Minimally Invasive Surgery in Periodontal Regeneration: A Review of the Literature
John C. Tunnell, DDS, MS; and Stephen K. Harrel, DDS, MS
Abstract
New techniques have emerged to achieve periodontal regeneration without the drawbacks associated with conventional flap surgery. Minimally invasive surgery (MIS) is a method of surgical access that minimizes flap reflection and tissue trauma, resulting in maintenance of critical blood supply, stability of the blood clot within the wound site, and less postoperative recession over time. As advancements in materials and techniques are made, MIS in periodontal regeneration continues to evolve. The emergence of a videoscope for use during MIS (VMIS) has improved visual access to surgical sites, facilitating improved defect debridement and root planing. As demonstrated, VMIS has resulted in an actual gain in soft-tissue height up to 3 years postoperatively.
In recent decades, regenerative procedures have become a preferred method for treating destruction caused by periodontal disease. Classic techniques such as guided tissue regeneration (GTR), which involves multitooth full-thickness flap reflection, defect debridement, thorough root planing, and placement of a barrier membrane to exclude downgrowth of epithelium facilitate regeneration of lost alveolar bone, cementum, and periodontal ligament of the periodontal attachment apparatus. While GTR results in improved clinical parameters such as reduced probing depth (PD) and increased clinical attachment level (CAL),1 the need for large flap reflection for access compromises blood supply to the area, which possibly reduces regenerative potential and causes gingival recession over time.2 Thus, new techniques have been sought to achieve periodontal regeneration without the drawbacks associated with conventional flap surgery.
Harrel and Rees3 first introduced minimally invasive surgery (MIS) in the periodontal literature as a method of surgical access that minimizes flap reflection and tissue trauma, resulting in maintenance of critical blood supply, stability of the blood clot within the wound site, and less postoperative recession over time. This is accomplished through the use of tiny incisions, sharp dissection of soft tissue to gain access to the defect, and minimal suturing to avoid trauma to papillary tissues.4 Other minimally invasive approaches to achieve periodontal regeneration include periodontal microsurgery5 and the minimally invasive surgical technique (MIST) and modified MIST (M-MIST) described by Cortellini and Tonetti.6,7 MIST is a modification of MIS that uses elements of papilla preservation procedures and small incision designs for surgical access. M-MIST was subsequently introduced as an even less invasive modification of MIST; it attempts to leave the interproximal tissue attached to the tooth root surface, thereby introducing an added dimension of stability. Reported advantages of M-MIST include less trauma, increased flap and wound stability, better primary closure, faster completion of surgery, and minimized patient discomfort during and after treatment.6,7
While these techniques are all based on the same principles of minimal flap reflection and soft-tissue manipulation, MIS preferably involves access from the palatal or lingual surface while MIST and M-MIST generally attempt buccal or facial access. Regardless of the location of entry, minimal flap reflection reduces visual access to the site. To address this limitation common to MIS, MIST, M-MIST, and periodontal microsurgery, surgeons have utilized surgical loupes, a fiberoptic endoscope, a surgical microscope, and later a videoscope to improve visualization during treatment.6,8-10 Though loupes often provide only limited magnification, the fiberoptic endoscope becomes contaminated with blood and debris as it is inserted into the surgical site, and the surgical microscope can be difficult to learn and use properly, all were applied effectively in minimally invasive techniques prior to the advent of the videoscope.10
Nevertheless, as advancements in materials and techniques have been made, minimally invasive surgery in periodontal regeneration has continued to evolve. Because a comprehensive review of minimally invasive techniques that focused on MIST and M-MIST has previously been published,11 the authors’ purpose is to review the literature on MIS as used in periodontal regenerative procedures.
Review of the Literature
In their original article, Harrel and Rees described a rotary degranulating instrument that required only small incisions for surgical site access, which they referred to as a “minimally invasive surgery technique.” They also proposed the minimally invasive technique for use in periodontal regenerative procedures utilizing bone grafts and barrier membranes. They stressed the importance of making as small of an incision as possible, arguing that “excessive reflection can damage the blood supply to the flap and can lead to increased tissue loss during initial healing” and “maintenance of the blood supply to the flap and minimal trauma of the tissue are critical to the success of all regenerative surgical procedures.”3
While the initial paper alluded to the use of “small incisions,” Harrel and Rees did not thoroughly discuss their minimally invasive surgical technique. Two follow-up papers12,13 by Harrel described the procedure in more detail, highlighted the use of MIS for periodontal bone grafting, and offered several author observations during and after treatment. Per Harrel, ideal situations for MIS included isolated interproximal defects and periodontal defects bordering edentulous areas. The author suggested that while MIS could be used to treat defects extending to the buccal and/or lingual from the interproximal area, generalized horizontal bone loss and contiguous defects may be best treated with conventional flap surgery. Harrel stressed that the incisions used in MIS were designed to preserve as much of the soft tissue surrounding the defect as possible. Importantly, incisions were limited to the palatal or lingual surface whenever feasible, especially in esthetic areas. Limiting incisions to palatal or lingual tissue while leaving buccal tissue and underlying blood supply intact resulted in an improved esthetic result with minimal recession. Tissues were elevated through use of sharp dissection only, which reduced trauma to the periosteum. According to Harrel13, this minimized the “postsurgical flattening of the papilla, interproximal cratering, and loss of soft-tissue height” that often occurs with blunt flap dissection. Once access was achieved, the defect was visualized with magnification and a light source. Granulation tissue was removed using curettes, ultrasonics, and a granulation tissue removal instrument in a way that prevented trauma to the flap followed by final root planing and smoothing with a high-speed surgical-length finishing bur. Roots were then burnished with citric acid and sites were grafted with demineralized freeze-dried bone allograft (DFDBA) and covered with a surgical mesh. Finally, wound closure was accomplished with a single vertical mattress suture at the base of the papilla and papilla tips gently compressed with moist sterile gauze.12,13
Using this technique, 10 consecutively treated patients who received MIS, bone grafting, and a surgical mesh membrane had postoperative measurements taken an average of 25.1 months after surgery. Results revealed a mean PD reduction of 4.10 mm and a mean gain in CAL of 4.20 mm. Further, all teeth that were initially mobile improved in mobility and no teeth were lost.12 A subsequent paper described results obtained from 87 patients with 194 surgical sites treated in a similar fashion in a private practice setting with at least 9 months of follow-up. Postsurgical measurements recorded during periodontal maintenance appointments with a mean follow-up time of 21.7 months showed that, for all teeth included, an average reduction in PD of 4.58 mm and an average improvement in CAL of 4.87 mm occurred along with minimal changes in soft tissue height during the healing period. Additionally, improvements in PD and CAL were maintained over time.4 Thus, results from MIS are consistent with previous studies using combination GTR, bone grafting, and long-span flap reflection2 with the added advantage of improved “retention of soft-tissue height and contour, patient acceptance, and less postoperative discomfort.”4,12
In these early studies, MIS utilized bone grafting and a resorbable barrier membrane. With evidence suggesting some biologic growth factors, such as enamel matrix derivative (EMD), provide clinically similar results compared to GTR,2 several surgeons now favor these in lieu of barrier membranes. EMD has been shown in laboratory studies to upregulate production of growth factors that aid in periodontal regeneration while limiting inflammation and epithelial cell proliferation.14
To observe the effect of EMD on periodontal regeneration, Harrel et al published another case series that utilized EMD in conjunction with MIS to treat 160 sites in 16 patients in a multicenter private practice study.8 Included patients had sites with a 6 mm or greater PD after initial nonsurgical periodontal therapy. Following minimally invasive surgical access, defect debridement, root planing, and root surface modification with either citric acid or 24% ethylenediaminetetraacetic acid (EDTA), the surgeon applied EMD directly in the defect and grafted with DFDBA. Flaps were replaced and sutured, and patients were enrolled in a 3-month periodontal maintenance program. Postsurgical measurements taken at follow-up visits ranging from 11 to 24 months showed mean PD reductions of 3.56 mm, mean CAL gains of 3.57 mm, and mean gains in gingival margin to cementoenamel junction (CEJ) measurements of 0.01 mm. In drawing a comparison to summary results obtained by Garrett in his review of various periodontal regenerative procedures,1 Harrel et al determined that MIS with combination EMD and DFDBA resulted in superior reductions in PD and gains in CAL with minimal to no gingival recession as compared to flap surgery, synthetic grafts, bone grafts, or traditional GTR.8 Further, the authors reported that improvements in clinical parameters remained virtually unchanged 6 years after treatment.9
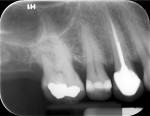
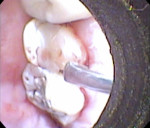
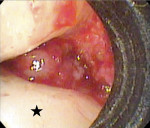
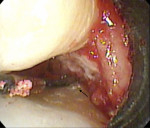
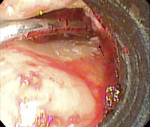
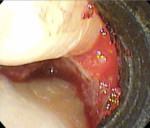
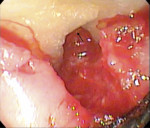
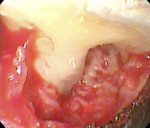
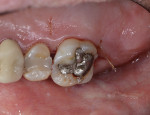
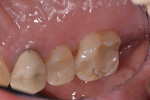
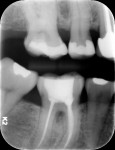
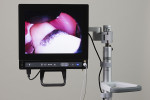
While the small incisions and minimal tissue reflection contributed to the superior results obtained using these techniques, visualization remained a primary limiting factor of MIS until the recent introduction of the videoscope (MicroSight, Q-Optics Inc, www.q-optics.com) described by Harrel et al. The videoscope is a high-definition digital camera placed on the end of a flexible tube 2.7 mm in diameter featuring a carbon fiber retractor that may be rotated to aid in flap reflection and a gas-shield that prevents blood and contamination from obscuring the camera, allowing improved visualization of the defect once inserted into the surgical site. Images taken with the videoscope are seen on a nearby surgical monitor at 20X to 60X magnification (Figure 12). Clinically, the enhanced visualization achieved with the videoscope facilitates improved defect debridement and root planing.10 Figure 13 shows the hand piece containing the videoscope that is used during surgery. A sample case treated with the videoscope and MIS (ie, VMIS) can be seen in Figure 1 through Figure 11.
Some concern exists over development of soft tissue emphysema when the gas-shield device on the end of the videoscope is inserted into the surgical site. However, a study observing the oral soft tissue resistance to emphysema in minimally invasive surgical sites in fresh pig jaws showed that the risk of soft tissue emphysema is minimal when pressures of +15 psi or less are used. As the gas-shield device is routinely used at a pressure of only 3-5 psi, there appears to be minimal risk for development of soft tissue emphysema when the videoscope is used for MIS procedures.16
To highlight the capabilities of VMIS, Harrel et al15 presented a clinical outcomes case series that featured an original cohort of 30 patients with 110 qualifying sites treated with VMIS and followed for at least 36 months. For inclusion, patients had an isolated interproximal area of periodontal destruction with a PD of at least 5 mm, CAL of at least 2 mm, and radiographic evidence of bone loss on periapical radiographs following initial nonsurgical therapy. The authors obtained minimally invasive surgical access, debrided the sites and planed the roots, biomodified root surfaces with 24% EDTA, and grafted with EMD and DFDBA. As before, flaps were repositioned and sutured while closure was ensured by digital pressure applied to the papilla tips with moist sterile gauze. Clinical measurements were recorded at baseline, 6 months, 12 months, and at least 36 months post-operatively. All 30 patients were available for the 6-month data, 18 patients were available for the 12-month data, and 14 patients were available for the 36-month or greater data, with an average post-operative time of 46.5 months ranging from 36 to 58 months. Since no statistically significant difference was found between measurements obtained for 1-wall, 2-wall, and 3-wall defects, data was combined for patient-level measurements of all surgical sites for the 14 patients who completed the study through the final evaluation. Results for these 14 patients revealed that a mean baseline PD of 6.41 ± 0.66 mm improved to 2.81 ± 0.58 mm at 6 months, 2.18 ± 0.58 mm at 12 months, and 2.61 ± 0.88 mm at 36 months+ for a mean improvement from baseline to 36 months+ of 3.80 ± 1.18 mm (P < 0.001). Measurements for gingival recession revealed that a mean gingival recession at baseline of 0.74 ± 0.95 mm improved to 0.69 ± 0.98 mm at 6 months, 0.31 ± 0.39 mm at 12 months, and 0.38 ± 0.67 mm at 36 months+ for a mean improvement (ie, gain in soft tissue height) of 0.36 ± 0.64 mm (P = 0.057) from baseline to the final post-operative visit. Finally, measurements for CAL improved from a mean baseline of 7.14 ± 0.97 mm to 3.50 ± 1.15 mm at 6 months, 2.49 ± 0.62 mm at 12 months, and 2.99 ± 1.05 mm at the 36 months+ post-operative for a mean improvement of 4.16 ± 1.18 mm (P < 0.001) from baseline to the final post-operative evaluation. Results of a visual analogue scale (VAS) patient survey indicated that only one patient experienced minor pain on the day of surgery while a different patient noted minor pain at the one and two week post-operative visits. All other patients reported no pain on the day of surgery or at any of the subsequent appointments.15 Additionally, all patients were satisfied with their treatment and said they would undergo the procedure again if indicated and would recommend the treatment to a friend.17 The results of the VAS survey highlighted one of the most important advantages to MIS, namely that patients generally experience minimal to no discomfort and have a high degree of satisfaction with the procedure.
Summary
MIS may result in reductions in PD and gains in CAL similar to conventional flap surgery, such as GTR, with less postoperative recession when used for periodontal regeneration. The emergence of VMIS has improved visual access to surgical sites and has resulted in an actual gain in soft tissue height up to 3 years postoperatively. Further, based on satisfaction surveys, patients report minimal to no postoperative pain and a high degree of satisfaction with MIS procedures. Thus, while further research into the technique as part of a randomized controlled trial is needed, MIS has proved capable of providing clinically and statistically significant improvements in PD, CAL, and soft-tissue height when used in periodontal regenerative procedures.
About the Authors
John C. Tunnell, DDS, MS
Texas A&M University College of Dentistry
Department of Periodontics
Private Practice
Dallas, Texas
Stephen K. Harrel, DDS, MS
Texas A&M University College of Dentistry
Department of Periodontics
References
1. Garrett S. Periodontal regeneration around natural teeth. Ann Periodontol. 1996;1(1):621-666.
2. Kao RT, Nares S, Reynolds MA. Periodontal regeneration - intrabony defects: a systematic review from the AAP Regeneration Workshop. J Periodontol. 2015;86(2 Suppl):S77-S104.
3. Harrel SK, Rees TD. Granulation tissue removal in routine and minimally invasive procedures. Compend Contin Educ Dent. 1995;16(9):960-964.
4. Harrel SK, Nunn ME, Belling CM. Long-term results of a minimally invasive surgical approach for bone grafting. J Periodontol. 1999;70(12):1558-1563.
5. Tibbetts LS, Shanelec D. Periodontal microsurgery. Dent Clin North Am. 1998;42(2):339-359.
6. Cortellini P, Tonetti MS. A minimally invasive surgical technique with an enamel matrix derivative in the regenerative treatment of intra-bony defects: a novel approach to limit morbidity. J Clin Periodontol. 2007;34(1):87-93.
7. Cortellini P, Tonetti MS. Improved wound stability with a modified minimally invasive surgical technique in the regenerative treatment of isolated interdental intrabony defects. J Clin Periodontol. 2009;36(2):157-163.
8. Harrel SK, Wilson TG, Nunn ME. Prospective assessment of the use of enamel matrix proteins with minimally invasive surgery. J Periodontol. 2005;76(3):380-384.
9. Harrel SK, Wilson TG Jr, Nunn ME. Prospective assessment of the use of enamel matrix derivative with minimally invasive surgery: 6-year results. J Periodontol. 2010;81(3):435-441.
10. Harrel SK, Wilson TG Jr, Rivera-Hidalgo F. A videoscope for use in minimally invasive periodontal surgery. J Clin Periodontol. 2013;40(9):868-874.
11. Cortellini P. Minimally invasive surgical techniques in periodontal regeneration. J Evid Based Dent Pract. 2012;12(3 Suppl):89-100.
12. Harrel SK. A minimally invasive surgical approach for periodontal bone grafting. Int J Periodontics Restorative Dent. 1998;18(2):161-169.
13. Harrel SK. A minimally invasive surgical approach for periodontal regeneration: surgical technique and observations. J Periodontol. 1999;70(12):1547-1557.
14. Sculean A, Kiss A, Miliauskaite A, et al. Ten-year results following treatment of intra-bony defects with enamel matrix proteins and guided tissue regeneration. J Clin Periodontol. 2008;35(9):817-824.
15. Harrel SK, Nunn ME, Abraham CM, et al. Videoscope assisted minimally invasive periodontal surgery (VMIS): long term outcomes. J Periodontol. 2017; DOI: 10.1902/jop.2017.160705.
16. Harrel SK, Abraham CM, Rivera-Hidalgo F. Tissue resistance to soft tissue emphysema during minimally invasive periodontal surgery. J Contemp Dent Pract. 2012;13(6):886-891.
17. Harrel SK, Abraham CM, Rivera-Hidalgo F, et al. Videoscope-assisted minimally invasive periodontal surgery: one-year outcome and patient morbidity. Int J Periodontics Restorative Dent. 2016;36(3):363-371.