Nanotechnology in Dentistry: Clinical Applications, Benefits, and Hazards
Govind Shashirekha, MDS; Amit Jena, MDS; and Satyajit Mohapatra, MDS
Abstract
Nanotechnology is emerging as an interdisciplinary field that is undergoing rapid development and has brought about enormous changes in medicine and dentistry. Nanomaterial-based design is able to mimic some of the mechanical and structural properties of native tissue and can promote biointegration. Nanotechnology has various applications in dentistry, including dentition renaturalization, therapy for dentin hypersensitivity, complete orthodontic realignment in a single visit, covalently bonding diamondized enamel, enhancing properties of root canal sealers, and continuous oral health maintenance using mechanical dentifrobots. A range of synthetic nanoparticles such as hydroxyapatite, bioglass, titanium, zirconia, and silver nanoparticles are proposed for dental restoration. This review focuses on the developments in the field of nanomaterials in dentistry in the form of tissue regeneration materials, implantable devices, nanocomposites, endodontic sealers, etc, and issues of patient safety.
The prefix nano emanated from the Greek word nanos, which means dwarf, and is used to describe materials whose molecular size lies in the range of 0 to 100 nanometers. One nanometer (nm) is one-billionth or 10-9 of a meter. Japanese scientist Dr. Nori Taniguchi in 1974 coined the term nanotechnology and defined it as “the processing of separation, consolidation, and deformation of materials by one atom or one molecule.”1 The possibility of molecular engineering was first described by Nobel laureate physicist Richard Feynman in 1959 at the American Physical Society meeting at the California Institute of Technology in a lecture titled There’s Plenty of Room at the Bottom. In 1987, one-time student of Feynman K. Eric Drexler, published a book titled Engines of Creation-The Coming Era of Nanotechnology. In 1991 Sumio Iijima described carbon nanotubes in an article titled Helical Microtubules of Graphitic Carbon.2
Nanomaterials
Nanomaterials are materials whose component size ranges from 0 to 100 nm in at least one dimension. These materials may be present in the form of a cluster of atoms, grains, fibres, or films, or as nanoholes. Nanomaterials possess improved properties as compared to the parent material. The change is basically of two types: (1) due to increase in the surface area; and (2) quantum effects. As the material size approaches the nanoscale dimensions, a greater number of these nanoscale materials can be incorporated with the resultant increase in surface area.3 Quantum effects refer to the optical, electric, and magnetic properties that are altered when the material approaches the smaller end of the nanoscale.
Nanomedicine, Nanorobots, and Implantable Devices
The concept of nanomedicine was first introduced by Robert A Frietas, Jr. in 1993 and was defined, as follows: “Nanomedicine is the preservation and improvement of human health using molecular tools and molecular knowledge of the human body.”4 It has diversified actions ranging from target drug delivery to creating tissue scaffolds based on nanoscale molecules.
Nanorobots are about 0.5 µm to 3 µm in diameter and made of components in nanoscale dimensions. Carbon in the form of diamonds or fullerenes is the chief component. Once inside the target tissues, these nanorobots respond to definite programs. This gives the flexibility to control and execute procedures at the cellular and molecular level. Nanorobots have found their use into various areas of medicine, including pharmaceutics, diagnostics, gene therapy, and dentistry.5
Nano implantable devices are applied in various fields ranging from tissue regeneration materials, implantable devices, osseous repair, implant coating materials, bioresorbable materials, smart materials, tissue replacement materials, and diagnostic and therapeutic aids, to cochlear materials.
Approaches to Nanodentistry
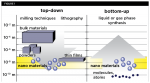
In the top-down technique, larger devices are used to assemble smaller devices (Figure 1).1 Milling, machining, and lithography are a few examples of this technique.6 The bottom-up technique refers to the use of smaller components into a more complex assembly. It involves the process of designing custom-made molecules that reorganize to higher scale structures. This method is most widely used as it proves to be more economical and cost effective.
Nanorobotic Local Anesthetics
Nanorobotic local anesthetics are composed of a colloidal solution of activated nanosized local anesthetic molecules. When applied to the gingival or the oral mucosa and signaled, the anesthetic travels via the epithelial and connective tissues of the gingiva to reach the pulp, thus providing selective anesthesia, which is under the control of the clinician. These ambulatory nano-active solutions are directed to the target site by chemical and temperature gradients. Upon reaching the pulp and establishing control over the nerve-impulse traffic, these nanorobots may be commanded to shut all neurosensory sensations to a particular tooth or multiple teeth as desired by the dentist. On completion of the procedure, the nanorobots may again be signaled to restore the sensation and, following this, they are aspirated. The advent of this technology offers greater patient comfort with minimal patient anxiety, precise selectivity, and controllability of the analgesic effect, as well as complete reversibility of the analgesic.7
Dentin Hypersensitivity
Reconstructive dental nanorobots are able to selectively and precisely block dentinal tubules, offering a quick and permanent cure. These nanorobots travel toward the dental pulp via the dentinal tubules. Desensitizing toothpaste containing 15% hydroxyapatite nanoparticles has been found to be effective in reduction of dentin hypersensitivity clinically even after single application for a period of 4 weeks.8
Dentifrobots
Dentifrobots are nanorobots incorporated into dentifrices and mouthwashes that help to clean organic residues by moving throughout the gingival tissues at a speed of approximately 10 microns/second, continuously preventing the accumulation of calculus. They can also be deactivated when accidentally swallowed by the patient.9 Dentifrobots can selectively identify and destroy pathogenic bacterial species in plaque biofilms and prevent halitosis.10
Nanodiagnostics
Nanoscale cantilevers: Flexible beams resembling rows of divided boards that bind to cancer-associated molecules.
Nanopores: These act as filters for DNA that allow only single strands of DNA to pass through them, thus allowing efficient DNA sequencing.
Nanotubes: These are nanosized carbon rods that are about half the diameter of a DNA molecule. They help to detect and identify the exact location of altered genes.
Quantum dots: These nanomaterials fluoresce when illuminated by ultraviolet light. When coated appropriately, they bind to proteins associated with cancer cells.
Nano Electromechanical Systems (NEMS): Nanotechnology-based NEMS biosensors are used for analyte detection. They help in converting (bio)chemical to an electrical signal.11
Oral fluid nanosensor test (OFNASET): This technology helps detect salivary biomarkers for oral cancers. It incorporates self-assembled monolayers (SAM), bionanotechnology, cyclic enzymatic amplification, and microfluidics for detection of salivary biomarkers. In a study, it was demonstrated that a combination of two salivary proteomic biomarkers (thioredoxin and IL-8) and four salivary mRNA biomarkers (SAT, ODZ, IL-8, and IL-1b) had high sensitivity and specificity in detection of oral cancer.12
Optical Nanobiosensor: These are minimally invasive, fiberoptic-based nanobiosensors that allow analysis of intracellular components and proteins.13
Lab-on-a-chip methods: Lab-on-a-chip combines various laboratory functions on a single chip. The analysis is done on chemically activated beads embedded onto silicon wafers. The advantage of this method includes a small sample requirement, short analysis time, and reduced cost. In dentistry, these have been used to assess the levels of interleukin-1 beta (IL-1ß), C-reactive protein (CRP), and matrix metalloproteinase-8 (MMP-8) in the whole saliva, which acts as biomarkers for diagnosing and categorizing the severity and extent of periodontitis.14
Nanomaterials
Nanocomposites: These are composite resins containing homogenously distributed nano-agglomerated nanoparticles. Aluminosilicate powder in 1:4 ratio is the most commonly used filler, with an average particle size of 80 nm.15 These nanofillers have a refractive index of approximately 1.503 and have advantages over conventional microfilled and hybrid resin-bonded composite (RBC) systems. These nanofilled composites have increased polishability, smoothness, flexural strength, and color characteristics compared to other posterior RBC.
Nanosolution (Nano adhesives): Nanosolutions manufactured using soluble nanoparticles when used in bonding agents lead to a homogenous and well-mixed adhesive consistently.16 They have high bond strength, long shelf-life, good marginal seal, fluoride release, and good stress absorption.8,15
Nano Light-curing Glass-Ionomer Restorative: Application of nanotechnology to glass-ionomer cement (GIC) was first developed for Ketac™ Nano (3M ESPE, 3mespe.com) with fluor aluminum-silicate technology having nanoparticles in the range of 1 µm.17 The addition of nanoparticles resulted in improved esthetics and polishability of the restoration.18 Another nanofilled light-cured varnish (G-Coat Plus™, GC Europe, gceurope.com) is applied onto the surface of viscous GIC (Fuji IX GP® Extra, GC Europe). This commercial product is the EQUIA (Easy-Quick-Unique-Intelligent-Aesthetic) system, which contains inorganic silica nanofillers (15 wt. % and 40-nm size) dispersed in a liquid. Nanofillers resulted in improved wear resistance by avoiding initial water intake and dehydration and decreased initial setting time. Friedl et al19 in a retrospective study of nanomodified GICs evaluated their performance over conventional GICs and concluded EQUIA restorations to be superior. Earlier study of antimicrobial property of silver nanoparticles in GIC against Streptococcus mutans has shown to be highly effective in reducing the bacterial load.20
Endodontic Sealers
Application of nanotechnology has been extended to the field of endodontics as well. Bioceramic-based sealer EndoSequence BC Sealer™ (Brasseler USA, brasselerusadental.com) containing calcium silicate, calcium phosphate, calcium hydroxide, zirconia, and a thickening agent, has been developed. The addition of nanoparticles resulted in improved handling and physical properties. When introduced into root canals, a hydration reaction occurs where a nanocomposite structure of calcium silicate and hydroxyapatite is formed. Water is essential for the setting reaction. Thus, in over-dried canals, setting time is prolonged.21 The addition of nanoparticles also facilitates delivery of material from 0.012 capillary needles and its adaption to irregular dentin surfaces. It sets hard in few hours and has good sealing ability along with dimensional stability. Its alkaline pH of (12.8) gives antimicrobial properties as well.21,22
Silicon-based sealer containing gutta-percha powder and silver nanoparticles less than 30 µm in size has been introduced as well (GuttaFlow® 2, Coltene Whaledent, nam.coltene.com). It is available as capsules that can be mixed and injected as a cold flowable filling system.21 It has good biocompatibility and dimensional stability with good sealing ability and is resistant to bacterial penetration. Recently, antibacterial quaternary ammonium polyethyleneimine (QPEI) nanoparticles have been incorporated into other sealers (eg, AH Plus®, Dentsply DeTrey, dentsply.com; Epiphany®, Pentron Clinical Technologies, pentron.com; Guttaflow, Coltene), which are relatively stable, have good biocompatibility,23 and resulted in good antibacterial activity without affecting mechanical properties.24
Impression Materials
Nanofillers integrated into vinylpolysiloxanes resulted in impression materials with better flow, improved hydrophilic properties, and enhanced reproduction of surface details.25
Nanocomposite Artificial teeth
Artificial composite teeth containing homogeneously diffused nanofillers have been reported to be superior to conventional acrylic teeth in terms of surface smoothness, abrasion resistance, and color stability.26,27 Enhanced antifungal activity along with increased fracture toughness is seen in silver nanoparticle modified denture teeth.28,29
Orthodontic Nanorobots
Orthodontic nanorobots could directly manipulate the periodontal tissues, thereby allowing a painless and rapid method for correcting malocclusion.30
Nanomaterials for Tissue Regeneration
Nanobiomaterial-based tissue scaffolds are used for pulpal cell culture. Scaffolds based on nanofibers of biodegradable type I collagen or fibronectin are used for regeneration.31,32 Pulp tissue regeneration is possible using self-assembling polypeptide hydrogels. The nanofiber mesh formed supported growing pulpal cells.33 A study conducted by Misawa et al34 has shown that cell growth was enhanced using Puramatrix containing repeats of essential amino acids (alanine, arginine, and aspartate). Other nanobiomaterials with potential dental applications for cell culture include natural silk35 and an injectable self-assembling collagen-I scaffold, which when loaded with exfoliated teeth stem cells resulted in the formation of pulp-like tissue and functional odontoblasts.36 Nanohydroxyapatite-based bone graft materials are also being used to treat large bone defects.37
Nanomaterials for Periodontal Drug Delivery
Experimented nanomaterials explored for controlled drug delivery include nanotubes, hollow spheres, core-shell structure, and nanocomposite. Drugs incorporated into nanospheres of a biodegradable polymer allows for timed release of the drug.
Triclosan-loaded nanoparticles manufactured using poly(d,l-lactide-coglycolide), poly(d,l-lactide), and cellulose acetate phthalate was found to be effective in reducing periodontal inflammation.38,39 Microspheres containing tetracycline are available as Arestin for controlled drug delivery into the periodontal pocket.10 An in vivo study observed nanostructured 8.5% doxycycline gel preserved the periodontal surface following experimentally induced periodontal disease in rats.40
Photodynamic Therapy
Antimicrobial photodynamic therapy (aPDT) is a relatively newly introduced treatment modality for removal of infectious pathogens. It uses a photosensitizer and light of a specific wavelength, eg, toluidine blue at 600-nm wavelength. Effective oral biofilm destruction with methylene blue dye (photosensitizer) encapsulated within poly(D, L-lactide-co-glycolide) (PLGA) nanoparticles (≈ 150 nm to 200 nm in diameter). A newly developed photosensitizer, indocyanine green (ICG), with loaded nanospheres when activated with 805-nm wavelength using a diode laser, has an a PDT-like effect and may serve as a potential photodynamic periodontal therapy.41
Implants
Surface characterizations of implant surfaces are increasingly being used to achieve higher success rates.42 The nanostructured implant coatings developed include (1) nanostructured diamond, with ultra-high hardness, improved toughness, low friction, and good adhesion to titanium alloys; (2) hydroxyapatite implant coatings manufactured using nanostructured processing, which has been found to increase the osteoblastic activity in terms of its adhesion, proliferation, and mineralization; and (3) nanostructured metalloceramic coatings on implant surface, which augments the osseointegration of dental implants by forming a nanocrystalline metallic bond with the implant surface and another hard ceramic bond on its surface.43
Nanoneedles
Nanosized stainless steel crystals incorporated into commercially available needles have been developed (Sandvik Bioline 1RK91™, Sandvik, smt.sandvik.com). Current research is being conducted for the development of nanotweezers that would make cell surgery possible in the near future.15
Safety Issues
Nanobiomaterial technology is extensively being applied in healthcare services largely because of its various advantages. However, with increased use, concerns about the safety of these nanobiomaterials are being raised. The increased rate of absorption associated with manufactured nanoparticles is the main concern. Nanoparticles have an increased surface area:volume ratio, which leads to increased absorption of these particles through the skin, lungs, and digestive tract. Nonbiodegradable nanoparticles when accumulated within the body may be deposited in various organs and may lead to an unwanted reaction within biological tissues.
A study conducted by the Swedish Karolinska Institute revealed that iron-oxide nanoparticles were nontoxic on human lung epithelial cells and caused no DNA damage. Zinc-oxide nanoparticles were slightly worse. Titanium dioxide caused only DNA damage; carbon nanotubes caused DNA damage at low levels. Copper oxide was found to be highly toxic and was categorized as a health risk.44
Conclusion
Advances in nanotechnology are paving the future of healthcare management. Nanodevices cannot be seen by the naked eye yet possess powerful capabilities. They have the potential to bring about significant benefits, such as improved health, better use of natural resources, and reduced environmental pollution. However, these nanodevices are also associated with significant potential misuse and abuse.
Nanodentistry aims to ensure comprehensive oral healthcare of the patient and emphasizes the primary prevention of oral diseases. With the availability of advanced and accurate diagnostic methods, a number of oral diseases can be prevented or treated at early signs.
Challenges in terms of basic molecular engineering methods, mass production techniques, and simultaneous coordination of a large number of nanorobots must be addressed prior to any large-scale application of nanotechnology.
Though the science of nanotechnology may appear as fiction in the present scenario, the future holds strong promise for utilizing and maximizing this technology for the benefit of mankind. Nanotechnology will change dentistry, healthcare, and human life profoundly. However, at the same time, social issues of public acceptance, ethics, regulation, and human safety will need to be addressed before molecular nanotechnology can enter the modern medical and dental armamentarium.
About the Authors
Govind Shashirekha, MDS
Professor
Department of Conservative Dentistry & Endodontics
Institute of Dental Sciences
Siksha ‘O’ Anusandhan University
Bhubaneswar, Odisha, India
Amit Jena, MDS
Professor and Department Head, Department of Conservative Dentistry & Endodontics
Institute of Dental Sciences, Siksha ‘O’ Anusandhan University
Bhubaneswar, Odisha, India
Satyajit Mohapatra, MDS
Private Practice
Cuttack, Odisha, India
References
1. Taniguchi N. On the Basic Concept of 'Nano-Technology, Proc. Intl. Conf. Prod. Eng. Tokyo, Part II, Japan Society of Precision Engineering, 1974.
2. Iijima S. Helical microtubles of graphitic carbon. Nature. 1991;354:56-58.
3. Drexler KE. Nanosystems: Molecular machinery manufacturing and computation. New York, New York: John Wiley and Sons; 1992.
4. Freitas RA Jr. Available from: https://www.nanomedicine.com/
5. Kubik T, Bogunia K, Sugisaka M. Nanotechnology on duty in medical applications. Curr Pharm Biotechnol. 2005;6(1):17-33.
6. Zhang L, Webster TJ. Nanotechnology, and nanomaterials: Promises for improved tissue regeneration. Nano Today. 2009;4(1):66-80.
7. Freitas RA Jr. Nanodentistry. J Am Dent Assoc. 2000;131(11):1559-1566.
8. Jena A, Shashirekha G. Comparison of efficacy of three different desensitizing agents for in-office relief of dentin hypersensitivity: A 4 weeks clinical study. J Conserv Dent. 2015;18(5):389-393.
9. Saravana KR, Vijayalakshmi R. Nanotechnology in dentistry. Ind J Dent Res. 2006;17(2):62-65.
10. Kong LX, Peng Z, Li SD, Bartold PM. Nanotechnology and its role in the management of periodontal diseases. Periodontol. 2006: 40(1):184-196.
11. Shi F, Qi B, Wu L, Wolinsky DT. Wong. The oral fluid MEMS/NEMS chip (OFMNC): diagnostic and translational applications. Adv Dent Res. 2005;18(1):3-5.
12. Gau V, Wong D. Oral fluid nanosensor test (OFNASET) with advanced electrochemical-based molecular analysis platform. Ann N Y Acad Sci. 2007;1098:401-410.
13. Song JM, Kasili PM, Griffin GD, Vo-Dinh T. Detection of cytochrome C in a single cell using an optical nanobiosensor. Anal Chem. 2004;76(9):2591-2594.
14. Christodoulides N, Floriano PN, Miller CS, et al. Lab-on-a-chip methods for point-of-care measurements of salivary biomarkers of periodontitis. Ann N Y Acad Sci. 2007;1098:411-428.
15. Jhaver HM. Nanotechnology: the future of dentistry. J Nanosci Nanotechnol. 2005;5(1):15-17.
16. Patil M, Mehta DS, Guvva S. Future impact of nanotechnology on medicine and dentistry. J Indian Soc Periodontol. 2008;12(2):34-40.
17. Chandki R, Kala M, Kumar KN, et al. Nanodentistry: exploring the beauty of miniature. J Clin Exp Dent. 2012;4(2):119-24.
18. Moshaverinia A, Ansari S, Moshaverinia M, et al. Effects of incorporation of hydroxyapatite and fluoroapatite nanobioceramics into conventional glass ionomer cements (GIC). Acta Biomater. 2008;4(2):432-440.
19. Friedl K, Hiller KA, Friedl KH. Clinical performance of a new glass ionomer based restoration system: A retrospective cohort study. Dent Mater. 2011;27(10):1031–1037.
20. Magalhães APR, Santos LB, Lopes LG, et al. Nanosilver application in dental cements. ISRN Nanotechnol. 2012;2012:e1-6.
21. Koch K, Brave D. A new day has dawned: the increased use of bioceramics in endodontics. Dentaltown. 2009;10(4):39-43.
22. Zoufan K, Jiang J, Komabayashi T, et al. Cytotoxicity evaluation of Gutta Flow and Endo Sequence BC sealers. Oral Surg Oral Med Oral Pathol Oral Radiol. Endodontol. 2011;112(5): 657-661.
23. Abramovitz I, Beyth N, Weinberg G, et al. In Vitro biocompatibility of endodontic sealers incorporating antibacterial nanoparticles. J Nanomater. 2012;2012:e1-9.
24. Beyth N, Houri-Haddad Y, Baraness-Hadar L, et al. Surface antimicrobial activity and biocompatibility of incorporated polyethyleneimine nanoparticles. Biomaterials. 2008;29(31): 4157-4163.
25. Jhaveri HM, Balaji PR. Nanotechnology: the future of dentistry. J Ind Prosthodont Soc. 2005;5(1):15-17.
26. Suzuki S. In vitro wear of nano-composite denture teeth. J Prosthodont. 2004;13(4): 238-243.
27. Ghazal M, Hedderich J, Kern M. Wear of feldspathic ceramic, nanofilled composite resin and acrylic resin artificial teeth when opposed to different antagonists. Eur J Oral Sci. 2008;116(6):585-592.
28. Chladek G, Barszczewska-Rybarek I, Lukaszczyk J. Developing the procedure of modifying the denture soft liner by silver nanoparticles. Acta Bioeng Biomech. 2012;14(1):23-29.
29. Acosta-Torres LS, Mendieta I, Nuñez-Anita RE, et al. Cytocompatible antifungal acrylic resin containing silver nanoparticles for dentures. Int J Nanomedicine. 2012;7:4777-4786.
30. Shellart WC, Oesterle LJ. Uprighting molars without extrusion. J Am Dent Assoc. 1999;130(3):381-385.
31. Imamura S, Takahashi H, Hayakawa I, et al. Effect of filler type and polishing on the discoloration of composite resin artificial teeth. Dent Mater J. 2008;27(6):802-808.
32. Fukuda J, Khademhosseini A, Yeh J, et al. Micropatterned cell co-cultures using layer-by-layer deposition of extracellular matrix components. Biomaterials. 2006;27(8):1479-1486.
33. Galler KM, Cavender A, Yuwono V, et al. Self-assembling peptide amphiphile nanofibers as a scaffold for dental stem cells. Tissue Eng Part A. 2008;14(12):2051-2058.
34. Misawa H, Kobayashi N, Soto-Gutierrez A, et al. PuraMatrix facilitates bone regeneration in bone defects of calvaria in mice. Cell Transplant. 2006;15(10):903-910.
35. Zafar MS, Al-Samadani KH. Potential use of natural silk for bio-dental applications. J Taibah Univers Med Sci. 2014;9(3):171-177.
36. Demarco FF, Conde M, Cavalcanti BN, et al. Dental pulp tissue engineering. Braz Dent J. 2011:22(1):3-14.
37. Paul W, Sharma CP. Nanoceramic matrices: biomedical applications. Am J Biochem Biotech. 2006;2(2):41-48.
38. Kohli P, Martin CR. Smart nanotubes for biomedical and biotechnological applications. Drug News Proespect. 2003;16(9):566-573.
39. Piñón-Segundo E, Ganem-Quintanar A, Alonso-Pérez V, Quintanar-Guerrero D. A preparation and characterization of triclosan nanoparticles for periodontal treatment. Int J Pharm. 2005;294(1-2):217-232.
40. Botelho MA, Martins JG, Reula RS, et al. Nanotechnology in ligature-induced periodontitis: protective effect of a doxycycline gel with nanoparticules. J Appl Oral Sci. 2010;18(4):335-342.
41. Nagahara A, Mitani A, Fukuda M, et al. Antimicrobial photodynamic therapy using a diode laser with a potential new photosensitizer, indocyanine green-loaded nanospheres, may be effective for the clearance of Porphyromonas gingivalis. J Periodontal Res. 2013;48(5):591-599.
42. Catledge SA, Fries MD, Vohra YK, et al. Nanostructured ceramics for biomedical implants. J Nanosci Nanotechnol. 2002;2(3-4):293-312.
43. Colon G, Ward BC, Webster TJ. Increased osteoblast and decreased Staphylococcus epidermidis functions on nanophase ZnO and TiO2. J Biomedical Mater Res. 2006;78(3):595-604.
44. Karlsson HL, Cronholm P, Gustafsson J, Moller L. Copper oxide nanoparticles are highly toxic: a comparison between metal oxide nanoparticles and carbon nanotubes. Chem Res Toxicol. 2008;21(9):1726-1732.