Soft-Tissue Grafting Techniques Associated With Immediate Implant Placement
Mark Bishara, DDS; Gregori M. Kurtzman, DDS; Waji Khan, DDS; Joseph Choukroun, MD; and Richard J. Miron, DDS, dr. med dent, MSc, PhD
ABSTRACT
Immediate implant placement often presents challenges in terms of predictably obtaining soft-tissue coverage over the implant site. While delayed implant placement offers the ability for soft tissues to grow and invade the extraction socket making their attachment around implants more predictable, immediate implant placement poses a significant risk of bacterial invasion towards the implant surface as a result of insignificant soft-tissue volume. Soft-tissue grafting techniques have often been proposed for use during immediate implant placement to augment soft-tissue deficiencies, including the use of either palatal connective tissue grafts (CTGs) or collagen-derived scaffolds. However, both of these approaches have significant drawbacks in that CTGs are harvested with high patient morbidity and collagen scaffolds remain avascular and acelluar posing a risk of infection/implant contamination. More recently, platelet-rich fibrin (PRF) has been proposed as an economical and biological means to speed soft-tissue wound healing. In combination with immediate implant placement, PRF offers an easily procurable low-cost regenerative modality that offers an efficient way to improve soft-tissue attachment around implants. Furthermore, the supra-physiological concentration of defense-fighting leukocytes in PRF, combined with a dense fibrin meshwork, is known to prevent early bacterial contamination of implant surfaces, and the biological concentrations of autologous growth factors in PRF is known to increase tissue regeneration. This article discusses soft-tissue grafting techniques associated with immediate implant placement, presents several cases demonstrating the use of PRF in routine immediate implant placement, and further discusses the biological and economic advantages of PRF for the management of soft-tissue grafting during immediate implant placement.
Implant placement often presents challenges in terms of predictable soft-tissue coverage. This is especially true in the esthetic zone where a lack of supporting keratinized soft tissue during implant placement often compromises the final esthetic outcomes when mucosal recession occurs around these implants. Timing of implant placement has mainly been characterized into four categories: (1) immediate implant placement (type 1) occurring when implants are placed at the same time as the surgical extraction of teeth; (2) early implant placement (type 2) occurring when soft-tissue healing has transpired and implants are placed typically within 4 to 8 weeks following extraction; (3) early implant placement (type 3) when partial bone healing has occurred in the socket by typically 12 to 16 weeks after extraction; and lastly, (4) late implant placement (type 4) when the extraction socket has fully healed after 16 weeks.1 The principles that will be discussed in this article may be applied with either a one-stage or two-stage surgical approach, depending on the practitioner’s preferences and the clinical situation that presents.
Type 1 immediate implant placement is deemed the most surgically challenging and possesses additional risks in that soft-tissue coverage over the extraction site as primary closure is not easily achievable without flap elevation and repositioning. Unlike types 2 through 4, where soft tissue has formed over the socket, the practitioner is left with limited methods to protect the implant site during the initial healing period. Traditionally, soft tissue is relieved (often requiring vertical releasing incisions) at the time of implant placement by undermining the periosteum, thus allowing tissue mobilization without tension to achieve a passive primary closure over the wound site.2 This often results in distortion of the mucogingival junction and a lack of adequate attached gingiva on the buccal/facial aspect of the site. Furthermore, an apical repositioning of the flap at second-stage surgery (implant exposure) and/or soft-tissue grafting is often required to allow for sufficient keratinized tissue coverage around the implant emergence. An associated increase in swelling with associated pain while utilizing this approach due to manipulation of the periosteum related to undermining to permit flap release has also been noted.
Other techniques for wound coverage allow the site to heal by secondary intention and use barrier membranes to protect the implant site and any associated bone graft that may have been placed simultaneously during implant placement. Such techniques include the use of d-polytetrafluoroethylene (d-PTFE) membranes, which require removal of the nonresorbable membrane at a later date. Expanded polytetrafluoroethylene (e-PTFE) membranes have also been widely used for guided bone regeneration (GBR) procedures in implant dentistry since the early 1980s, and their long-term use has been well-documented.3,4 Several disadvantages of e-PTFE include wound dehiscence and early membrane exposure that may compromise the bone augmentation procedure and lead to localized infection.5 d-PTFE can also be used in an open membrane technique (part of the membrane is left exposed due to an inability to achieve primary closure) as introduced in 2005 by Funakoshi.6 The advantage of this technique over the use of e-PTFE is d-PTFE has a very low membrane porosity and, thus, prevents bacterial penetration through the membrane to the underlying graft or implant.7 The smooth nonporous surface of this membrane allows portions to be left exposed while protecting the underlying graft and implant during the initial healing period. Because this approach does not require periosteal releasing incisions, postoperative pain and swelling are minimized. The margins of the socket are undermined and the membrane is tucked under the soft tissue on the buccal and lingual, then sutures are placed to retain the membrane (Figure 1). These membranes are typically removed without local anesthesia at 4 to 6 weeks by grasping the exposed portion and gently tugging it out of the site, as tissue does not grow into the membrane. New gingival tissue grows under the membrane and completely covers the site. While this method is often utilized, it is expected that soft-tissue healing is slightly slower and the nonresorbable membranes are known to create a foreign body reaction with host tissues.8
While nonresorbable barrier membranes were first utilized, an entire line of resorbable membranes fabricated from collagen derived from various allograft and xenograft sources have seen a steady increase in use due to their added biocompatibility and the elimination of the need for a second procedure to remove the membrane.9 Therefore, collagen-derived membranes were introduced as an alternative either by acting as a barrier or by being utilized as a socket seal as a collagen plug to protect the wound site.10,11 This technique reduces postoperative discomfort because there is no need for primary closure or a second harvest site. Naturally, the gold standard for soft-tissue coverage of either a graft or implant site is autologous tissue, including a free gingival graft or connective tissue graft.12 While clinically the final esthetic outcomes remain optimal, use of these grafts is also associated with a higher incidence of postoperative discomfort due to graft harvesting at the donor site.10
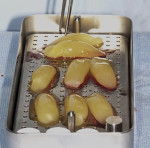
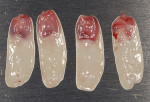
Deriving biological membranes from blood drawn from the patient’s peripheral veins has recently been proposed as an alternative approach to packaged membranes. Briefly, blood is collected and immediately centrifuged for 8 to 14 minutes to reach supra-physiological doses of growth factors and leukocytes producing what has been termed platelet-rich fibrin (PRF) (Figure 2). PRF can similarly be used in an open membrane technique to protect the implant site and may be left exposed to the oral cavity without major risk of infection due to its incorporation of supra-physiological concentration of leukocytes, defense-fighting cells that engulf incoming pathogens. Furthermore, the dense fibrin meshwork also prevents bacteria from penetrating toward the implant surface. Once centrifuged the fibrin clot portion in the tube (PRF) is removed (Figure 3) and pressed to create an autologous membrane derived without the use of anticoagulants (Figure 4). The advantages of this approach include reduced cost and complete biocompatibility with host tissues, thus avoiding the use of creating a foreign body reaction. Additionally, remodeling of the PRF membrane naturally occurs within 2 weeks and acts as a “tissue glue” when placed, aiding in containing the graft placed into the site and protecting it from saliva and its bacterial components (Figure 5). The PRF membrane may also be used in a “poncho” technique, retaining the membrane with the healing cap or cover screw to provide wound protection and stabilization of soft tissues while further speeding the regenerative outcomes of soft tissues as a result of the supra-physiological doses of growth factors found in PRF.
The following case reports illustrate the typical steps undertaken to obtain advanced PRF (A-PRF) membranes used to cover an immediate implant placement.
Case 1
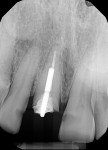
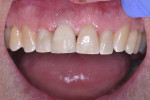
A 44-year-old healthy man presented following a work-related incident in which his endodontically treated maxillary right central incisor, which had been restored with a post/core and crown, was fractured. The tooth fracture occurred at the gingival margin (Figure 6). Replacing the crown would require osseous crown lengthening to achieve a restorative ferrule, thereby compromising the bone level on the adjacent teeth. Clinically, no evidence of root fracture was noted (Figure 7) and probing depths were within normal levels (1 mm to 3 mm). Treatment options presented to the patient included: osseous crown lengthening with placement of a new post/core and crown, extraction with placement of a fixed bridge using the adjacent teeth as abutments, or extraction and implant placement with a single-crown restoration. The patient chose the implant treatment option.
A thorough medical history was collected and extraction consent given. Two carpules of 1.8 ml 2% Xylocaine Dental with 1:100,000 epinephrine (Dentsply Sirona, dentsplysirona.com) was administered as infiltration locally and an incisive nerve block was achieved. A disposable tourniquet was placed on the patient’s right arm, 10 centimeters above the elbow. A vein was visually and physically located in the antecubital fossa. An alcohol wipe was used to prepare the site to eliminate potential of dermal bacteria at the needle site to collect blood, and the site was allowed to dry for 30 seconds. A 23-gauge venipuncture needle was attached to a vacutainer holder and two 10-ml red vacutainer tubes were used to harvest two vials of blood. This was then immediately centrifuged at 1,300 rpm for 8 minutes as per the A-PRF protocol.4,5
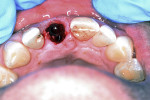
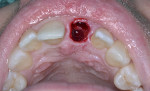
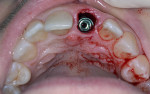
With the use of a Woodson elevator (Hu-Friedy, hu-friedy.com), the gingiva was delicately dissected from the fractured central incisor; this was done in a circumferential manner to expand the bone around the tooth. The tooth was atraumatically extracted using a #76S forcep (Hu-Friedy) employing a rotation/counter-rotation technique to prevent fracture of the thin buccal plate. The extraction socket was examined; the buccal plate was noted to be intact and no fenestration or dehiscence was observed (Figure 8). The extraction socket was debrided using a curette to remove any residual soft tissue and then irrigated with saline solution. A precision drill (MIS Implants, mis-implants.com) was then used to engage the dense palatal cortical bone for the pilot osteotomy to act as a guide for the subsequent osteotomy drills. This was followed with use of the 2.3-mm pilot drill (MIS Implants), placing the implant angulation to accommodate a screw-retained position for the planned crown. The drilling sequence, thus, was completed.
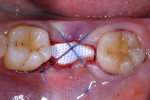
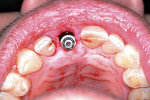
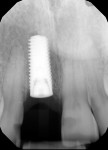
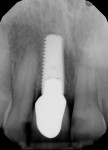
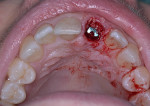
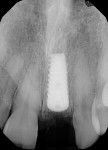
A UNC 15 periodontal probe (Hu-Friedy) was then used to assess the osteotomy site to ensure no fenestrations in the buccal plate had occurred following osteotomy preparation. A 5-mm x 13-mm C1 implant (MIS Implants) was placed into the preparation (Figure 9). Implant primary stability was evidenced by a 30 Ncm insertion torque. Lateral stability was questionable based on the gap/jump junction observed between the buccal plate and the implant. A cover screw was placed into the implant. The fibrin clots were removed from the glass vials to produce A-PRF membranes, and one of these membranes was then placed to augment the gap on the buccal and cover the extraction site (Figure 10). A figure 8 suture wasangiotech.com placed over the site with 3-0 silk on a reverse cutting C6 needle (LOOK™ Sutures, Surgical Specialties Corp., angiotech.com). A periapical radiograph was taken to document the implant placement (Figure 11).
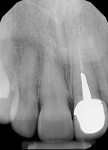
The patient returned a week later for postoperative evaluation and suture removal and reported very minimal to no pain following the procedure. At 3 months post implant placement, the patient returned for stage two surgery and a periapical radiograph was taken (Figure 12). Half a carpule of 2% Xylocaine Dental with 1:100,000 epinephrine was administered into the gingiva overlaying the implant. An incision was made with a 15c scalpel blade mesial-distally to the palatal side of the mid-crestal line. The tissue was positioned to the facial to preserve adequate attached gingiva and horizontal mattress sutures with 6-0 Prolene® (Ethicon, ethicon.com) were used to secure the repositioned flap. A screw-retained provisional crown was created to develop the emergence profile of the soft tissue around the uncovered implant.
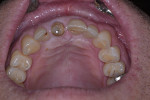
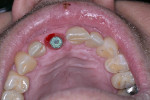
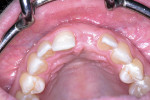
Approximately 6 weeks later, the patient presented for impressions and the screw-retained provisional crown was removed (Figure 13). An open-tray impression coping was placed and a periapical radiograph was taken to ensure the impression coping was properly seated. An open-tray fixture level impression was taken using Maxill® Light and Heavy Body PVS (Maxill Dental, maxill.com) in a stock tray (Master Tray®, Waterpik Oral Health, waterpik.com) and was sent to the lab for fabrication of a screw-retained crown.
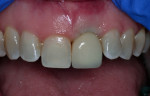
The laboratory returned the restoration for insertion. The patient presented and the provisional restoration was removed. The final screw-retained crown was inserted and the screw was tightened with finger pressure on a hex wrench. A radiograph was taken to verify complete mating of the parts. A torque wrench was used to tighten the fixation screw to the manufacturer’s recommendation of 30 Ncm. A ball of PTFE tape was placed into the screw channel and sealed with Filtek™ Flow composite (3M ESPE, 3m.com). Occlusion was checked and the patient dismissed (Figure 14). Follow-up with the patient at a post-insertion appointment demonstrated healthy non-inflamed gingival tissue surrounding the implant restoration with a natural emergence profile (Figure 15).
Case 2
A 38-year-old healthy woman presented to the clinic with issues pertaining to a previously endodontically treated and restored maxillary left central incisor. Radiographically the tooth appeared normal (Figure 16). Clinically, a vertical root fracture was noted as evidenced by an isolated probing depth on the facial of the tooth. Treatment options were presented to the patient, which included: extraction with placement of a fixed bridge using the adjacent teeth as abutments, or extraction and implant placement with a single-crown restoration. She decided to pursue the dental implant option.
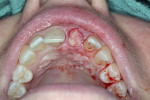
After a thorough medical history was collected, consent for treatment was provided. Two carpules of 1.8-ml 2% Xylocaine Dental with 1:100,000 epinephrine was administered as infiltration and an incisive nerve block. Blood was collected at the antecubital fossa as previously described in Case 1. A Woodson elevator was used to circumferentially dissect the gingiva around the central incisor. The tooth was atraumatically extracted using a #76S forcep in a rotation/counter-rotation manner (Figure 17). The extraction socket was then debrided using a curette and irrigated with saline solution. A precision drill was then used to engage the dense palatal cortical bone to guide the pilot drill for the osteotomy. The 2.3-mm pilot drill was used to align the angulation to allow a screw-retained crown implant position. The drilling sequence was then completed.
A UNC 15 periodontal probe was used to assess the osteotomy site to ensure no fenestrations in the buccal plate were present. An MIS SEVEN 5-mm x 13-mm implant (MIS Implants) was placed into the osteotomy with primary stability as evidenced by a 30-Ncm insertion torque (Figure 18). Again, the lateral stability was questionable as a result of the gap/jump junction that was observed between the buccal plate and implant. A cover screw was placed into the implant.
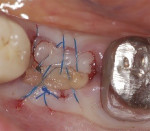
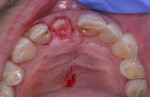
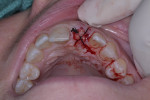
Because this case had a larger gap/jump junction then the previous case discussed, 0.25 cc cortical/cancellous bone allograft with a particle size ranging from 250 µm to 1000 µm (OraGRAFT®, LifeNet Heath, lifenethealth.org) was mixed with an injectable PRF (i-PRF, centrifuged at 700 rpm for 3 minutes) to create a putty-like graft, which was placed into the buccal gap (Figure 19). The fibrin clots were removed from the centrifuged vials to produce A-PRF membranes as previously described in Case 1, and an A-PRF membrane was then used to cover the extraction site (Figure 20). This was fixed in place using a figure 8 suture utilizing 3-0 silk on a reverse cutting C6 needle (Figure 21).
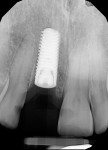
The patient returned a week later for postoperative evaluation and suture removal. Three months post implant placement the patient returned for stage two surgery demonstrating healthy non-inflamed soft tissue overlaying the implant (Figure 22). A periapical radiograph was taken to assess implant healing (Figure 23). Half a carpule of 2% Xylocaine Dental with 1:100,000 epinephrine was administered locally and the implant cover screw was exposed. An open-tray impression coping was placed and a periapical radiograph was taken to verify that the impression coping was fully seated. An open-tray fixture level impression was taken and sent to the lab for fabrication of a screw-retained crown. A healing abutment was placed and the patient was dismissed after modification of the removable provisional prosthesis. The crown was returned from the lab and inserted onto the implant (Figure 24).
Discussion
The present case reports highlight the use of autologous blood concentrates (ie, PRF) for everyday dental use. While PRF has gained much attention as a regenerative agent capable of further speeding tissue regeneration across many fields of medicine, the authors focused the present article on its use in immediate implant dentistry. Currently, the trend in implant placement has slowly shifted from a delayed approach toward immediate/early placement as patients continue to seek more rapid treatment protocols and final restorations. As a result, immediate implant placement has received considerable attention in recent years.
One of the main limitations of immediate implant placements is that, unlike with early implant placement, soft tissue has not fully matured over the extraction socket. This makes immediate implant placement somewhat more biologically demanding as it is now known that poor integration of soft tissues to implants drastically increases the risk of peri-implantitis and decreases long-term implant survival.13-16 While hard-tissue integration into bone was the primary focus of the majority of research in the 1990s and early 2000s, over the past decade more research has been done on soft-tissue integration around dental implants as a primary requirement for their long-term success. Simply put, to maintain long-term integration of dental implants, both soft and hard tissues must adequately be preserved. Especially critical to implant dentistry is soft-tissue attachment to the coronal portion of the implant. Should this criterion fail to be fulfilled during implant placement, the implant is at greater risk for bacterial contamination whereby incoming pathogens are able to attach to the roughened portion of the implant posing a major risk of peri-implantitis, a disease that is difficult to resolve once progression begins.
For these reasons, soft-tissue management around immediate implant placement is critically important. While historically, most cases with deficient soft-tissue volume were regenerated with either a connective tissue graft (CTG) or a collagen-based scaffold, major drawbacks were also noted. CTG harvesting is associated with high morbidity to the patient in the palate, and acellular collagen-based scaffolds pose a risk of infection if left exposed to the oral cavity because they are avascular. More recently, PRF has been utilized as a completely natural way of improving soft-tissue wound closure and delivering localized growth factors to the microenvironment.17 PRF poses several advantages when compared to other modalities: (1) It contains a host of leukocytes entrapped within its fibrin matrix. This serves the important role of preventing bacterial contamination.18 (2) PRF has also been shown to deliver supra-physiological doses of important growth factors for regeneration, including platelet-derived growth factor (PDGF), TGF-beta, vascular endothelial growth factor (VEGF), and insulin-like growth factor (IGF), which support cell growth and new blood vessel revascularization.19-22 (3) PRF also supports faster soft-tissue wound healing when compared to hard tissues.17 (4) Recently, it was shown that PRF specifically is able to support better soft-tissue attachment of gingival fibroblast to implant surfaces.22 This favors the quick colonization of host-regenerative cells toward the implant surface as opposed to bacterial pathogens. (5) Lastly, it has also been reported that PRF supports less morbidity to patients whereby they take less medications and report less pain as a result of faster soft-tissue defect closure.23,24
The present report also utilized the recent modifications to PRF centrifugation speeds highlighted by the low-speed centrifugation concept. While the first protocols of PRF designed by Dr. Joseph Choukroun used high g-force with a 2700 RPM for 12 minutes,25 more recently it has been proposed that lower centrifugation speeds and time are favored to produce higher platelet, leukocyte, and growth factor concentrations.20,21,25,26 Therefore, recent modifications to centrifugation speeds and times (1300 RPM, 200 g-force for 8 minutes) have been proposed, leading to higher levels of growth factor release, cellular collagen synthesis, and more synergistic cellular activity when compared to the original leukocyte- and platelet-rich fibrin (L-PRF) formulations.20 Furthermore, even shorter and slower centrifugation speeds (700 RPM, 60 g-force for 3 minutes) have further been shown to result in i-PRF, a liquid version of PRF that further enhances leukocyte numbers and growth factor release that may be used in combination with bone grafting materials as demonstrated.5,26-29
Conclusion
The use of A-PRF as a fully biocompatible patient-generated biological membrane lowers the cost of rendering treatment while simultaneously favoring faster soft-tissue wound healing around immediate implant placement. Additionally, PRF is well-tolerated by the patient, leading to better healing with fewer postoperative complications as a result of its incorporation of defense-fighting leukocytes when compared to routine traditional membrane use. While treatment with PRF is still considered in its infancy, especially around immediate implant placement, many advantages, both biological and economic, are thought to further increase its use in everyday dental practice.
Disclosure
Dr. Joseph Choukroun is the developer of A-PRF/i-PRF and owns distribution rights to the equipment to process the PRF products.
ABOUT THE AUTHORS
Mark Bishara, DDS
Private Practice
Bowmanville, Ontario, Canada
Gregori M. Kurtzman, DDS
Private Practice
Silver Spring, Maryland
Master, Academy of General Dentistry
Diplomate, International Congress of Oral Implantologists
Waji Khan, DDS
Private Practice
Kingston, Ontario, Canada
Joseph Choukroun, MD
Private Practice
Nice, France
Richard J. Miron, DDS, dr. med dent, MSc, PhD Adjunct Faculty
Department of Periodontics
Nova Southeastern University
Fort Lauderdale, Florida
REFERENCES
1. Hämmerle CH, Chen ST, Wilson TG. Consensus statements and recommended clinical procedures regarding the placement of implants in extraction sockets. Int J Oral Maxillofac Implants. 2004;19(suppl):26-28.
2. Greenstein G, Greenstein B, Cavallaro J, et al. Flap advancement: practical techniques to attain tension-free primary closure. J Periodontol. 2009;80(1):4-15.
3. Zhang Y, Zhang X, Shi B, Miron RJ. Membranes for guided tissue and bone regeneration. Annals of Oral & Maxillofacial Surgery. 2013;1(1):10.
4. von Arx T. 5 years of guided bone regeneration (GBR) in implant dentistry. A report on the membrane symposium of 10 and 11 December 1993 at Basel [in German]. Schweiz Monatsschr Zahnmed. 1994;104(4):494-496,515-517.
5. Choukroun J, Ghanaati S. Reduction of relative centrifugation force within injectable platelet-rich-fibrin (PRF) concentrates advances patients' own inflammatory cells, platelets and growth factors: the first introduction to the low speed centrifugation concept. Eur J Trauma Emerg Surg. 2017. doi: 10.1007/s00068-017-0767-9. [Epub ahead of print]
6. Funakoshi E, Yamashita M, Maki K, et al. Guided bone regeneration with open barrier membrane technique. Presented at: 22nd Annual Meeting of the Academy of Osseointegration; 2007.
7. Tal H, Artzi Z, Moses O, et al. Spontaneous early exposure of submerged endosseous implants resulting in crestal bone loss: a clinical evaluation between stage I and stage II surgery. Int J Oral Maxillofac Implants. 2001;16(4):514-521.
8. Tatullo M, Marrelli M, Cassetta M, et al. Platelet Rich Fibrin (P.R.F.) in reconstructive surgery of atrophied maxillary bones: clinical and histological evaluations. Int J Med Sci. 2012;9(10):872-880.
9. Barber HD, Lignelli J, Smith BM, Bartee BK. Using a dense PTFE membrane without primary closure to achieve bone and tissue regeneration. J Oral Maxillofac Surg. 2007;65(4):748-752.
10. Ghanaati S, Booms P, Orlowska A, et al. Advanced platelet-rich fibrin: A new concept for cell-based tissue engineering by means of inflammatory cells. J Oral Implantol. 2014;40(6):679-689.
11. Kotsakis G, Chrepa V, Marcou N, et al. Flapless alveolar ridge preservation utilizing the “socket-plug” technique: clinical technique and review of the literature. J Oral Implantol. 2014;40(6):690-698.
12. Sun Y, Wang CY, Wang ZY, et al. Test in canine extraction site preservations by using mineralized collagen plug with or without membrane. J Biomater Appl. 2016;30(9):1285-1299.
13. Sculean A, Chappuis V, Cosgarea R. Coverage of mucosal recessions at dental implants. Periodontol 2000. 2017;73(1):134-140.
14. Buser D, Chappuis V, Belser UC, Chen S. Implant placement post extraction in esthetic single tooth sites: when immediate, when early, when late? Periodontol 2000. 2017;73(1):84-102.
15. Kinaia BM, Ambrosio F, Resident P, et al. Soft tissue changes around immediately placed implants: a systematic review and meta-analyses with at least 12 months follow up after functional loading. J Periodontol. 2017;88(9):876-886.
16. Poskevicius L, Sidlauskas A, Galindo-Moreno P, Juodzbalys G. Dimensional soft tissue changes following soft tissue grafting in conjunction with implant placement or around present dental implants: A systematic review. Clin Oral Implants Res. 2017;28(1):1-8.
17. Zafiropoulos GG, John G. Use of collagen matrix for augmentation of the peri-implant soft tissue at the time of immediate implant placement. J Contemp Dent Pract. 2017;18(5):386-391.
18. Miron RJ, Fujioka-Kobayashi M, Bishara M, et al. Platelet-rich fibrin and soft tissue wound healing: A systematic review. Tissue Eng Part B Rev. 2017;23(1):83-99.
19. Hoaglin DR, Lines GK. Prevention of localized osteitis in mandibular third-molar sites using platelet-rich fibrin. Int J Dent. 2013;2013:875380.
20. Chen FM, Wu LA, Zhang M, et al. Homing of endogenous stem/progenitor cells for in situ tissue regeneration: Promises, strategies, and translational perspectives. Biomaterials. 2011;32(12):3189-3209.
21. Fujioka-Kobayashi M, Miron RJ, Hernandez M, et al. Optimized platelet-rich fibrin with the low-speed concept: Growth factor release, biocompatibility, and cellular response. J Periodontol. 2017;88(1):112-121.
22. Kobayashi E, Fluckiger L, Fujioka-Kobayashi M, et al. Comparative release of growth factors from prp, prf, and advanced-prf. Clin Oral Investig. 2016;20(9):2353-2360.
23. Wang X, Zhang Y, Choukroun J, et al. Behavior of gingival fibroblasts on titanium implant surfaces in combination with either injectable-PRF or PRP. Int J Mol Sci. 2017;18(2):E331.
24. Bilginaylar K, Uyanik LO. Evaluation of the effects of platelet-rich fibrin and piezosurgery on outcomes after removal of impacted mandibular third molars. Br J Oral Maxillofac Surg. 2016;54(6):629-633.
25. Ustaoglu G, Ercan E, Tunali M. The role of titanium-prepared platelet-rich fibrin in palatal mucosal wound healing and histoconduction. Acta Odontol Scand. 2016;74(7):558-564.
26. Miron RJ, Fujioka-Kobayashi M, Hernandez M, et al. Injectable platelet rich fibrin (i-PRF): Opportunities in regenerative dentistry? Clin Oral Investig. 2017;21(8):2619-2627.
27. Roy S, Driggs J, Elgharably H, et al. Platelet-rich fibrin matrix improves wound angiogenesis via inducing endothelial cell proliferation. Wound Repair Regen. 2011;19(6):753-766.
28. Choukroun J, Adda F, Schoeffler C, Vervelle A. Une opportunité en paro-implantologie: Le prf. Implantodontie. 2001;42(55):e62.
29. El Bagdadi K, Kubesch A, Yu X, et al. Reduction of relative centrifugal forces increases growth factor release within solid platelet-rich-fibrin (PRF)-based matrices: A proof of concept of LSCC (low speed centrifugation concept). Eur J Trauma Emerg Surg. 2017. doi: 10.1007/s00068-017-0785-7. [Epub ahead of print]