Controlled Palatal Harvest (CPH) Technique for Harvesting a Palatal Subepithelial Connective Tissue Graft
Neel B. Bhatavadekar, BDS, MS, MPH; and Amit S. Gharpure, BDS
ABSTRACT
Subepithelial connective tissue grafts (SCTGs) have been extensively used to augment soft-tissue volume on dental implants and natural teeth. The authors present a technique called the controlled palatal harvest (CPH) to obtain SCTGs. CPH provides an alternative to the trap-door and single-incision techniques currently utilized. The objective of this article is to describe this harvesting technique for SCTGs. A case report is presented in which the CPH technique was successfully used to obtain a SCTG. Contrary to current techniques where the SCTG is usually taken from the palatal (bone) side, this technique involves the dissection of a thick split-thickness flap in which the graft is harvested from the raised flap. Further, an L-shaped incision (with an anterior release) improves visibility and dexterity during dissection of the connective tissue graft. This technique enables the operator to maintain a certain minimum overlying flap thickness, and excellent control is ensured over the thickness of the harvested SCTG. The thickness of the remaining connective tissue overlying the palatal can also be controlled by the operator. Thus, the chances of leaving behind a thin tissue covering the palatal bone are reduced, minimizing necrosis and flap sloughing, which further improves grafting success. While the CPH can be used as an alternative to currently employed conventional harvest techniques, it may require greater operator skill. To the best of the authors' knowledge, this is the first report that documents such a technique.
Autogenous subepithelial connective tissue grafts (SCTGs) have been well documented as an effective treatment modality to enhance soft-tissue volume in periodontal plastic surgery and implantology. They have been extensively used for periodontal procedures such as esthetic soft-tissue thickening, treatment of recession, and papilla reconstruction, as well as for soft-tissue augmentation for dental implants.1-3
Various surgical techniques have been proposed to harvest SCTGs from the palate. The trap-door technique introduced by Edel involves a single horizontal incision parallel to the gingival margin and two vertical releasing incisions to achieve sufficient visual access.4 Similarly, Langer and Langer have documented a technique in which a second horizontal parallel incision is made to obtain a graft with an epithelial margin.5 A technique by Bruno involves two incisions, one perpendicular to the long axis of teeth and the second parallel to it, and the graft tissue is removed with the aid of a periosteal elevator.6 A wedge of connective tissue with an epithelial collar has been proposed by Raetzke using two semilunar converging incisions.7
Another technique by Harris involves the use a double-bladed scalpel to harvest a 1.5-mm-thick graft with an epithelial margin.8 Hurzeler and Weng have presented a single-incision technique that promotes primary healing and improves patient comfort.3 Although it provides better healing and patient comfort than the trap-door technique,9 it is technically more demanding due to limited visual access.Finally, Liu and Weisgold have demonstrated various incision techniques and have a proposed classification of palatal incision design for SCTG.10
The objective of this article is to demonstrate a technique called the controlled palatal harvest (CPH) to obtain a SCTG of optimum thickness. The salient features of the technique involve an L-shaped incision (one vertical release anteriorly) and the graft being harvested from a thick palatal flap rather than from the palatal bone side. The article describes the harvesting technique of an SCTG and demonstrates its application in a case report, with primary focus on the harvest procedure. To the best of the authors' knowledge, this is first report documenting such a technique.
Anatomic Considerations of the Donor Site
The palatal harvest is commonly used for SCTGs, and the choice of surgical technique is often determined by the anatomic considerations of the donor site.1,9 The thickness of the palatal mucosa plays an integral role in the amount of graft tissue thickness. The second premolar and molar region has an average thickness of about 3 mm.11 Its thickness increases from the canine to premolar region, decreases at the first molar region, and increases again in the second molar region.12 Thus, the canine to premolar region seems to be the most appropriate donor site with a uniform thickness.
Other factors that determine the graft dimensions are the greater palatine artery (GPA) and relatively medially placed greater palatine nerve (GPN), which emerge from the greater palatine foramen (GPF), run anteriorly along the palate in a groove, and end at the incisive canal.13 In the canine area, the GPA usually is located 12 mm from the gingival margins, and in the first and second molar regions, this distance is 12 mm to 14 mm.13,14 Additionally, the shape of the palatal vault has been shown to have a correlation with the location of the GPA.15 In shallow palates, the GPA is closer to the gingival margin.15 The distance of the GPA from the gingival margin determines the width of the graft that can be obtained. The GPF, which is usually located in the second or third molar region,16,17 determines the posterior extent of the incision.
Surgical Technique
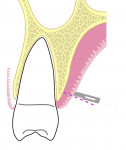
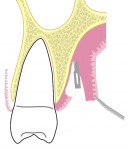
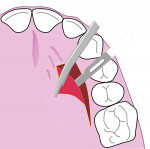
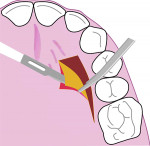
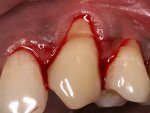
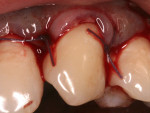
All surgical procedures are performed under local anaesthesia with 2% lignocaine containing 1:100,000 epinephrine. After preparation of the recipient site, the dimensions of the desired graft are assessed using a standardized periodontal probe. As shown in Figure 1 and Figure 2 an incision is made 2 mm apical to the gingival margin extending anteriorly from the first molar using a standardized No. 15 scalpel blade. A vertical L-shaped incision, roughly 6 mm in length, is made at the anterior end of the first incision (Figure 1, Video 1). A thick split-thickness flap is raised from the edges of the incision leaving behind a thin periosteum covering the palatal bone (Figure 3 and Figure 4, Video 1).
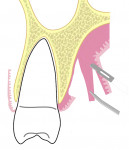
Care is taken to maintain a uniform thickness of the flap. An attempt is made to obtain the greatest possible flap thickness while keeping the periosteum and epithelium intact. The entire thickness of the flap is held with a tissue forceps, and the graft is then harvested from the raised flap by dissecting it, leaving a 1.5-mm-thick flap to ensure adequate flap healing (Figure 5 and Figure 6, Video 1). This ensures good control over the thickness of both the harvested graft and the flap. The graft can then be stabilized at the recipient site. Optionally, a bovine collagen tape can be placed at the harvested site,18,19 and primary closure is obtained for the palatal flap using 5-0 polyglactin 910 sutures (Video 2).
Clinical Presentation, Case Management, and Outcomes
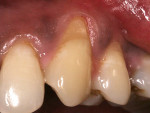
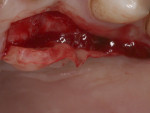
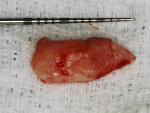
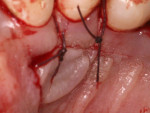
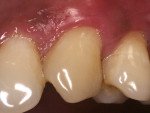
A 37-year-old male patient presented with sensitivity and esthetic complaints in the region of tooth No. 11 (Figure 7). The patient was a non-smoker and had no significant medical or dental histories. Oral examination of the tooth revealed a Miller's Class I recession defect (Figure 7). A treatment option involving the use of a SCTG from the palate for root coverage was presented to the patient. He agreed to the treatment plan, and a SCTG was performed using the CPH technique (Figure 8), enabling procurement of a graft of adequate thickness (Figure 9). The recipient site was prepared to receive the graft (Figure 10), and the graft was stabilized using 5-0 polyglactin 910 sutures (Figure 11). The raised palatal was sutured back using 5-0 polyglactin 910 sutures (Figure 12).
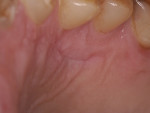
One-week recall demonstrated good healing and epithelial growth. At 6 weeks post-surgery, excellent healing was evident at the donor site (Figure 13) and the sensitivity was markedly decreased. At a 12-month follow-up, the recipient site showed excellent stability and the patient was happy with the esthetic outcome (Figure 14).
Discussion
This article demonstrates how the CPH technique can be successfully used to obtain an SCTG for soft-tissue augmentation around implants as well as natural teeth. This technique can also be used for harvesting autogenous connective tissue as an adjunct to augment a ridge preservation procedure. In the traditional approach to harvesting connective tissue grafts, the surgeon elevates a relatively thin split-thickness palatal flap and harvests the SCTG that remains attached to the underlying bone. In the CPH technique, a thick split-thickness flap is elevated, leaving minimal tissues and periosteum attached to bone, and the graft is harvested from the inner surface of the freely mobile split-thickness flap. In both cases, the external flap is replaced to cover the denuded bone. The advantages of this technique are adequate control due to good visibility, good predictability in ensuring adequate graft and flap thickness, ability to obtain uniform graft and flap thickness, and less chance of leaving behind a thin flap for closure at the donor site thereby minimizing necrosis and flap sloughing and improving grafting success.
Although this technique has many benefits, like all other SCTG techniques, it is technique-sensitive and dependent on the thickness of the palatal mucosa. In cases with thin mucosa, there is a chance of perforation of the flap while obtaining the graft. Also, while harvesting from under the ruggae area, perforation could occur due to the extent of the epithelial invaginations, which cannot be visualized. As with any incision perpendicular to the blood supply, increased bleeding is possible. Because the harvest procedure is done from a relatively mobile flap, greater clinician skill is required compared to conventional techniques.
The authors believe that the CPH technique may provide some advantages over the trap-door and single-incision techniques and could be a viable alternative for obtaining SCTGs. Although this article demonstrates successfully treated cases using the CPH technique, further research in the form of a randomized controlled clinical trial is needed to determine whether the CPH technique provides better outcomes and less morbidity than other palatal harvest techniques.
Video 1
Video 2
About the Authors
Neel B. Bhatavadekar, BDS, MS, MPH
Adjunct Faculty, University of North Carolina School of Dentistry, Chapel Hill, North Carolina; Adjunct Faculty, Bioengineering Department, Rice University, Houston, Texas; Director, Private Practice, Clarus Dental Specialities, Pune, India
Amit S. Gharpure, BDS
Resident, Graduate Periodontics, University of Washington School of Dentistry, Seattle, Washington
References
1. Zuhr O, Baumer D, Hurzeler M. The addition of soft tissue replacement grafts in plastic periodontal and implant surgery: critical elements in design and execution. J Clin Periodontol. 2014;41(suppl 15):S123-S142.
2. Thoma DS, Buranawat B, Hammerle CH, et al. Efficacy of soft tissue augmentation around dental implants and in partially edentulous areas: a systematic review. J Clin Periodontol. 2014;41(suppl 15):S77-91.
3. Hurzeler MB, Weng D. A single-incision technique to harvest subepithelial connective tissue grafts from the palate. Int J Periodontics Restorative Dent. 1999;19(3):279-287.
4. Edel A. Clinical evaluation of free connective tissue grafts used to increase the width of keratinised gingiva. J Clin Periodontol. 1974;1(4):185-196.
5. Langer B, Langer L. Subepithelial connective tissue graft technique for root coverage. J Periodontol.1985;56(12):715-720.
6. Bruno JF. Connective tissue graft technique assuring wide root coverage. Int J Periodontics Restorative Dent. 1994;14(2):126-137.
7. Raetzke PB. Covering localized areas of root exposure employing the “envelope” technique. J Periodontol. 1985;56(7):397-402.
8. Harris RJ. A comparison of two techniques for obtaining a connective tissue graft from the palate. Int J Periodontics Restorative Dent. 1997;17(3):260-271.
9. Fickl S, Fischer KR, Jockel-Schneider Y, et al. Early wound healing and patient morbidity after single-incision vs. trap-door graft harvesting from the palate--a clinical study. Clin Oral Investig. 2014;18(9):2213-2219.
10. Liu CL, Weisgold AS. Connective tissue graft: a classification for incision design from the palatal site and clinical case reports. Int J Periodontics Restorative Dent. 2002;22(4):373-379.
11. Muller HP, Schaller N, Eger T, Heinecke A. Thickness of masticatory mucosa. J Clin Periodontol. 2000;27(6):431-436.
12. Song JE, Um YJ, Kim CS, et al. Thickness of posterior palatal masticatory mucosa: the use of computerized tomography. J Periodontol. 2008;79(3):406-412.
13. Monnet-Corti V, Santini A, Glise JM, et al. Connective tissue graft for gingival recession treatment: assessment of the maximum graft dimensions at the palatal vault as a donor site. J Periodontol. 2006;77(5):899-902.
14. Benninger B, Andrews K, Carter W. Clinical measurements of hard palate and implications for subepithelial connective tissue grafts with suggestions for palatal nomenclature. J Oral Maxillofac Surg. 2012;70(1):149-153.
15. Reiser GM, Bruno JF, Mahan PE, Larkin LH. The subepithelial connective tissue graft palatal donor site: anatomic considerations for surgeons. Int J Periodontics Restorative Dent. 1996;16(2):130-137.
16. Klosek SK, Rungruang T. Anatomical study of the greater palatine artery and related structures of the palatal vault: considerations for palate as the subepithelial connective tissue graft donor site. Surg Radiol Anat. 2009;31(4):245-250.
17. Ikuta CR, Cardoso CL, Ferreira-Junior O, et al. Position of the greater palatine foramen: an anatomical study through cone beam computed tomography images. Surg Radiol Anat. 2013;35(9):837-842.
18. Shaw N. Textured collagen, a hemostatic agent. A pilot study. Oral Surg Oral Med Oral Pathol. 1991;72(6):642-645.
19. Bhatavadekar N. Peri-implant soft tissue management: Where are we? J Indian Soc Periodontol. 2012;16(4):623-627.