's health and the durability of the appliance. The study design threshold was a 27% decrease in overall tics. The 27% reduction in tics was chosen because it was considered clinically meaningful to the patient.
Each of the separate dimensions (motor and vocal) were secondary measurements of efficacy. The major secondary endpoint was safety and efficacy changes in the YTTSS for the Tic Guard at week 11.
Sham Comparator: Occlusal Bite Guard
The occlusal bite guard oral appliance used as a sham comparator has a lower vertical dimension than the Tic Guard. The occlusal bite guard was used to demonstrate that an oral appliance (such as a night/occlusal guard) would not elicit enough of a clinically significant improvement (pre-specified to be >27% when measured on the YTTSS) in tic severity or frequency to make the clinical claim that a sensory "trick" is occurring in the subject's mouth and that a specific vertical dimension of the Tic Guard is what has made the difference. The tics were measured using the YTTSS. A tic measurement with the occlusal bite guard was made at initial insertion of the appliance on the second visit and again at the 1-week follow-up, prior to inserting the Tic Guard.
Investigational Appliance: Tic Guard
The Tic Guard is an oral appliance made to a specific vertical dimension. The appliance opens the vertical dimension between the upper and lower jaw. The height is determined by adjusting the vertical dimension in 1-mm increments. After the increase in vertical dimension is attained, the tic severity and frequency is assessed. Once a vertical dimension is established that reaches a threshold of maximum medical improvement, a bite registration is taken using fast set polyvinylsiloxane and the definitive appliance is fabricated.
Statistical Analysis
Analyses were conducted using SAS for Windows version 9.3 or later. Data was summarized using descriptive statistics. For continuous measures, this included number (n), mean, standard deviation (SD), median, and range (minimum and maximum). For categorical measures, counts and percentages were provided. All statistical tests were conducted at α = 0.05 with two-sided significance level. The first day of placebo run-in was considered as Day 1, and the day prior to placebo run-in was Day -1. Summaries were based on the database visits, regardless of the relative day of the visit.
The primary parameters were the YTTSS (range 0 to 50), defined as the sum of the Motor Tic Severity Score (range 0 to 25) and the Vocal Tic Severity Score (range 0 to 25). Each separate dimension (motor and vocal) was a secondary parameter. Adverse events and device deficiencies also were collected.
The null hypothesis was that a placebo orthotic and the Tic Guard orthotic are not different in mitigating the severity and frequency of tics associated with medically diagnosed TS and CTD when measured using the YTTSS. The parameters of the design dictating the recommended sample size of 65 patients were based upon the following measurements: significance level: 0.05; SD: 0.5; power number: 0.80; minimal detectable difference: 0.27; dropout rate: 14%.
Results
A total of 77 subjects enrolled in the study, 67 (87%) started the study, and 58 (75%) completed the study. Nine (13.4%) subjects discontinued from the study. Ten of the 77 patients never received or wore the standard mouth guard or the Tic Guard. The reason for discontinuation was that the subjects were lost to follow-up (ie, did not return for Visit 2 or the Follow-up Visit).
The mean (SD) age of subjects was 15.5 (8.56) years. The majority of subjects were male (74.6%) and white (94%). The average (SD) age when motor tics and vocal tics first occurred was 6.9 (3.12) years and 8 (3.48) years, respectively. The average (SD) age when worst motor tics and vocal tics first occurred was 11.4 (6.57) years and 11.2 (5.49) years, respectively. A total of 36 (53.7%) subjects used concomitant medications.
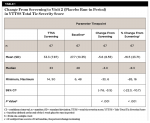
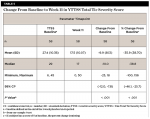
At Visit 2, subjects received their placebo/sham orthotic and started the placebo run-in period. During this 1-week period, the mean (SD) change from screening in the total tic severity score was -5.6 (8.55). This change represented a mean percentage change of -16.5% (23.71%) from the screening score (Table 1).
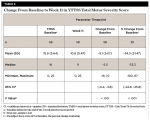
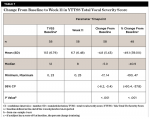
By Week 3, a statistically significant mean (SD) decrease was observed in the total tic severity score: -6.6 (7.75), P < .001. This change represented a mean (SD) percentage decrease from baseline of 22.1% (44.03%) (Table 2). By Week 3, a statistically significant mean (SD) decrease was observed in the total motor severity score: 6.6 (7.75), P < .001. This change represented a mean (SD) percentage decrease from baseline of 22.1% (44.03%) (Table 3). By Week 3, a statistically significant mean (SD) decrease was observed in the total vocal severity score: 3.6 (5.05), P < .001. This change represented a mean (SD) percentage decrease from baseline of 23.9% (31.54%) (Table 4).
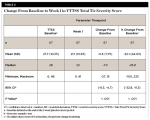
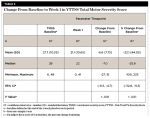
By Week 11, a statistically significant mean (SD) decrease was observed in the total tic severity score: -9.9 (8.13), P < .001. This change represented a mean (SD) percentage decrease from baseline of 35.9% (38.70%). This also represented a median (SD) change of 38.8% from baseline (Table 5). By Week 11, a statistically significant mean (SD) decrease was observed in the total motor score: -5.1 (5.07), P < .001. This change represented a mean (SD) percentage decrease from baseline of 34.3% (31.47%) (Table 6).
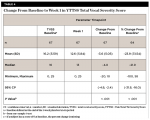
By Week 11, a statistically significant mean (SD) decrease was observed in the total vocal score: -4.8 (5.43), P < .001. This change represented a mean (SD) percentage decrease from baseline of 49.1% (39%) (Table 7).
Discussion
Several treatment options for tics associated with TS and CTD exist, including prescription drugs, CBIT,4 and deep brain stimulation (DBS).5 DBS has not been cleared by the US Food and Drug Administration for the treatment of TS, and it has had some setbacks with regard to CTD because of the surgical protocol. The three most commonly prescribed drugs for TS and CTD are medications that are commonly used to treat other maladies: clonidine (high blood pressure), topiramate (anti-seizure/anti-convulsant), and pimozide (anti-psychotic). Unfortunately, these drugs can have severe adverse side effects, and most child neurologists try to avoid pharmacologic treatments unless tics are significantly interfering with the child's academic or social functioning or causing physical pain. Non-pharmacologic therapies, such as CBIT, have advanced the treatment and understanding of TS/CTD and provided patients a means of control over tics without exposure to medication side effects.
Sutcher and colleagues were the first to describe the use of an oral appliance for the treatment of movement disorders,6 and anecdotal clinical reports describe the elimination of tics associated with TS by changing the vertical dimension or relationship of a patient's maxilla and mandible. The orthotic oral appliance's mechanism of action for decreasing tics is likely through its effects on the gross and neuroanatomy of the TMJ. The TMJ is the general area where the mandible connects to the temporal bone of the skull via a series of tendons, ligaments, and muscles. The auriculotemporal nerve is the main sensory nerve supply to the TMJ, with contributions from the masseteric and deep temporal nerves that come off the mandibular nerve at the same level as the auriculotemporal nerve. Most of the nerve endings are contained in the loose vascular connective tissue located posterior to the TMJ disc called the retrodiscal pad. There are three types of nerve receptors in the TMJ: un-encapsulated or Ruffini endings, encapsulated or Pacinian corpuscles, and free nerve endings.7 The collective role of these receptors is perception of pain and joint position, and the proprioception sensory fibers are critical to the mechanism of action of the appliance study. Thus, the action of separating the posterior opposing teeth (typically in excess of 4 mm to 5 mm) causes the condyle to rotate within the condylar fossa of the TMJ and translate down the articulate eminence of the temporal bone. Opening the bite in this way creates neurostimulation of the sensory fibers of the joint capsule to create a shutoff mechanism in the brain to prevent the motor nerves from leaving the brain to fire. The signal received by the brain via the Vth cranial nerve reduces or eliminates the motor signal from the brain to the III, IV, V, VI, VII, IX, and XI cranial nerves.
The development of a non-invasive, minimal side-effect device that is proven to effectively eliminate or minimize tics would greatly benefit patients with TS and CTD. The present research details the results from a clinical study evaluating the effectiveness of the oral orthotic device, the TicTocStop Tic Guard, developed by TicTocStop, Inc, against a sham/placebo device. The authors monitored the patients' dental/oral health, measured their baseline symptoms, and compared these findings after use of the oral orthotic device and sham. The objective of this study was to: (1) evaluate the safety of a custom-fitted oral orthotic device in patients with TS or CTD, and (2) evaluate and demonstrate the efficacy of an oral orthotic device to decrease motor and vocal tic severity in patients with TS or CTD.
This study assessed the effectiveness of the Tic Guard in mitigating motor and vocal tics in children and adults with TS and CTD. The results shown here describe a statistically significant improvement in the YTTSS with both the sham appliance and the Tic Guard with more robust improvement with the Tic Guard. The sham appliance resulted in a 25% overall reduction in tic severity and frequency while the Tic Guard resulted in a 39% overall reduction in tic severity and frequency. No serious adverse events were reported with either device. Because this study demonstrated significant reduction in YTTSS using the Tic Guard with no reported serious adverse events, this device could be added to the treatment modalities offered to patients with TS/CTD.
Safety was assessed by adverse events and device deficiencies at each appointment. Patients were evaluated for adverse events such as headaches, migraines, TMJ symptoms, oral sore spots, appliance break down, and device defects. Adverse events were limited to two reports of the patient reporting the mouth was uncomfortable to open while wearing the orthotic devices.
The study does not answer "why" the Tic Guard works to reduce motor and vocal tics in children and adults with TS and CTD, and this still remains an area of active investigation. The information obtained from the radiographs will give the authors insight into the skeletal relationship of the mandible to the cranium.
Conclusion
This study clearly demonstrated that tics in all test subjects were mitigated by optimizing the opening of the vertical dimension. Understanding where the condyle is in relationship to the articulate eminence and fossa will provide guidance as to "why" this works effectively. The long-term implications outpace this study design. Perhaps a treatment modality would be craniofacial surgery to correct a maxilla/mandibular discrepancy. Possibly, interceptive orthodontics might be appropriate on patients with a family history of TS and a propensity toward a class II occlusion; this would be a patient population that would require early screening and treatment.
Disclosures
This work was supported through a grant funded by TicTocStop, Inc, New York, New York, Non-Profit 501C3. Dr. Balanoff is chief medical officer of the funding agency for this research but had no part in the clinical patient treatment or collection of data. The authors report no other conflicts.
About the Authors
Timothy L. Hottel, DDS
Professor, College of Dentistry, University of Tennessee Health Science Center, Memphis, Tennessee
Robin L. Jack, MD
Associate Professor, University of Tennessee Health Science Center, Le Bonheur Children's Hospital, Memphis, Tennessee
Elliott Taynor, DDS
Adjunct Assistant Professor, College of Dentistry, University of Tennessee Health Science Center, Memphis, Tennessee
Martha Wells, DDS
Associate Professor and Director of PG Pediatrics, College of Dentistry, University of Tennessee Health Science Center, Memphis, Tennessee
Wainscott Hollis, DDS
Associate Professor, College of Dentistry, University of Tennessee Health Science Center, Memphis, Tennessee
Robert Brandt, DDS
Adjunct Professor, College of Dentistry, University of Tennessee Health Science Center, Memphis, Tennessee
Stefan A. Hottel
College of Biology, College of Dentistry, University of Tennessee Health Science Center, Memphis, Tennessee
William L. Balanoff, DDS
Chief Medical Officer, TicTocStop Inc, New York, New York
References
1. What is Tourette Syndrome? Bayside, NY: Tourette Association of America; https://tourette.org/media/WhatisEnglish.proof_.r1.pdf. Accessed November 30, 2018.
2. Leckman JF, Riddle MA, Hardin MT, et al. The Yale Global Tic Severity Scale: initial testing of a clinician-rated scale of tic severity. J Am Acad Child Adolesc Psychiatry. 1989;28(4)566-573.
3. Storch EA, Murphy TK, Geffken, GR, et al. Reliability and validity of the Yale Global Tic Severity Scale. Psychol Assess. 2005;17(4):486-491.
4. Piacentini J, Woods DW, Scahill L, et al. Behavior therapy for children with Tourette disorder: a randomized controlled trial. JAMA. 2010;303(19):1929-1937.
5. Servello D, Porta M, Sassi M, et al. Deep brain stimulation in 18 patients with severe Gilles de la Tourette syndrome refractory to treatment: the surgery and stimulation. J Neurol Neurosurg Psychiatry. 2008;79(2):136-142.
6. Sutcher HD, Underwood RB, Beatty RA, Sugar O. Orofacial dyskinesia. A dental dimension. JAMA. 1971;216(9):1459-1463.
7. Keller JH, Moffett BC Jr. Nerve endings in the temporomandibular joint of the Rhesus Macaque. Anat Rec. 1968;160(3):587-594.