Use of the Straumann® AlloGraft Ring With Simultaneous Implant Placement: A Novel Approach
Robert J. Miller, DMD; Randi J. Korn, DMD; and Ross J. Miller
Abstract:
Recent advances in guided bone regeneration include the use of the Straumann® AlloGraft Ring, which can be used for the repair of both horizontal and vertical ridge deficiencies. Augmentation procedures are performed in conjunction with simultaneous placement of a bone-level and bone-level tapered implant, which is inserted through the allograft ring and stabilized in the patient’s native bone. Indications for this one-stage procedure include augmentation therapy such as fenestration- or dehiscence-type lesions, thin buccal plates, gap-type lesions, and sinus floor elevations. The concept typically reduces treatment time and can be considered in cases involving large extraction sites.
The concept of guided bone regeneration (GBR) has its roots in guided tissue regeneration (GTR), which first appeared in the dental literature in the late 1970s.1 It was later defined in the 1980s as the use of a cell occlusive barrier membrane to affect bone growth in periodontal defects around teeth.2 The use of a Millipore filter to exclude soft tissue and allow the pluripotential cells from the native bone to restore the lost periodontium was considered a paradigm shift by some.3 GBR defined as the use of cell occlusive membranes to regenerate bone for the placement of dental implants follows many of the principles of GTR and has become a predictable therapeutic modality used routinely in many practices.4-6 Advances in the procedure have come primarily from membrane or bone technology and biologics such as bone morphogenic proteins.7
GBR can be performed as either a one-stage or two-stage procedure. A two-stage procedure involves the use of a bone graft with the goal of regenerating a ridge defect or extraction site for the ultimate placement of a dental implant. Soft-tissue compression of the graft will often limit the bone volume yield, necessitating secondary grafting at the time of implant placement. This would ensure a “restoratively driven” implant placement with a thick buccal plate and a more esthetic rehabilitation with the re-establishment of a “root eminence.”8,9
A one-stage procedure refers to the placement of a dental implant with a simultaneous GBR procedure.10 This is often used to cover exposed threads, repair a buccal plate, or treat “gap-type” lesions.11 Simultaneous placement requires good initial stability for predictability. Typically, surgeons measure stability with either insertion torque or implant stability quotient (ISQ) values. Adverse events, including implant failure, are a consequence of placing implants with less-than-optimal stability.12 A recommendation for immediate placement in extraction sites with small gap-type lesions is to ensure that at least 3 mm to 4 mm of the implant is secured in native bone.13 Other potential problems include apical fenestration, dehiscence lesions, or placement of implants that may be outside of the “bony envelope.”14 Many times in these instances a grey hue from the implant shows through in thin tissue biotypes and there is a potential for failure. Immediate placement in a pneumatized sinus has never been predictable due to an inability to stabilize the fixture.
Several options are available to surgeons with respect to bone grafting materials. Autogenous bone is considered by many to be the “gold standard.” This is due to both its osteoconductive and osteoinductive capabilities. Autogenous bone typically has the most osteogenic potential among bone grafting materials because of an abundance of pluripotential cells and their ability to stimulate growth of new bone. However, the need for a secondary site from which to harvest donor material, morbidity, additional cost, and potential limitations in the quantity of material that can be harvested often lessens patient acceptance.15 Many practitioners are also concerned about the rapid resorption rate of autogenous bone, which limits its ability to act as a scaffold. Typically, patients seek alternatives; these include allografts, xenografts, and synthetic bone substitutes.16
The use of mineralized and demineralized freeze-dried bone allograft has gained acceptance, as it is morphologically similar to the patient’s own bone. Though it is typically not osteoinductive, some research does suggest there is an osteogenic potential for demineralized freeze-dried bone.17,18 An advantage of this material is its abundant availability for larger grafts; however, due to its relatively rapid rate of resorption secondary grafting is often necessary.19
Other grafting material options for GBR include xenografts, such as bovine bone, and synthetic materials, including hydroxyapatite (HA) and beta-tricalcium phosphate (ß-TCP). Advantages of these materials are their osteoconductive nature and relatively long resorption time. This becomes advantageous if used in tandem with autogenous bone or an allograft when building or maintaining a buccal plate or sinus floor elevation.20
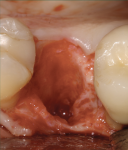
The allograft ring can be used for the repair of both horizontal and vertical ridge deficiencies (Figure 1). Augmentation procedures are performed in conjunction with simultaneous implant placement. A bone-level tapered implant is inserted through the allograft ring and stabilized in the patient’s native bone (Figure 2). Aggressive thread design provides the initial stability necessary to ensure predictable osseointegration. Indications include augmentation procedures such as fenestration- or dehiscence-type lesions, thin buccal plates, gap-type lesions, and sinus floor elevations. The time-efficient concept has been gaining acceptance, as it enables the simultaneous placement of the dental implant.21 In addition, patients are often pleased that it is a one-stage procedure.
This article depicts four case studies that describe the use of an allograft ring to treat horizontal and vertical deficiencies, with the latter two cases demonstrating its use in large extraction sites.
Case 1
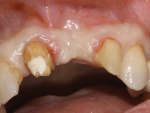
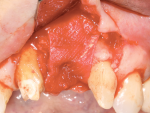
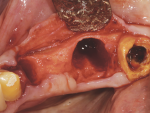
The patient was a healthy 57-year-old woman who lost tooth No. 9 due to root fracture after endodontic therapy and post and crown placement. The tooth was removed and an implant was placed after tissue maturation. Unfortunately, the fixture failed to osseointegrate and was subsequently removed. She was placed in a three-unit provisional, which became frustrating to her, as she was hoping for individual teeth (Figure 3). Ultimately, the patient sought a second opinion seeking an implant solution. She presented to the authors hopeful that an implant could be placed and asking if the labial ridge defect could be repaired.
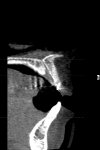
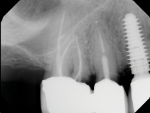
After a thorough restorative evaluation a computed tomography (CT) scan was exposed, revealing a buccal/palatal ridge dimension of approximately 5.8 mm. A relatively large incisive foramen was noted that would interfere with a restoratively driven implant placement without encroachment (Figure 4). It was also determined that an augmentation procedure would help to create a “root eminence.” The authors discussed the need to reduce the size of the canal and potential for some minor paresthesia. The allograft ring concept appeared to be the optimal treatment because it would enable a simultaneous augmentation and implant placement. The case was treatment planned with the patient’s restorative dentist and presented to the patient as a team.
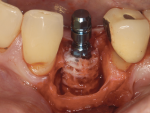
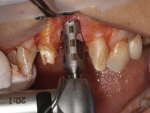
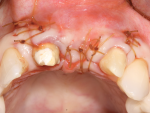
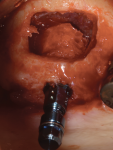
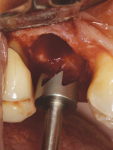
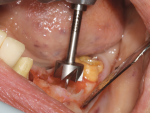
The procedure was performed using an aseptic surgical technique. The area was anesthetized with lidocaine 2% (1:100,000 epinephrine). Full-thickness flap elevation was performed from teeth Nos. 7 through 11 exposing the osseous crest. A vertical releasing incision and split-thickness flap elevation apical to the cementoenamel junction was performed to create a tension-free flap. The incisive nerve was carefully dissected to avoid trauma and palatal paresthesia (Figure 5). Standard drilling procedure was performed as described by the implant manufacturer for a 3.3-mm-diameter bone-level tapered implant (Straumann® Bone Level Tapered Implant).22 Precise 3-dimensional implant positioning was determined for a restoratively driven implant placement. After use of the initial 2-mm twist drill, a 7-mm-diameter trephine drill was used to create an osteotomy that could receive a 7-mm allograft ring (Figure 6). Note that at least 1 mm of distance must be preserved between the allograft ring and an adjacent tooth.
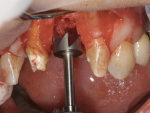
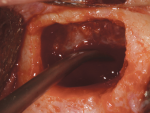
The residual excess bone was removed using a blunt instrument. Use of a planator (Planator 7mm – stainless steel, Straumann) was necessary to flatten the floor of the osteotomy, enabling secure seating of the allograft ring (Figure 7). A diamond disc was used to trim the ring to the appropriate height of approximately 6.5 mm (Figure 8).
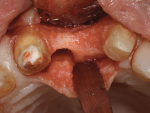
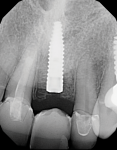
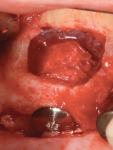
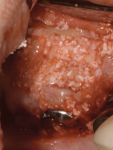
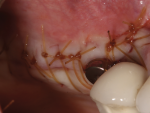
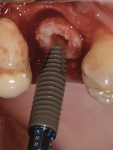
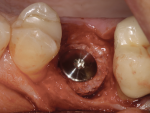
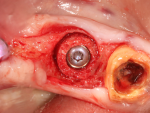
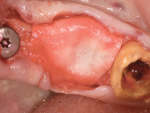
After completing the osteotomy with the 2.8-mm twist drill, a 3.3-mm x 12-mm bone-level implant (Straumann) was placed through the allograft ring and secured in the native bone (Figure 9). The implant had good initial stability, as more than 4 mm of the implant body was in native bone. All sharp edges were removed with a slow-speed diamond bur. A bovine xenograft was used to augment the buccal plate, and a collagen resorbable membrane used to cover the graft was secured under the buccal and palatal flaps (Figure 10). The site was closed using 4-0 mild chromic gut sutures (Figure 11). Initial healing was uneventful with resolution of the soft and hard tissue noted after 4 weeks (Figure 12 and Figure 13).
Case 2
In this case the patient, a healthy 67-year-old man, presented with recurrent decay under the crowns on teeth Nos. 3 and 4. Due to the extent of the caries and nonfeasibility of restoring the teeth predictably it was decided that strategic removal of teeth and placement of dental implants would be the treatment of choice. The plan included a two-stage procedure in which teeth Nos. 3 and 4 would be removed, and along with their removal the decay would be eliminated, followed by immediate placement of a dental implant in position No. 4 (Figure 14). Due to sinus proximity and a paucity of native bone it was decided to allow the upper right molar extraction socket to heal prior to a simultaneous augmentation procedure and fixture placement. The treatment plan was presented to the patient who accepted therapy, as the total treatment time would be reduced and secondary surgery would be avoided because of the simultaneous implant placement and sinus floor elevation.
The upper left posterior sextant was anesthetized with lidocaine 2% (1:100, 0000 epinephrine). Buccal and palatal full-thickness flap elevation was performed from Nos. 13 through 15. Standard surgical protocol was followed for the extraction of teeth Nos.13 and 14 with simultaneous placement of a 4.8-mm x 12-mm Regular Connection Straumann® Bone Level Tapered Implant in the extraction socket of No. 13. A freeze-dried bone graft and resorbable collagen membrane were used to regenerate the extraction site of position No. 14. The initial GBR procedure would ensure a straightforward second surgery, enabling the authors to attain primary closure and provide a stable sinus floor to secure an allograft ring and implant.
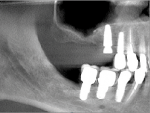
The area was reevaluated for surgery in position No. 14 (Figure 15) after exposure of a CBCT (Figure 16). The scan revealed approximately 3 mm of native bone and a clear sinus, making the patient a good candidate for sinus floor elevation and simultaneous implant placement. The area was anesthetized with lidocaine 2% (1:100,000 epinephrine). Full-thickness flap elevation was utilized to gain access to the lateral wall of the maxillary right sinus. A piezo-surgery unit was used to carefully create the lateral window in the area of position No. 14. The Schneiderian membrane was carefully detached and reflected from the inner aspect of the sinus cavity, allowing the visualization of the floor and medial aspect (Figure 17).
A 7-mm-diameter allograft ring, which would subsequently accept a 4.1-mm x 10-mm Regular Connection Straumann Bone Level Tapered Implant, was trimmed to enable its placement through the lateral window. Preparation of the implant bed included use of 2-mm, 2.8-mm, and 3.5-mm twist drills. Under-preparation and compression necrosis was avoided by using the profile drill. The implant was placed via a transalveolar approach into the allograft ring and secured to the residual bone using a closure screw and fixation cap (Figure 18 and Figure 19). A combination of particulate mineralized freeze-dried bone and bovine bone xenograft was used to fill the remaining sinus cavity (Figure 20), and a resorbable collagen membrane was placed to cover the lateral window. The site was closed with 4-0 mild chromic gut sutures using a combination of horizontal mattress and interrupted suturing (Figure 21).
Case 3
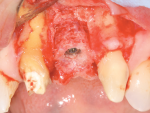
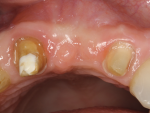
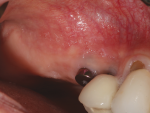
A 46-year-old female patient presented with a hopelessly involved upper left first bicuspid. Strategic extraction was the treatment of choice due to recurrent decay and circumferential bone loss. The tooth was removed, which left her with a large extraction socket that did not lend itself to immediate placement of a dental implant with a particulate graft (Figure 22). It was determined, however, that immediate placement could be achieved using an allograft ring.
A 7-mm-diameter trephine was used to prepare the site to a depth of approximately 6 mm. After reshaping of the extraction socket was done with a trephine and flattening of the floor with a planator (Figure 23), an allograft ring was able to be placed within the osteotomy. A 4.1-mm x 12-mm Regular Connection Straumann Bone Level Tapered Implant was placed and stabilized by apical native bone and an allograft ring (Figure 24). A particulate allograft bone was used to fill the remainder of the gap-type lesion and augment the buccal plate, and the implant was secured with a cover screw and fixation cap (Figure 25). The site was covered with a resorbable collagen membrane and closed with 4-0 mild chromic gut sutures.
Case 4
The patient, a 67-year-old woman in good health, presented with recurrent decay under teeth Nos. 18 and 20, which were pier abutments for a five-unit bridge. The treatment plan was to provisionalize the area from Nos. 17 through 21, strategically extract teeth Nos. 18 and 20, and place implants in the extraction sites. The site would be provisionalized with an acrylic fixed partial denture from Nos. 17 through 21 with metal reinforcement. Tooth No. 17 would be extracted after delivery of the final prosthesis.
After extraction of teeth Nos. 18 and 20 it was discovered that immediate placement was not possible in position No. 18 due to root morphology and the site’s large circumference (Figure 26). Treatment options included a two-stage procedure with particulate regenerating the ridge prior to implant placement or a single-stage approach with an allograft ring and simultaneous implant placement. The CT scan revealed a favorable ridge morphology, indicating there was adequate bone medial to the mandibular canal and mental foramen for implant placement.
The site required minimal preparation with a 7-mm trephine due to the conical nature of the extraction socket. The planator was then used to flatten the floor (Figure 27), enabling the placement of a stable allograft ring. The allograft ring and implant were secured using the apical native bone (Figure 28). An allograft particulate was used to fill in the residual gap-type lesion and was covered with a resorbable collagen membrane (Figure 29). Tension-free primary closure was attained using 4-0 mild chromic gut sutures (Figure 30).
Discussion
From a historical perspective, before the widespread acceptance of GBR as a predictable treatment modality, implant placement was typically confined to areas where clinically adequate quality and quantity of bone was present. Predictable areas included the mandibular anterior sextant where dense bone is typically located.23
The initial Brånemark protocol encompassed the extraction of hopelessly involved teeth, with a thorough debridement of the affected sites. Recommendations included 12 months of hard- and soft-tissue maturation prior to implant placement. Six months of healing in the mandible or 8 months in the maxilla were necessary for osseointegration, which was needed for the initial smooth-surface, commercially pure titanium external hex implants prior to stage II implant uncovering.24 Treatment could last as long as 2 years, which confounded many patients.
Prior to the advent of sinus augmentation, tilted implants were used in the maxillary tuberosity to facilitate molar occlusion or obtain a greater anterior-posterior spread, increasing the cross-arch stability.25 Other procedures that helped to enable implant placement in the maxillary molar region included the transplantation of osseointegrated implants from the chin area. Typical clinical presentation included sinus proximity in the molar region. Sinus floor elevation procedures were still being refined and alternative procedures such as this were being investigated. Autogenous transplantation was a relatively predictable procedure; however, it did not become mainstream because of the morbidity associated with multiple procedures necessary to access the chin area apical to the mandibular anterior teeth and the potential for devitalizing the lower incisors.26
The advent of the allograft ring enables predictable immediate placement of a dental implant in many clinical situations that would otherwise require a two-stage approach using particulate bone. This technique affords the surgeon ample flexibility that can be incorporated in a variety of situations. The allograft ring lends itself effectively to immediate placement in many sites requiring horizontal and vertical augmentation.
Other situations in which an allograft ring can be considered involve larger extraction sites, including those of single- and multi-rooted teeth that typically require multiple procedures. Traditionally, a GBR procedure would be necessary to regenerate the extraction socket prior to placement of a dental implant, adding time, cost, and a second surgical procedure. As demonstrated in Case 3 and Case 4 above, this can be achieved in a single procedure with use of an allograft ring if adequate apical native bone is present to provide good initial stability of the implant.
Conclusion
The allograft ring is an innovative tool surgeons can use to repair both horizontal and vertical ridge defects in a safe and predictable fashion. This case study, which serves to display technique, indications, and a brief historical perspective, reports the initial results of the authors, who are among the early US practitioners performing procedures using this method. The authors have seen patient acceptance that has been gratifying, as implants can be placed at the time of the regenerative procedure, reducing total treatment time and number of procedures. This has been particularly valuable in sinus floor elevation procedures, as implants can be loaded after 6 months of hard-tissue maturation.
About the Authors
Robert J. Miller, DMD
Private Practice
Periodontist
South Florida Periodontics and Implant Solutions
Plantation, Florida
Fellow
International Team for Implantology (ITI)
Randi J. Korn, DMD
Private Practice
Periodontist
South Florida Periodontics and Implant Solutions
Plantation, Florida
Diplomate
American Board of Periodontology
Ross J. Miller
DMD candidate 2018
University of Pennsylvania School of Dental Medicine
Philadelphia, Pennsylvania
References
1. Melcher AH. On the repair potential of periodontal tissues. J Periodontol. 1976;47(5):256-260.
2. Gottlow J, Nyman S, Lindhe J, et al. New attachment formation in the human periodontium by guided tissue regeneration. Case reports. J Clin Periodontol. 1986;13(6):604-616.
3. Becker W, Becker B, Prichard J, et al. Root isolation for new attachment procedures. A surgical and suturing method: three case reports. J Periodontol. 1987;58(12):819-826.
4. Dahlin C, Linde A, Gottlow J, Nyman S. Healing bone defects using guided tissue regeneration. Plast Reconstr Surg. 1988;81(5):672-676.
5. Buser D, Dula K, Belser U, et al. Localized ridge augmentation using guided tissue regeneration. 1. Surgical procedure in the maxilla. Int J Periodontics Restorative Dent. 1993;13(1):29-45.
6. Hammerle CH, Karring T. Guided bone regeneration at oral implant sites. Periodontol 2000. 1998;17:151-175.
7. Jones AA, Buser D, Schenk R, et al. The effect of rhBMP-2 around endosseous implants with and without membranes in the canine model. J Periodontol. 2006;77(7):1184-1193.
8. Weber HP, Fiorellini JP, Buser DA. Hard-tissue augmentation for the placement of anterior dental implants. Compend Contin Educ Dent. 1997;18(8):779-791.
9. Tonetti MS, Cortellini P, Graziani F, et al. Immediate versus delayed implant placement after anterior single tooth extraction: the timing randomized controlled clinical trial. J Clin Periodontol. 2017;44(2):215-224.
10. Lazzara RJ. Immediate implant placement into extraction sites: surgical and restorative advantages. Int J Periodontics Restorative Dent. 1989;9(5):332-343.
11. Botticelli D, Berglundh T, Buser D, Lindh J. The jumping distance revisited: An experimental study in the dog. Clin Oral Implants Res. 2003;14(1):35-42.
12. Ostman PO, Hellman M, Wendelhag I, Sennerby L. Resonance frequency analysis measurements of implants at placement surgery. Int J Prosthodont. 2006;19(1):77-83.
13. Lang NP, Pun L, Lau KY, et al. A systematic review on survival and success rates of implants placed immediately into fresh extraction sockets after at least 1 year. Clin Oral Implants Res. 2012;23 suppl 5:39-66.
14. Stavropoulos A, Nyengaard JR, Kostopoulos L, Karring T. Implant placement in bone formed beyond the skeletal envelope by means of guided tissue regeneration: an experimental study in the rat. J Clin Periodontol. 2005;32(10):1108-1115.
15. Sakkas A, Wilde F, Heufelder M, et al. Autogenous bone grafts in oral implantology–is it still a “gold standard”? A consecutive review of 279 patients with 456 clinical procedures. Int J Implant Dent. 2017;3(1):23.
16. Aghaloo TL, Moy PK. Which hard tissue augmentation techniques are the most successful in furnishing bony support for implant placement? Int J Oral Maxillofac Implants. 2007;22(suppl):49-70.
17. Buser D, Hoffmann B, Bernard JP, et al. Evaluation of filling materials in membrane-protected defects. A comparative study in the mandible of miniature pigs. Clin Oral Implants Res. 1998;9(3):137-150.
18. Bosshardt DD, Wallkamm B, Schenk RK, et al. Uptake of bone-related matrix proteins into implanted deproteinized bovine bone mineral. Bone. 2006,38(3 suppl 1):S8-S9.
19. Committee on Research, Science, and Therapy of the American Academy of Periodontology. Tissue banking of bone allografts used in periodontal regeneration. J Periodontol. 2001;72(6):834-838.
20. Fugazzotto PA. GBR using bovine bone matrix and resorbable and nonresorbable membranes. Part 1: histologic results. Int J Periodontics Restorative Dent. 2003;23(4):360-369.
21. Giesenhagen B. Klinische Fünfjahresergebnisse nach einzeitiger Augmentation mit autologen Knochenringen und Ankylos-Implantaten im atrophierten Unterkiefer. Z Zahnärztl Implantol. 2015;31(1):52–63.
22. Properative planning. In: Basic information on the Surgical Procedures for the Straumann® Dental Implant System. Andover, MD: Straumann USA, LLC; NAMLIT 1017:12-23. https://www.straumann.us/dam/internet/straumann_us/resources/brochurecatalogue/brochures/en/NAMLIT_1017.pdf. Accessed October 5, 2017.
23. Adell R, Lekholm U, Brånemark PI. A 15-year study of osseointegrated implants in the treatment of the edentulous jaw. Int J Oral Surg. 1981;10(6):387-416.
24. Brånemark PI, Zarb GA, Albrektsson T. Tissue-Integrated Prostheses: Osseointegration in Clinical Dentistry. Chicago, IL: Quintessence Publishing; 1985.
25. Bahat O. Osseointegrated implants in the maxillary tuberosity: report on 45 consecutive patients. Int J Oral Maxillofac Implants. 1992;7(4):1-21.
26. Lazzara, RJ. Transplantation of a preosseointegrated implant from the mental area to a maxillary sinus graft. Int J Periodontics Restorative Dent. 1995;15(6):538-547.