Treatment of an Unusual Non-Tooth-Related Enamel Pearl (EP) and 3 Teeth-Related EPs with Localized Periodontal Disease Without Teeth Extractions: A Case Report
Simón Pardiñas López, DDS, MS; Roger N. Warren, DDS; Timothy G. Bromage, PhD; Ioana Chesnoiu Matei, DDS, MSc; and Ismael Khouly, DDS, MS, PhD
Abstract: Enamel may be found ectopically as enamel pearls (EPs), which are frequently associated with advanced localized periodontal (LP) destruction. This study presents a case in which an unusual non–tooth-related enamel pearl and three teeth-related enamel pearls with LP disease were found and treated without teeth extractions. A 47-year-old female patient presented at the New York University College of Dentistry with four EPs, three of which were associated with periodontal pockets and/or bleeding on probing (BOP), and one of them not related to any tooth. Periodontal therapy included scaling and root planing and open flap debridement with removal of the EPs. Two pearls were histologically analyzed by polarizing microscopy and scanning electron microscope (SEM). Clinical periodontal parameters, including probing depth and BOP, were measured. All enamel found in the pearls had the same general morphologic appearance when examined by a SEM. The non–tooth-related pearl could be classified as Rodriguez Ponte “adamantinóidea” pearl. Probing depths at 3 months and 9 months after EP removal diminished considerably. Only after the treatment was there no BOP. Before treatment it was in some areas, as shown in Table 1. The LP related to the EP was resolved. The authors conclude that early recognition of enamel pearls is important in the prevention of periodontal destruction, and removal of EPs by a surgical approach as an adjunct to mechanical periodontal treatment resulted in resolution of the LP. The authors state that this is the first time an “adamantinóideas” pearl is being reported on clinically in the literature.
Periodontal diseases are a family of maladies that affect dental supporting tissues. They are often caused by periodontal pathogens, which lead to loss of supporting tissues around the teeth.1,2 Susceptibility to these diseases is highly variable and depends on host responses to periodontal pathogens. Although bacterial plaque is the primary etiologic factor,3,4 progression and clinical characteristics of these diseases are influenced by acquired, local, systemic, and genetic factors that can modify susceptibility to infection.2,5
It is also well established that variations in tooth morphology and local anatomy can predispose an isolated area to inflammation by providing a niche for periodontopathic bacteria.6 Enamel, which is normally restricted to the anatomic crowns of teeth, may be found ectopically on the root, either as enamel pearls (EPs) or as cervical enamel projections.7 EPs are a developmental anomaly of teeth, also called enamelomas, enamel droplets, enamel globules, enamel nodules, enamel knots, and enamel exostoses.8 They are frequently associated with advanced localized periodontal destruction.9 A contributing factor to this localized periodontal destruction is suggested to be the EP size and its topographic relation with the furcation.7,10 Some studies have suggested that the enamel covering of the pearl would preclude a connective tissue attachment, allowing only a hemidesmosomal attachment in the region of the pearl, which is less resistant to periodontal breakdown by bacterial plaque.11,12 Its inaccessibility for proper oral hygiene and its proximity to the furcation can predispose the region to further progression of periodontal disease.11 Once the pearls become exposed to the pocket environment, they provide a larger area for bacterial attachment and also a barrier from mechanical plaque control.13
There is limited literature referring to the treatment of localized periodontitis (LP) related to the presence of an EP without extracting the tooth involved. Only two studies reported clinical benefit with the use of surgical therapy to eliminate the EP in combination with scaling and root planing (SRP), ostectomy, and/or osteoplasty.11,13 In both studies, LP was resolved successfully and patients maintained oral health, comfort, and function. However, in most cases, the EPs were a major cause of the localized severe periodontitis favoring the extraction of the teeth involved.7,12 The aim of this study is to present a case in which four EPs related to localized periodontal disease were found and treated without teeth extractions in three maxillary molars in different locations. Unusual finding of an “adamantinóideas” pearl was also histologically analyzed.
Case Report Patient Evaluation and Treatment
The data and case in this study were obtained from the records of a patient who underwent periodontal treatment in the Department of Periodontology and Implant Dentistry at the New York University College of Dentistry (NYUCD).
A 47-year-old female patient was referred for evaluation and treatment of inflammation and attachment loss around teeth Nos. 2, 7 through 10, 14, and 15. The patient also complained about food impaction and soreness between teeth Nos. 14 and 15. The patient’s general health history was non-contributory, and she reported no history of smoking.
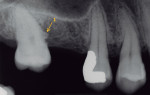
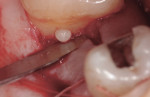
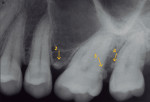
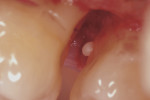
A complete periodontal evaluation was performed. Probing depths (PD), bleeding on probing (BOP), and clinical attachment loss (cemento-enamel junction to gingival margin [CEJ/GM]) were recorded (Table 1). Probing depth measurements of greater than 3 mm and/or bleeding were considered to be pathologic.14 The radiographic findings and intraoral photographs revealed the presence of four EPs: one pearl was on the mesial aspect of tooth No. 2 (pearl No. 1) (Figure 1 and Figure 2); one was on the alveolar bone in the interproximal area between teeth Nos. 13 and 14 (pearl No. 2) (Figure 3); one was on the distal of tooth No. 14 (pearl No. 3) (Figure 3 and Figure 4); and one was on the mesial of tooth No. 15 (pearl No. 4) (Figure 3 and Figure 5). Pearl No. 3 was also visible and accessible clinically. The teeth exhibited no mobility or caries, and pulp tests showed values within normal limits.
Informed consent was obtained and initial periodontal therapy consisting of a complete debridement with SRP under local anesthesia (2% lidocaine with 1:100,000 epinephrine) was performed for all upper teeth in order to reduce inflammation and pain. The patient was instructed to use a mouth rinse (0.12% chlorhexidine gluconate [Peridex™, 3M ESPE, www.3MESPE.com]) twice daily for 10 days. In addition, ibuprofen (600 mg three times a day for 3 days) was prescribed for pain relief and as an anti-inflammatory. The patient returned to the clinic 10 days later for postoperative site evaluation.
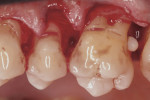
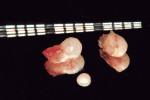
Nine weeks after initial therapy, an open flap debridement (OFD) under local anesthesia (2% lidocaine with 1:100,000 epinephrine) of the upper left molar area was performed following a protocol used in a previous study.15 During this procedure, three pearls were removed successfully with no complications. Pearl No. 2 (3 mm x 2 mm in size) was removed using college pliers (Figure 6). Pearl No. 3 (2 mm x 2 mm in size) (Figure 4 and Figure 6) and pearl No. 4 (1 mm x 1 mm in size) (Figure 5 and Figure 6) were removed with a rongeur. Calculus was detected apical to pearl No. 3 and removed. SRP was performed to eliminate all calculus after removing the pearls. Primary closure of the flap was attained using VICRYL™ 4.0 (Ethicon, www.ethicon.com), and hemostasis was achieved. Pearls Nos. 2 and 3 were sent for histologic analysis. The patient was given postoperative instructions, which included using chlorhexidine gluconate 0.12% mouth rinse twice daily for 10 days until brushing could be resumed. In addition, ibuprofen (600 mg three times a day for 3 days) was prescribed.
Two weeks after the OFD of the upper left molar area, an OFD of the upper right molar area was performed, and pearl No. 1 (1.5 mm x 1 mm) was removed with college pliers from the mesial aspect of tooth No. 2 (Figure 1). Mouth rinses (chlorhexidine gluconate 0.12% mouth rinse twice daily for 10 days) and ibuprofen (600 mg three times a day for 3 days) were prescribed. The patient returned to the clinic 10 days after surgery for suture removal and a postoperative site evaluation.
During follow-up visits at 3 months and 9 months after initial OFD, clinical periodontal parameters (PD, BOP, and CEJ/GM) were recorded (Table 1).
Histologic Analysis
Only the pearls found in the periodontium between teeth Nos. 13 and 14 (pearl No. 2) and on the distal of No. 14 (pearl No. 3) were used for histologic analysis. Specimens were subjected to graded ethanol substitution (70%, 85%, 95%, and 100%) every 12 hours, which was then replaced with 100% methyl salicylate solution (2x) every 8 hours. This was followed by infiltrations of polymethyl methacrylate (PMMA) (Fisher Scientific, www.fishersci.com) each day for 3 days, followed by a final PMMA embedding medium. The embedding medium was cured at 37°C for 3 days, followed by exposure to ultraviolet light for 3 more days. A low-speed saw (IsoMet®, Buehler LTD, www.buehler.com) was used to vertically section the pearls in a buccolingual plane through their centers. The sample blocks were mounted on plexiglass slides and cut off from the slide, leaving a 150-mm-thick section, which was then ground and polished to a thickness of 50 mm to 60 mm with hand-polishing instruments to a 1-mm surface finish. The slides were evaluated unstained in a polarized light microscopy (Leica DM5000, Leica Microsystems Inc, www.leica.com).
Specimen Preparation for Scanning Electron Microscopy
Two histologic thin sections examined by polarizing microscopy were also examined by backscattered electron imaging in the scanning electron microscope (BSE-SEM). The sections were etched for 5 seconds with 37% phosphoric acid and rinsed with running distilled water for 15 seconds. They were air-dried and imaged uncoated by BSE-SEM in a Zeiss EVO 50 variable-pressure SEM (Zeiss, www.zeiss.com) at 8.5-mm working distance, 15kV accelerating voltage, 230 pA to 600 pA current, and 30 Pa to 50 Pa pressure.
Results
All enamel found in the pearls had the same general morphological appearance when examined in a SEM. Only minor differences were found between the specimens.
Pearl No. 2
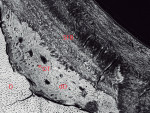
Pearl No. 2, which was found in the periodontium between teeth Nos. 13 and 14, illustrated a crescent-shaped exposure of enamel in cross-section at left, dentin at center, and a mineralized matrix surrounding the dentin and small portions of the “cervical” enamel (Figure 7). The enamel structure was normal, exhibiting prisms and daily cross-striations indicative of daily secretion rates of about 4 mm. Interestingly, overlying the prismatic enamel, there was a substantial layering of aprismatic enamel increasing in width toward the right, each daily layer of enamel being uniformly laid down over one another (Figure 7). At the center of the dentin, where the presumptive pulp cavity would normally exist, secondary dysplastic dentin was observed (Figure 7 and Figure 8). The cervical enamel was covered by a very thin layer of presumed cementum, over which was a layer of Sharpey’s fiber bone (SFB), followed by a very thin layer of enamel, a moderately thick layer of cementum, and, finally, a thick layer of SFB. Toward the right side, adhering to the SFB that had cracked away from the thick cementum layer, a very thin layer of enamel was found. The surface contour was furrowed on one side of the pearl dentin. The dentin surface facing a dysplastic dentin showed evidence of clastic activity, indicating that this furrow was formed by cellular-based removal of the mineralized matrix. The secondary matrix that had filled in this furrow appears to be dysplastic dentin, containing some odontoblast tubules. Overlying this secondary tissue, SFB with layers of various fiber directions was found. It is apparent that the dysplastic dentin was well integrated to the SFB (Figure 8). At the junction of dentin, dysplastic dentin, and SFB, a small loop of enamel laid down was encapsulated around and within by SFB.
Pearl No. 3
The enamel structure and dentin of pearl No. 3 were normal (Figure 9), and the enamel exhibited prisms and daily cross-striations (Figure 10). Higher magnification showed thick aprismatic enamel and dentin at the center of this pearl specimen.
Periodontal Evaluation
Periodontal destruction after 3 months and 9 months post EP removal diminished considerably (Table 1). No BOP was present in any of the areas. LP related to the EP was resolved after EP removal, SRP, and surgical treatment.
Discussion
Anatomical factors, including EP, can predispose a particular site to periodontitis, increasing plaque retention and shielding organisms from the action of salivary enzymes and oral hygiene measures.12 Previous studies reported that EPs were associated with LP.7,10,11 An early diagnosis of an EP may aid in the selection of an appropriate treatment to prevent progression of the periodontal breakdown.10,16 Different treatment therapies have been reported, including odontoplasty, tunneling, root separation, resection, intentional replantation, or extraction.17-19
The present case report showed that the EPs were related to LP, which was successfully treated by EP removal and a combination of SRP and surgical treatment. Different studies showed a successful outcome of the LP related to EPs with similar techniques.11,13 The EP No. 3 on the distal aspect of tooth No. 14 became a reservoir for plaque and calculus as well as food impaction. This pearl was exposed to the oral environment, and proper oral hygiene in this area was very difficult for the patient to maintain. The aim of the therapy was to surgically eliminate the EP to facilitate good plaque control by the patient.12
At 3 months and 9 months after periodontal treatment, which included SRP and OFD on teeth Nos. 2, 14, and 15, the results were stable and favorable. There was neither PD of greater than 3 mm nor was BOP observed. It must be noted that due to the presence of EPs, the PD measurements were not completely reliable as they interfered physically with the probe measurements preoperatively.
The histomorphologic appearance of some areas in which dysplastic enamel and dentin are seen in the polarized light microscopy was previously observed.9,20 These studies reported the presence of prisms and interprisms, which were also found in the present study. Cross-striations of prisms seen in the enamel structure in the present case report showed a periodicity of 4 mm, which is in contrast with other studies that showed a periodicity of 3.2 mm and 3 mm, respectively.7,20 Sharpey’s fibers, which appear in proximity to the cementum of pearl No. 2, were also present in the cementum in another study.9
There is limited literature related to the prevalence of EPs. In two studies on extracted teeth,10,17 it was found that the incidence of EPs was between 0.82% to 2.28%, more often in third molars, second molars, and first molars—65.9%, 15.2%, and 11%, respectively. The prevalence of EPs on maxillary molars was more than four times greater than on mandibular molars, and EPs were found most frequently on the mesial and distal aspects. The mean pearl diameter measured was 0.96 mm to 1.9 mm, and the mean distance to the cemento-enamel junction was 2.8 mm.17,21 In the present case report, three of the four EPs were related to first or second molars, and the mean pearl diameter was 1.5 mm.
Genetic factors do not play a decisive role in enamel pearl formation.9 In 1965, Cavanha22 proposed a classification based on the histological content and localization of the EP on the tooth. Microscopically, the EP can be classified as: true enamel pearls; enamel dentin pearls; enamel dentin pearls with pulp; Rodriguez Ponte’s “adamantinóideas” pearls (which are small enamel nodules found in the periodontium), and intradental enamel pearls included in the dentin.
Three of the pearls in this case report, EP Nos. 1, 3, and 4, classified as enamel-dentin pearls and were directly related to teeth Nos. 2, 14, and 15, respectively.22 The other pearl, EP No. 2, which was located in the interproximal between teeth Nos. 13 and 14, with no signs of LP and unrelated to any tooth, could be classified as Rodriguez Ponte’s adamantinóideas pearls,22 although the present authors found only one reference in the literature related to this type of EP. The reason for this may be explained by the location of the EP, which was not in direct contact to the sulcus. This EP was located in the periodontium over the alveolar bone completely covered by the keratinized attached gingiva, and was therefore protected from the oral environment. This pearl appearance may be explained by the fact that the embryogenesis of pearls is similar to supernumerary tubercules or cusps, and so the pearl’s presence is related to something that happened prior to the initiation of dentin formation.23
Conclusions
This is the first time that a non–tooth-related pearl located in the periodontium is being reported clinically in the literature. A surgical approach to removing enamel pearls as an adjunct to mechanical periodontal treatment resulted in resolution of the localized periodontitis. These results were maintained and stable for 9 months after surgical treatment. Further long-term studies and more cases, however, are needed to confirm the trends that are reflected in this case report, as well as the predictability of treating enamel pearls surgically.
Acknowlegments
The authors thank Dr. Pablo Garcia Canas for his help in conducting this research. Research support was provided by the 2010 Max Planck Research Award (awarded to TGB), administered by the Max Planck Society and the Alexander von Humboldt Foundation in respect of the Hard Tissue Research Program in Human Paleobiomics, and by the Fundación de Salud y Evolución Humana, Madrid, Spain.
About the Authors
Simón Pardiñas López, DDS, MS Former Resident, Department of Periodontology and Implant Dentistry, New York University College of Dentistry, New York, New York; Private Practice, Clínica Pardiñas, Coruña, Spain
Roger N. Warren, DDS Clinical Professor, Department of Periodontology and Implant Dentistry, New York University College of Dentistry, New York, New York
Timothy G. Bromage, PhD Professor, Hard Tissue Research Unit, Department of Biomaterials and Biomimetics and Department of Basic Science and Craniofacial Biology, New York University College of Dentistry, New York, New York
Ioana Chesnoiu Matei, DDS, MSc ITI (International Team for Implantology) Scholar, Department of Oral Surgery and Stomatology, University of Bern School of Dental Medicine, Bern, Switzerland
Ismael Khouly, DDS, MS, PhD Assistant Clinical Professor, Department of Oral and Maxillofacial Surgery and Bluestone Center for Clinical Research, New York University College of Dentistry, New York, New York
References
1. Marchetti E, Monaco A, Procaccini L, et al. Periodontal disease: the influence of metabolic syndrome. Nutr Metab (Lond). 2012;9(1):88.
2. The pathogenesis of periodontal diseases. J Periodontol. 1999;70(4):457-470.
3. Loe H, Theilade E, Jensen SB. Experimental gingivitis in man. J Periodontol. 1965;36:177-187.
4. Lindhe, J, Hamp SE, Loe H. Experimental periodontitis in the beagle dog. Int Dent J. 1973;23(3):432-437.
5. Armitage GC. Development of a classification system for periodontal diseases and conditions. Ann Periodontol. 1999;4(1):1-6.
6. Listergarten M. Influence of anatomical structures on the development of periodontal disease. J Can Dent Assoc. 1959;25:494.
7. Risnes S, Segura JJ, Casado A, Jimenez-Rubio A. Enamel pearls and cervical enamel projections on 2 maxillary molars with localized periodontal disease: case report and histologic study. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;89(4):493-497.
8. Moskow BS, Canut PM. Studies on root enamel (2). Enamel pearls. A review of their morphology, localization, nomenclature, occurrence, classification, histogenesis and incidence. J Clin Periodontol. 1990;17 (5):275-281.
9. Gaspersic D. Enamel microhardness and histological features of composite enamel pearls of different size. J Oral Pathol Med. 1995;24(4):153-158.
10. Chrcanovic BR, Abreu MH, Custódio AL. Prevalence of enamel pearls in teeth from a human teeth bank. J Oral Sci. 2010;52(2):257-260.
11. Goldstein AR. Enamel pearls as a contributing factor in periodontal breakdown. J Am Dent Assoc. 1979;99(2):210-211.
12. Romeo U, Palaia G, Botti R, et al. Enamel pearls as a predisposing factor to localized periodontitis. Quintessence Int. 2011;42(1):69-71.
13. Skinner MA, Shiloah J. The role of enamel pearls in localized severe periodontitis. Quintessence Int. 1989;20(3):181-183.
14. Tanner AC, Kent R Jr, Van Dyke T, et al. Clinical and other risk indicators for early periodontitis in adults. J Periodontol. 2005;76(4):573-581.
15. Froum SJ, Weinberg MA, Rosenberg E, Tarnow D. A comparative study utilizing open flap debridement with and without enamel matrix derivative in the treatment of periodontal intrabony defects: a 12-month reentry study. J Periodontol. 2001;72(1):25-34.
16. Versiani MA, Cristescu RC, Saquy PC, et al. Enamel pearls in permanent dentition: case report and micro-CT evaluation. Dentomaxillofac Radiol. 2013;42(6):20120332.
17. Risnes S. The prevalence, location, and size of enamel pearls on human molars. Scand J Dent Res. 1974;82(6):403-412.
18. Lin HJ, Chan CP, Yang CY, et al. Cemental tear: clinical characteristics and its predisposing factors. J Endod. 2011;37(5):611-618.
19. Darwazeh A, Hamasha AA. Radiographic evidence of enamel pearls in jordanian dental patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;89(2):255-258.
20. Risnes S. Ectopic tooth enamel. An SEM study of the structure of enamel in enamel pearls. Adv Dent Res. 1989;3(2):258-264.
21. Loh, HS. A local study on enamel pearls. Singapore Dent J. 1980;5(1): 55-59.
22. Cavanha AO. Enamel pearls. Oral Surg Oral Med Oral Pathol. 1965;19: 373-382.
23. Malassez L, Gallipe V. Notes on “enamel pearls.” Abstract. The Dental Cosmos. 1908;49:1286.



![Figure 7 Fig 7. Histologic section of EP No. 2 imaged by polarizing microscopy, which illustrates a crescent-shaped exposure of enamel in cross-section at left (E), dentin at center (D), and a mineralized matrix (Sharpey’s fiber bone [SFB]) surrounding the dentin and small portions of the “cervical” enamel. Field width = 3.8 mm.](/media/thumbnail/48694/150x150)
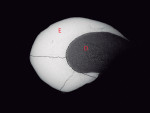
![Figure 10 Fig 10.Electron microscopy of a histologic section of EP No. 3. The enamel structure is normal, exhibiting prisms (coursing from upper right to lower left) and daily cross striations (appearing as dark lines that cross the diameters of prisms [see arrows]). Note also the aprismatic enamel (AE) containing daily incremental lines (arrows). Field width = 100 μm.](/media/thumbnail/48692/150x150)


