Treatment of Peri-implantitis Using a Combined Decontaminative and Regenerative Protocol: Case Report
Nicola Alberto Valente, DDS, MS; and Sebastiano Andreana, DDS, MS
Abstract: Peri-implantitis is a long-term complication that frequently occurs after implant surgery. Several treatment methods have been proposed that are aimed at arresting the disease and potentially regenerating bone defects related to it. The use of a combination of proposed decontaminative treatments could possibly improve outcomes. In this case report the authors describe a combined mixed protocol, in which chemical decontamination and a laser-assisted decontamination technique are used in a surgical regenerative approach to successfully manage and treat a case of peri-implantitis. The combined surgical and antimicrobial protocol described can be a suitable treatment option in cases in which implants show severe bone resorption as a result of peri-implant disease.
The prevention and treatment of peri-implant disease has become a primary topic of interest in the dental literature and has gained the attention of various major international dental associations. Two consecutive consensus conferences were organized by the European Federation of Periodontology in 2008 and 2011, which helped define the disease, assess prevalence, and determine risk factors associated with peri-implantitis.1,2 The third European Association for Osseointegration Consensus Conference in 2012 outlined the most current data regarding prevalence, role of occlusal overload, and outcomes of surgical and nonsurgical treatment for peri-implantitis.3 In 2013, the American Academy of Periodontology released an official statement regarding diagnosis and clinical implications of peri-implantitis.4
All of these aforementioned reports agree on the importance of detection of bleeding on probing (BoP) and suppuration as well as progressive radiographical bone loss for the diagnosis of peri-implant disease. However, as of yet there is no common agreement on the diagnostic parameters.5
The onset of peri-implant disease is a result of an imbalance between bacterial load and the host defense.2 Probing and radiographic assessment are the primary diagnostic means. Probing should be performed using a force of 0.25 N without damaging the peri-implant tissues, with the intention being to assess the presence of BoP or suppuration.6 BoP indicates the presence of inflammation in the peri-implant mucosa and is a sign of tissue loss and that future support of the dental implant will be needed. Probing depth (PD) should be assessed regularly at every maintenance visit for the detection of BoP in order to determine any increase in pocket depth over time. Radiographs also should be taken regularly—at least once per year—to assess bone loss.
Despite the large quantity of studies available, a lack of consensus still remains regarding protocols of treatment.7 Most authors agree that two fundamental aspects must be controlled: inflammation and degranulation. However, the devices and instruments used to obtain control of these factors are extremely varied and remain objects of debate. Interestingly, most of the studies available involve the use of a laser, with its usage having exponentially increased in recent years.8 The diode laser, in particular, has demonstrated encouraging results in terms of titanium surface decontamination in in vitro studies, reosseointegration in animal studies, and clinical parameters in clinical trials.9-14
This case report describes a successfully treated case of peri-implantitis using a protocol that involves debridement, decontamination, and guided bone regeneration.
Case Description
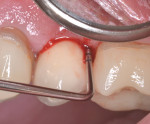
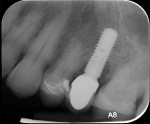
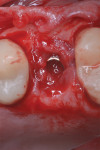
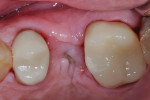
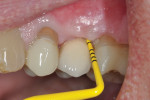
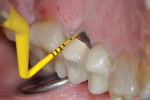
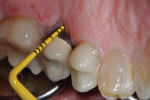
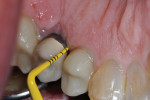
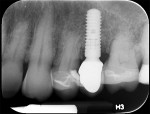
A healthy 58-year-old Caucasian male patient presented with extensive bone resorption, BoP, and suppuration affecting an implant placed in the maxillary left second premolar area (Figure 1). Probing depths were 6 mm on the mesial-buccal aspect, 6 mm on the mid-buccal aspect, 9 mm on the distal-buccal aspect, 5 mm on the mesial-palatal aspect, 3 mm on the mid-palatal aspect, and 4 mm on the distal-palatal aspect. Radiographic assessment revealed bone resorption of about 25% of the total length of the fixture on the mesial aspect and about 40% of the total length of the fixture on the distal aspect (Figure 2). Based on Froum and Rosen's classification of peri-implant disease,15 the patient was diagnosed with moderate peri-implantitis.
During the first visit 1-mg minocycline hydrogen chloride microspheres were locally delivered in the pocket surrounding the implant. After 1 month the peri-implant mucosa was no longer showing signs of BoP or suppuration, though the PD remained the same. The patient was then scheduled for surgical peri-implant debridement and bone grafting.
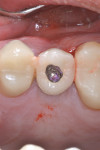
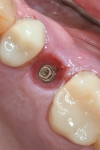
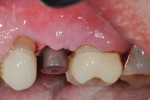
Prior to surgery a hole was drilled through the occlusal aspect of the prosthetic crown to enable access to the abutment screw in an attempt to preserve the suprastructure for future reuse while allowing for a full post-surgical closure and buried healing (Figure 3). The crown was then unscrewed and a cover screw was applied on the implant (Figure 4).
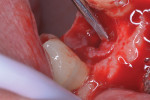
On the day of surgery the area was anesthetized via local infiltration of mepivacaine 2% 1:50,000. A crestal incision was performed extending mesially and distally to the affected implant with a sulcular incision around the adjacent teeth. Reflection of the flap revealed a circumferential defect around the fixture occupied by granulation tissue (Figure 5). The bone resorption did not involve the buccal or palatal walls or the alveolar bone of the adjacent teeth. A thorough degranulation was performed with a metal curette and a rotating, round diamond bur mounted on a high-speed handpiece to ensure complete removal of inflammatory tissue residuals from the bony walls (Figure 6).
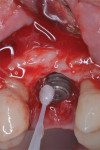
The implant surface was then decontaminated using a combined protocol in which the surface was exposed to a solution of 100-mg doxycycline hyclate powder and sterile water for 60 seconds, then rinsed thoroughly, followed by hydrogen peroxide 10% volume applied with a microbrush for 60 seconds, and rinsed again (Figure 7). Finally, a 810-nm diode laser set at 1 W in continuous mode non-initiated was used to complete the decontamination, with the fiber-optic tip being passed over the titanium surface 3 mm away from the surface in an up-and-down motion around the circumference of the fixture for 60 seconds.
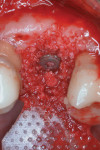
The peri-implant defect was then grafted with 1-cc cortico/cancellous particulate allograft. A 15-mm x 20-mm polylactic acid bioresorbable membrane was trimmed and adapted over the area, and the flap was sutured with 4-0 polytetrafluoroethylene (PTFE) in an attempt to obtain a primary closure (Figure 8).
Systemic antibiotics were prescribed consisting of 100-mg doxycycline hyclate twice per day for a total of 10 days starting the day before surgery. The patient was instructed to gently dab the surgical area with a local application of chlorhexidine gluconate 0.12% twice per day for 14 days. Suture removal was performed 14 days after surgery.
The case was monitored for 8 months with regular monthly oral hygiene recall and maintenance visits; no adverse events were noted. The membrane was slightly exposed during the first phases of healing with no harmful effects on the healing itself and full epithelization of the exposed area. The mucosal healing left the cover screw slightly uncovered, and a healing abutment was placed 4 months after surgery so food and plaque accumulation would be avoided and to facilitate the patient's independent maintenance of proper hygiene (Figure 9 and Figure 10).
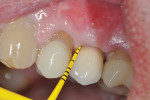
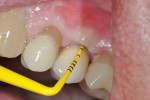
Eight months after the surgery the soft tissues showed no signs of inflammation or bleeding, and the probing depths around the healing abutment were 5 mm on the mesial-buccal aspect, 5 mm on the mid-buccal aspect, 5 mm on the distal-buccal aspect, 4 mm on the mesial-palatal aspect, 4 mm on the mid-palatal aspect, and 5 mm on the distal-palatal aspect (Figure 11 through Figure 16). Given the stability of the clinical condition it was decided to redeliver the crown. PD values were improved, inflammatory signs were stable post-surgery, and signs of regeneration were evident radiographically (Figure 17).
Discussion
The present case report documents a case of peri-implantitis treated by decontamination with a combined protocol and grafting of the defect. Although, as stated by Mombelli et al in a recent narrative review,16 the evidence available from the literature does not determine a specific protocol of treatment or recommendations for the surgical therapy of peri-implantitis, some elements of therapy seem to be commonly used and accepted as beneficial. One such treatment involves surgical access by a full-thickness flap, thorough degranulation followed by decontamination and cleaning of the implant surfaces, and stabilization of the defect with a bone substitute, combined with systemic antibiotics and oral chlorhexidine applications during the healing phase.
The use of doxycycline for treatment of peri-implantitis has shown good results in a randomized controlled trial in which, after local delivery of doxycycline following subgingival scaling of peri-implant lesions with a plastic curette, the sites treated demonstrated significant gain in probing attachment level, BoP, and PD at 18 weeks.17 The study was followed by a case report from the same group of authors, in which doxycycline was used to decontaminate the implant surface in the context of a surgical treatment with degranulation and autologous bone graft with good results in terms of inflammation, BoP, and PD.18 Muller et al reported a case followed up for 8 years using a protocol similar to that reported here.19
The use of hydrogen peroxide also has shown good results when utilized for implant surface decontamination. In a 5-year follow-up study, hydrogen peroxide chemical compound used after surgical exposure and degranulation was successful in 58% of 26 implants affected by peri-implantitis.20 In a different study, good results in terms of PD and bone gain also were reported after the use of hydrogen peroxide in combination with bone grafting at 1-year and 5-year follow-up visits.21,22 Interestingly, in an in vitro study the application of hydrogen peroxide on titanium discs seeded with human epithelial cells showed an increase in cell proliferation compared to the application of chlorhexidine gluconate.23
Use of a 810-nm diode laser has demonstrated encouraging results in terms of bacterial decontamination in in vitro studies, with marked reduction of bacterial colonization and without causing alterations on the titanium implant surface after irradiation.10,24,25 In a 5-year follow-up clinical study, the use of a 810-nm diode laser at 1 W in continuous mode for decontamination after surgical exposure of implants affected by peri-implantitis showed significant reduction of gram-negative anaerobic rods, with higher implant survival rates compared to the conventionally treated group and without harmful effects on soft and hard tissues.26
The scientific evidence for the use of a membrane is thus far contradictory. A review by Faggion et al indicated that the use of nonresorbable membranes leads to new bone gain,27 however Roos-Jansåker et al published opposite results that showed the noncritical effect of placing the membrane over the bone graft.22 It is the present authors' opinion that the use of a resorbable membrane favors peri-implant bone regeneration.
In the present case, it is unknown which factor was the most significant in halting the progression of peri-implant inflammation and bone loss, thus enabling the gain of new, radiographically detectable peri-implant bone; however, the speed of healing was remarkable, making this approach well-suited for the treatment of peri-implant inflammatory disease.
Conclusion
The combined surgical and antimicrobial protocol described here, which encompasses the use of open flap debridement, doxycycline, hydrogen peroxide and laser decontamination, and guided bone regeneration, can be a viable and recommendable treatment option in cases of implants showing severe bone resorption as a result of peri-implant disease.
Acknowledgment
The authors would like to thank Catherine Cagino, DDS, for her contribution in the prosthetic phase of the treatment of this case.
About the Authors
Nicola Alberto Valente, DDS, MS
Assistant Professor, Head of Clinical Activities, Unit of Oral Surgery and Implantology, University of Geneva and Geneva University Hospitals, Geneva, Switzerland; Adjunct Assistant Professor, Department of Periodontics and Endodontics, State University of New York at Buffalo, Buffalo, New York
Sebastiano Andreana, DDS, MS
Associate Professor, Director of Implant Dentistry, Department of Restorative Dentistry, State University of New York at Buffalo, Buffalo, New York
References
1. Lindhe J, Meyle J; Group D of European Workshop on Periodontology. Peri-implant diseases: Consensus Report of the Sixth European Workshop on Periodontology. J Clin Periodontol. 2008;35(suppl 8):282-285.
2. Lang NP, Berglundh T; Working Group 4 of Seventh European Workshop on Periodontology. Periimplant diseases: where are we now?—Consensus of the Seventh European Workshop on Periodontology. J Clin Periodontol. 2011;38(suppl 11):178-181.
3. Klinge B, Meyle J; Working Group 2. Peri-implant tissue destruction. The Third EAO Consensus Conference 2012. Clin Oral Implants Res. 2012;23(suppl 6):108-110.
4. Rosen P, Clem D, Cochran D, et al. Peri-implant mucositis and peri-implantitis: a current understanding of their diagnoses and clinical implications. J Periodontol. 2013;84(4):436-443.
5. Valente NA, Andreana S. Peri-implant disease: what we know and what we need to know. J Periodontal Implant Sci. 2016;46(3):136-151.
6. Heitz-Mayfield LJ. Peri-implant diseases: diagnosis and risk indicators. J Clin Periodontol. 2008;35(suppl 8):292-304.
7. Figuero E, Graziani F, Sanz I, et al. Management of peri-implant mucositis and peri-implantitis. Periodontol 2000. 2014;66(1):255-273.
8. Natto ZS, Aladmawy M, Levi PA Jr, Wang HL. Comparison of the efficacy of different types of lasers for the treatment of peri-implantitis: a systematic review. Int J Oral Maxillofac Implants. 2015;30(2):338-345.
9. Kreisler M, Kohnen W, Marinello C, et al. Antimicrobial efficacy of semiconductor laser irradiation on implant surfaces. Int J Oral Maxillofac Implants. 2003;18(5):706-711.
10. Sennhenn-Kirchner S, Klaue S, Wolff N, et al. Decontamination of rough titanium surfaces with diode lasers: microbiological findings on in vivo grown biofilms. Clin Oral Implants Res. 2007;18(1):126-132.
11. Shibli JA, Martins MC, Ribeiro FS, et al. Lethal photosensitization and guided bone regeneration in treatment of peri-implantitis: an experimental study in dogs. Clin Oral Implants Res. 2006;17(3):273-281.
12. Salmeron S, Rezende ML, Consolaro A, et al. Laser therapy as an effective method for implant surface decontamination: a histomorphometric study in rats. J Periodontol. 2013;84(5):641-649.
13. Dortbudak O, Haas R, Bernhart T, Mailath-Pokorny G. Lethal photosensitization for decontamination of implant surfaces in the treatment of peri-implantitis. Clin Oral Implants Res. 2001;12(2):104-108.
14. Papadopoulos CA, Vouros I, Menexes G, Konstantinidis A. The utilization of a diode laser in the surgical treatment of peri-implantitis. A randomized clinical trial. Clin Oral Investig. 2015;19(8):1851-1860.
15. Froum SJ, Rosen PS. A proposed classification for peri-implantitis. Int J Periodontics Restorative Dent. 2012;32(5):533-540.
16. Mombelli A, Moene R, Decaillet F. Surgical treatments of peri-implantitis. Eur J Oral Implantol. 2012;5(suppl):S61-S70.
17. Buchter A, Meyer U, Kruse-Losler B, et al. Sustained release of doxycycline for the treatment of peri-implantitis: randomised controlled trial. Br J Oral Maxillofac Surg. 2004;42(5):439-444.
18. Buchter A, Kleinheinz J, Meyer U, Joos U. Treatment of severe peri-implant bone loss using autogenous bone and a bioabsorbable polymer that delivered doxycycline (Atridox). Br J Oral Maxillofac Surg. 2004;42(5):454-456.
19. Muller E, Gonzalez YM, Andreana S. Treatment of peri-implantitis: longitudinal clinical and microbiological findings—a case report. Implant Dent. 1999;8(3):247-254.
20. Leonhardt A, Dahlen G, Renvert S. Five-year clinical, microbiological, and radiological outcome following treatment of peri-implantitis in man. J Periodontol. 2003;74(10):1415-1422.
21. Roos-Jansåker AM, Renvert H, Lindahl C, Renvert S. Surgical treatment of peri-implantitis using a bone substitute with or without a resorbable membrane: a prospective cohort study. J Clin Periodontol. 2007;34(7):625-632.
22. Roos-Jansåker AM, Persson GR, Lindahl C, Renvert S. Surgical treatment of peri-implantitis using a bone substitute with or without a resorbable membrane: a 5-year follow-up. J Clin Periodontol. 2014;41(11):1108-1114.
23. Ungvari K, Pelsoczi IK, Kormos B, et al. Effects on titanium implant surfaces of chemical agents used for the treatment of peri-implantitis. J Biomed Mater Res B Appl Biomater. 2010;94(1):222-229.
24. Tosun E, Tasar F, Strauss R, et al. Comparative evaluation of antimicrobial effects of Er:YAG, diode, and CO(2) lasers on titanium discs: an experimental study. J Oral Maxillofac Surg. 2012;70(5):1064-1069.
25. Stubinger S, Etter C, Miskiewicz M, et al. Surface alterations of polished and sandblasted and acid-etched titanium implants after Er:YAG, carbon dioxide, and diode laser irradiation. Int J Oral Maxillofac Implants. 2010;25(1):104-111.
26. Bach G, Neckel C, Mall C, Krekeler G. Conventional versus laser-assisted therapy of periimplantitis: a five-year comparative study. Implant Dent. 2000;9(3):247-251.
27. Faggion CM, Chambrone L, Listl S, Tu Y-K. Network meta-analysis for evaluating interventions in implant dentistry: the case of peri-implantitis treatment. Clin Implant Dent Relat Res. 2013;15(4):576-588.