Effect of an Herbal Mouth Rinse in Preventing
Periodontal Inflammation in an Experimental Gingivitis Model: A Pilot Study
Noah Samuels, MD; John T. Grbic, DMD, MMSc; Aron J. Saffer, DDS, MS; Isaiah D. Wexler, MD, PhD; and Ray C. Williams, DMD
Abstract
BACKGROUND: Gingivitis is a chronic inflammatory condition, resulting from gingival bacteria and bacterial byproducts. Antiplaque oral rinses reduce inflammation by removing or inhibiting plaque formation. The purpose of this pilot study was to examine the anti-inflammatory effects of HM-302, a mouth rinse based on natural products, on gingival inflammation. METHODS: A prospective, double-blinded, randomized parallel-group controlled trial involving 62 patients was conducted to assess efficacy and safety. During a 2-week period with no dental hygiene, subjects were randomized to receive either the study rinse (HM-302); a cetylpyridinium chloride (CPC) rinse; an essential oils (EO) rinse; or a water-only preparation. The gingival index (GI), plaque index (PI), and number of bleeding sites were measured at baseline and at the end of the study period. RESULTS: Progression of gingival inflammation resulting from lack of dental hygiene was lowest in patients treated with the HM-302 rinse, and was significantly less marked than in patients treated with the water-only preparation. When compared to the CPC and EO treatments, HM-302 was the only mouth rinse that was significantly better than the control, with respect to both the change in absolute GI scores (p = .006) and to the percent increase in GI scores (p = .012). No serious adverse effects were noted in any of the study groups. CONCLUSION: HM-302 is a safe and effective treatment for preventing the development of gingival inflammation in an experimental gingivitis model. Further research is needed to evaluate its long-term effects.
Gingivitis and periodontal inflammation are common diseases, prevalent in 50% to 90% of the general population worldwide, with more than 80% of those over the age of 65 living in the United States suffering from periodontal disease.1,2 Though considered a physiologic inflammatory response to plaque accumulation, gingivitis is the starting point for more serious and destructive periodontal disease.3,4
Gingivitis and periodontitis have also been linked to systemic inflammatory conditions such as atherosclerosis and macrovascular complications associated with diabetes mellitus.5-7 It is these systemic effects that have led to an increased understanding that finding an effective treatment for chronic gingival inflammatory disease is vital not only for preventing tooth loss but also as part of overall healthcare. As a result, increased efforts are being directed at the prevention and treatment of the inflammatory processes that are associated with these conditions.8
The etiology of gingivitis is multifactorial, with both host and environmental factors contributing to its pathogenesis.3,4,8-10 Bacteria and bacterial byproducts of periodontal pathogens form a biofilm above and below the gingival margin, stimulating a chronic inflammatory response, with decreased tissue repair leading to bone destruction.7-12 Current treatment of this condition consists of mechanical removal of plaque (eg, toothbrushing, flossing, and dental scaling and root planing procedures),13 topical and systemic antimicrobial therapy,14 and chemical removal or inhibition of plaque formation through the use of oral rinses.15
The underlying rationale for the use of antiplaque oral rinses is the understanding that plaque removal or inhibition will reduce inflammation.16,17 Many of the products use a cetylpyridinium chloride (CPC)-based formulation, because CPC is safe and has demonstrated significant plaque inhibition, which is attributed to its chemical and antimicrobial properties.18-21 Oral rinses are particularly useful for patients with severely inflamed gums who may not adequately brush because of the associated pain and discomfort, or for patients with physical and/or cognitive disabilities. The efficacy of oral rinses is based on their impact on preventing plaque development as well as reducing inflammation.15
A mouth rinse (HM-302, Izun Pharmaceuticals Corp., www.izunpharma.com) containing the herbs Centella asiatica, Echinacea purpurea, and Sambucus nigra was developed for the purpose of treating gingivitis. The herbs are prepared according to guidelines of the homeopathic pharmacopeia of the United States (HPUS) and authorized for sale by the Israeli health ministry. Clinical experience of the authors has shown this rinse to be effective in reducing gingival inflammation. The present study set out to investigate the safety and efficacy of HM-302 using a well-established experimental gingivitis model, as developed by Löe and colleagues.22 In this pilot clinical trial, each of the study rinses was compared to a water-based rinse vis-à-vis the effects on gingival inflammation and plaque. The primary outcome measured was the gingival index (GI), which is a measure of gingival inflammation. Other parameters assessed were the plaque index (PI) and number of bleeding sites.
Materials and Methods
Subject Population
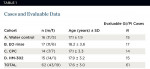
This study was designed as a prospective, interventional, double-blinded, randomized, placebo-controlled trial. Eligible subjects were paid volunteers attending a dental clinic located in Jerusalem, Israel. Subjects between the ages of 14 to 75 years with a clinical diagnosis of gingivitis were eligible for inclusion. Individuals with chronic illnesses (eg, diabetes, heart disease, chronic inflammatory disorders), those using anti-inflammatory or antibiotic medication, pregnant women, and smokers were excluded, as were those with less than 24 teeth or severe periodontitis (periodontal pockets > 5 mm in > three areas). After receiving an in-depth explanation of the study proceedings and possible adverse events, candidates were asked to sign a written informed consent form. The study protocol was approved by the Institutional Review Board at the Shaare Zedek Medical Center in Jerusalem, and was registered at ClinicalTrials.gov (NCT00885599).
Study Design
The study design was based on the experimental model developed by Löe et al, which requires participants to refrain from any type of oral hygiene for a pre-established period of time.22 Recruited subjects were randomly allocated to one of four groups, using the Moses-Oakford algorithm, as well as a clinical evaluation of the severity of gingival inflammation. The method for determining gingival inflammation and the extent of plaque was in accordance with that described by Löe and Sillness.23 Severity of gingival inflammation was evaluated using the GI as the primary outcome measure. The PI, which is a scoring system that characterizes the extent of dental plaque, was also utilized. The number of bleeding sites per patient was determined by assessment of bleeding-upon-probing (BoP). Following initial evaluation, all subjects underwent a full dental prophylaxis treatment, which included both scaling and tooth polishing.24
Following initial assessment, all subjects were instructed to abstain from brushing their teeth or using any other oral hygiene technique (eg, flossing, using commercial mouth rinses or breath mints, etc). The only treatment allowed was washing with 15 mL of one of the study mouth rinse products, which was to be used three times daily for 14 days. Patients were given bottles containing 250 mL of the test rinse product and bottles contained a 15 mL dose cap, which patients were asked to fill. On day 14, patients were re-examined, and GI, PI, and BoP were evaluated. Subjects were then given full dental prophylaxis and instructed to return to their original oral hygiene regimen.
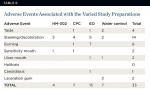
Throughout the study period, subjects were instructed to keep a log record in which they recorded the times that they took the mouth rinse. In order to assess adherence to the study plan, subjects were instructed to return the bottles of mouth rinse each week so that investigators could determine whether the volume of mouth rinse used corresponded with the declared use. Study participants were also instructed to record any adverse event that occurred during the study period. Adverse events were graded on a three-point scale (mild, moderate, serious) and reported to the study monitor.
Study Medications
HM-302 contains a proprietary mixture of extracts from the following herbs: Centella asiatica, Echinacea purpurea, and Sambucus nigra. The exact mix of herbs was selected after extensive pre-clinical testing, and HM-302 was chosen as the test drug after it was determined that this particular mixture of the three herbal extracts had the best anti-inflammatory profile of all the candidates tested.
The ingredients in HM-302 were formulated in an alcohol-free solution containing flavorants and a preservative. The other two test mouth rinses included CPC in a water base with flavorants and preservatives, and a blend of essential oils (EO) in an alcohol base (Listerine®, Johnson & Johnson, www.listerine.com). A water-based rinse that was color- and flavor-coordinated containing a preservative was used as a negative control.
Observer Reliability
To standardize measurements and minimize inter-observer variability, recommendations listed in the draft Guidance for Industry protocol publicized by the FDA were followed.25 Two licensed and practicing dentists who served as examiners were calibrated by one of the study coordinators (AJS). Calibration consisted of training the examiners prior to the study; pre-testing them on 20 patients, verifying that they assigned identical scores (designed as inter-observer variation of ≤ 10%); and monitoring during the study period, with periodic retraining by the study coordinator. In order to accurately compare final to baseline parameter outcomes, each participant was followed by the same examiner.
Statistical Analysis
This was a pilot study with no prior clinical data from which to estimate an effect for the purpose of sample size calculation. Based on the authors’ clinical experience, a 25% decrease in GI between the test drug and control cohort was chosen a priori as a clinically significant effect. Comparing the means of two independent samples using a t-test with a power of 80% at a significance level of < 0.05 (estimated standard deviation [SD] of .25 of the untreated cohort expected mean GI), the authors estimated that 16 patients would be needed per group. A target recruitment goal of 16 to 18 patients per group was established.
The outcome measures, including GI and PI, were calculated as the absolute difference (Δ = final - baseline mean GI or PI), with increases in GI or PI denoted as positive values. Hence, a positive difference meant worsening clinical disease or increased inflammation. The differences were also calculated as percent change to normal for baseline differences in these parameters. Data met the conditions for normalcy as determined by Goodness-of-Fit Test (Kolmogorov-Smirnov test). The paired t-test was used to determine whether there were significant differences between mean baseline and final values. The t-test assuming equal variances was used to compare the results of a specific arm with the water control. Significance was assessed using a two-tail t-test. To assess the effect of baseline values on the outcome variables, an analysis of covariance (ANCOVA) was performed using a general linearized model, with treatment as the main effect and baseline values as the covariate. Bleeding site data was analyzed as either the difference between baseline and final examinations, and response rate was defined as the number of patients with a reduction in the mean number of bleeding sites at the end of the trial. A two-tailed t-test was used for evaluating the difference in mean number of bleeding sites, and the χ2 statistic was used to evaluate response rate. Data were entered into a Microsoft Excel 2003 worksheet and transferred to SPSS version 16 (SPSS, Inc., www.spss.com) for statistical analyses. A p-value of less than or equal to 0.05 was considered statistically significant.
Results
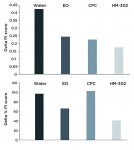
A total of 62 patients (41 male and 21 female) were enrolled in the study, which took place between June 2009 and June 2010. The mean age of the study group was 17.6 years of age (range: 14 to 28 years), with 61 completing the study protocol in its entirety. The number of patients per cohort for the reported data is shown in Table 1. GI and PI values at baseline and at the end of the 14-day study period are shown in Table 2 and Table 3. There was no difference in baseline levels of mean GI values between the cohorts, and only the water-control group displayed a statistically significant increase in GI score. The HM-302 treatment group was the only group that showed a minimal decrease in the GI score at the end of the trial period, indicating that gingival inflammation did not worsen over the 2-week course of the study in this cohort. All other study groups deteriorated with higher mean GI scores at the end of the study period. The HM-302 group was also the only one that was significantly better than the water group in terms of ΔGI and %ΔGI (p = .007 and p = .013, respectively; see Figure 1 and Table 4).
Baseline PI values were not significantly different between the study groups (Figure 2, Table 3), and in all groups there was significant or near-significant deterioration of PI over the 2-week study period. The HM-302 group, however, showed the smallest increase (ie, least severe exacerbation) in PI values, and when compared to the water-treated control group (Table 4) this was close to the level of significance (p = .081). Analysis of covariance of baseline GI scores as the covariate showed that the differences between pre- and posttreatment values continued to remain statistically significant for the HM-302 group when compared to the water-only group even when factoring in for the effect of baseline levels of GI (Table 4). In contrast, ΔPI and %ΔPI values were significantly related to baseline values, and the extent of plaque at the beginning of the study impacted on ΔPI and %ΔPI, which was close to significant (Table 4).
Analysis of gingival bleeding results showed only a minimal change in the number of bleeding sites in the HM-302 group from baseline when compared to the other treatment groups. The response rate for this outcome—defined as the number of patients per cohort with a reduction of bleeding sites at the end of the trial—was in favor of the HM-302 group (Table 5). When compared to the water-only group, the reduced number of bleeding sites in the HM-302 group was significant (p = .035), though the response rate was not. However, both the HM-302 and the EO rinse groups had response rates greater than 50% and an almost twofold improvement when compared to the water-only group, though this did not reach a level of significance (Table 5).
The HM-302 mouthwash was acceptable to all of the subjects in the treatment group, with no serious adverse effects reported. The most commonly reported effect (by 3 of 15 subjects) was a slight staining of the teeth, which was consistent with all other rinses in the trial. This effect did not lead to any of the participants discontinuing its use (Table 6).
Discussion
This small pilot study utilized an experimental gingivitis protocol and showed that there was a significantly lower increase (ie, less deterioration) in mean GI values in patients treated with HM-302 when compared to a water-only control treatment. HM-302 was the only agent in the trial that significantly minimized the increase in GI and was found to be an acceptable treatment as well, with no serious adverse effects.
The use of naturally derived anti-inflammatory agents is becoming more accepted in conventional medicine and dentistry.26 Many of these agents contain a mixture of active pharmaceutical ingredients that are combined based on either their additive or synergistic properties.27 The ideal anti-inflammatory phytotherapeutic agent for periodontal disease would be one that targets pro-inflammatory cytokines and enzymes involved in oral inflammation and tissue destruction.3,4,28 The list of pro-inflammatory cytokines and enzymes involved in periodontitis includes interleukin-1 beta (IL-1β), tumor necrosis factor-alpha (TNF-α), beta-glucuronidase (ß-glu), and metalloproteinases,8-12,28,29 and the three active herbal ingredients of HM-302 have been shown to have an impact on these cytokines.
The first herb, Centella asiatica (gotu kola), is commonly used for wound healing and microangiopathy.30-32 It has been found to increase collagen production and stimulate cellular hyperplasia in granulation tissue due to higher levels of DNA, protein, and hexosamine.33 In studies, the tensile strength of the healing wound was enhanced through greater cross-linking of collagen, as well as a more rapid re-epithelialization process.34,35 Centella asiatica has also been found to reduce activity levels of ß-glu, an enzyme that is often elevated at sites of gingival inflammation.35 Sastravaha et al studied the effects of Centella asiatica, implanted together with the herb Punica granatum as a biodegradable chip in the subgingiva of 15 patients. There was a significant improvement of the GI score with respect to subgingival implants when compared to controls at 3 and 6 months, while PI scores were not different between the groups.36 This herb has demonstrated antibacterial activity in an in vitro model.37
The second herbal component, Sambucus nigra, has been shown to modulate the levels of a number of pro-inflammatory cytokines, inhibiting pro-inflammatory properties of bacteria commonly found in periodontitis and exhibiting antibacterial and antiviral activity.38,39 An aqueous extract of this herb was shown to inhibit the pro-inflammatory properties of two periodontal pathogens, Porphyromonas gingivalis and Actinobacillus actinomycetemcomitans, with regard to cytokine production, oxidative burst induction in neutrophils, and integrin activation. This herb was also found to inhibit the activation of NFқB, a transcription factor that plays a key role in regulating the immune response to infection.38 This finding was significant because the innate immune responses leading to NFқB activation may underlie the link between periodontal inflammation and atherosclerosis.40
The third and final component of HM-302, Echinacea purpurea, is considered to have antiviral, antibacterial, and anti-cytotoxic effects,41,42 though the effects of this herb on gingival inflammation remain unclear. Echinacea purpurea, a popular medicinal herb, is used primarily for upper respiratory tract infections and has been found to inhibit pro-inflammatory interleukins such as interferon-g and TNF-α.41-44
The authors studied HM-302 using an experimental gingivitis protocol, which evaluates the efficacy of the test agent in patients for whom all mechanical tooth cleaning and oral preparations, except for the test agent, are stopped for a 2-week period. The advantage of a short-term experimental gingivitis model is that there is typically better participant adherence, thereby requiring a smaller sample size and allowing for the utilization of outcome parameters such as PI and GI, which are objective and easily measured.
There are a number of limitations to this study. In the study model, the prolonged abstention from brushing and other routine mechanical plaque control methods can lead to the creation of an artificial situation in which there is greater food and materia alba accumulation on the teeth, which in turn accelerates plaque development and gingivitis. In addition, the study population consisted of healthy individuals, a group that may respond differently from those suffering from significant chronic periodontal inflammation. Another potential limitation is that while not statistically different than other treatment, baseline GI scores were highest in the HM-302 group, possibly allowing for greater room for improvement in this group versus the other active and control groups. Finally, the study was not powered to determine the individual contributions of the anti-inflammatory and the antimicrobial components to the enhanced efficacy of HM-302. Nevertheless, the findings of this pilot study are significant and suggest that HM-302 has the potential to mitigate the host inflammatory response to plaque and reduce levels of gingival inflammation.
Conclusion
HM-302, an oral rinse containing herbs with reported antibacterial and anti-inflammatory properties, was shown to reduce gingival inflammation in an experimental gingivitis protocol. The possible synergistic effect of the combined ingredients, with both antibacterial and anti-inflammatory properties, may have contributed to this outcome. Larger-scale studies will be needed to confirm the therapeutic potential of HM-302 as a novel treatment for gingivitis.
Disclosure
The study drug was provided free of charge by Izun Pharmaceuticals Corp., Israel, which also funded the statistical analysis of the data. Dr. Saffer is senior vice president for research and development and Dr. Wexler is a scientific consultant for Izun Pharmaceuticals Corp.
References
1. Pihlstrom BL, Michalowicz BS, Johnson NW. Periodontal diseases. Lancet. 2005;366(9499):1809-1820.
2. Albandar JM. Epidemiology and risk factors of periodontal diseases. Dent Clin North Am. 2005;49(3):517-532.
3. Offenbacher S, Barros SP, Beck JD. Rethinking periodontal inflammation. J Periodontol. 2008;79(8 suppl):1577-1584.
4. Tatakis DN, Kumar PS. Etiology and pathogenesis of periodontal diseases. Dent Clin North Am. 2005;49(3):491-516.
5. Seymour GJ, Ford PJ, Cullinan MP, et al. Relationship between periodontal infections and systemic disease. Clin Microbiol Infect. 2007;13(suppl 4):3-10.
6. Williams RC, Barnett AH, Claffey N, et al. The potential impact of periodontal disease on general health: a consensus view. Curr Med Res Opin. 2008;24(6):1635-1643.
7. Ryan ME, Carnu O, Kamer A. The influence of diabetes on the periodontal tissues. J Am Dent Assoc. 2003;134(spec no):34S-40S.
8. Williams RC, Paquette DW. Understanding the pathogenesis of periodontitis: a century of discovery. J Int Acad Periodontol. 2000;2(3):59-63.
9. Kinane DF, Mark Bartold P. Clinical relevance of the host responses of periodontitis. Periodontol 2000. 2007;(43):278-293.
10. Madianos PN, Bobetsis YA, Kinane DF. Generation of inflammatory stimuli: how bacteria set up inflammatory responses in the gingiva. J Clin Periodontol. 2005;32(suppl 6):57-71.
11. Ebersole JL. Humoral immune responses in gingival crevice fluid: local and systemic implications. Periodontol 2000. 2003;(31):135-166.
12. Potempa J, Banbula A, Travis J. Role of bacterial proteinases in matrix destruction and modulation of host responses. Periodontol 2000. 2000;(24):153-192.
13. Van Dyke TE. The management of inflammation in periodontal disease. J Periodontol. 2008;79(8 suppl):1601-1608.
14. Herrera D, Alonso B, León R, et al. Antimicrobial therapy in periodontitis: the use of systemic antimicrobials against the subgingival biofilm. J Clin Periodontol. 2008;35(8 suppl):45-66.
15. Moran JM. Home-use oral hygiene products: mouthrinses. Periodontol 2000. 2008;(48):42-53.
16. Bhatavadekar NB, Williams RC. New directions in host modulation for the management of periodontal disease. J Clin Periodontol. 2009;36(2):124-126.
17. Salvi GE, Lang NP. The effects of non-steroidal anti-inflammatory drugs (selective and non-selective) on the treatment of periodontal diseases. Curr Pharm Des. 2005;11(14):1757-1769.
18. Llewelyn J. A double-blind crossover trial on the effect of cetylpyridinium chloride 0.05% (Merocet) on plaque accumulation. Br Dent J. 1980:148(4):103-104.
19. Albert-Kiszely A, Pjetursson BE, Salvi GE, et al. Comparison of the effects of cetylpyridinium chloride with an essential oil mouth rinse on dental plaque and gingivitis - a six-month randomized controlled clinical trial. J Clin Periodontol. 2007;34(8):658-667.
20. Haps S, Slot DE, Berchier CE, et al. The effect of cetylpyridinium chloride-containing mouth rinses as adjuncts to toothbrushing on plaque and parameters of gingival inflammation: a systematic review. Int J Dent Hyg. 2008;6(4):290-303.
21. Gunsolley JC. A meta-analysis of six-month studies of antiplaque and antigingivitis agents. J Am Dent Assoc. 2006;137(12):1649-1657.
22. Löe H, Theilade E, Jensen SB. Experimental gingivitis in man. J Periodontol. 1965;(36):177-187.
23. Löe H. The Gingival Index, the Plaque Index and the Retention Index Systems. J Periodontol. 1967;38(6)suppl:610-616.
24. Muhlemann, HR, Son S. Gingival sulcus bleeding—a leading symptom in initial gingivitis. Helv Odontol Acta. 1971;15(2):107-113.
25. Food and Drug Administration. Gingivitis: Development and Evaluation of Drugs for Treatment or Prevention. Rockville, MD: US Dept of Health and Human Services; updated June 28, 2005. Available at: https://www.fda.gov/ohrms/dockets/98fr/05-12764.htm. Accessed April 2011.
26. Groppo FC, Bergamaschi Cde C, Cogo K, et al. Use of phytotherapy in dentistry. Phytother Res. 2008;22(8):993-998.
27. Wagner H, Ulrich-Merzenich G. Synergy research: approaching a new generation of phytopharmaceuticals. Phytomedicine. 2009;16(2-3):97-110.
28. Reddy MS, Geurs NC, Gunsolley JC. Periodontal host modulation with antiproteinase, anti-inflammatory, and bone-sparing agents. A systematic review. Ann Periodontol. 2003;8(1):12-37.
29. Hannas AR, Pereira JC, Granjeiro JM, Tjäderhane L. The role of matrix metalloproteinases in the oral environment. Acta Odontol Scand. 2007;65(1):1-13.
30. Cesarone MR, Incandela L, De Sanctis MT, et al. Evaluation of treatment of diabetic microangiopathy with total triterpenic fraction of Centella asiatica: a clinical prospective randomized trial with a microcirculatory model. Angiology. 2001;52(suppl 2):S49-S54.
31. Punturee K, Wild CP, Kasinrerk W, Vinitketkumnuen U. Immunomodulatory activities of Centella asiatica and Rhinacanthus nasutus extracts. Asian Pac J Cancer Prev. 2005;6(3):396-400.
32. Shukla A, Rasik AM, Dhawan BN. Asiaticoside-induced elevation of antioxidant levels in healing wounds. Phytother Res. 1999;13(1):50-54.
33. Lu L, Ying K, Wei S, et al. Asiaticoside induction for cell-cycle progression, proliferation and collagen synthesis in human dermal fibroblasts. Int J Dermatol. 2004;43(11):801-807.
34. Shetty BS, Udupa SL, Udupa AL, Somayaji SN. Effect of Centella asiatica L (Umbelliferae) on normal and dexamethasone-suppressed wound healing in Wistar Albino rats. Int J Low Extrem Wounds. 2006;5(3):137-143.
35. Arpaia MR, Ferrone R, Amitrano M, et al. Effects of Centella asiatica extract on mucopolysaccharide metabolism in subjects with varicose veins. Int J Clin Pharmacol Res. 1990;10(4):229-233.
36. Sastravaha G, Gassmann G, Sangtherapitikul P, Grimm WD. Adjunctive periodontal treatment with Centella asiatica and Punica granatum extracts in supportive periodontal therapy. J Int Acad Periodontol. 2005;7(3):70-79.
37. Zaidan MR, Noor Rain A, Badrul AR, et al. In vitro screening of five local medicinal plants for antibacterial activity using disc diffusion method. Trop Biomed. 2005;22(2):165-170.
38. Harokopakis E, Albzreh MH, Haase EM, et al. Inhibition of proinflammatory activities of major periodontal pathogens by aqueous extracts from elder flower (Sambucus nigra). J Periodontol. 2006;77(2):271-279.
39. Krawitz C, Mraheil MA, Stein M, et al. Inhibitory activity of a standardized elderberry liquid extract against clinically-relevant human respiratory bacterial pathogens and influenza A and B viruses. BMC Complement Altern Med. 2011;11:16.
40. Hajishengallis G, Sharma A, Russell MW, Genco RJ. Interactions of oral pathogens with toll-like receptors: possible role in atherosclerosis. Ann Periodontol. 2002;7(1):72-78.
41. Woelkart K, Marth E, Suter A, et al. Bioavailability and pharmacokinetics of Echinacea purpurea preparations and their interaction with the immune system. Int J Clin Pharmacol Ther. 2006;44(9):401-408.
42. Sharma SM, Anderson M, Schoop SR, Hudson JB. Bactericidal and anti-inflammatory properties of a standardized Echinacea extract (Echinaforce): dual actions against respiratory bacteria. Phytomedicine. 2010;17(8-9):563-568.
43. Sasagawa M, Cech NB, Gray DE, et al. Echinacea alkylamides inhibit interleukin-2 production by Jurkat T cells. Int Immunopharmacol. 2006;6(7):1214-1221.
44. Mishima S, Saito K, Maruyama H et al. Antioxidant and immuno-enhancing effects of Echinacea purpurea. Biol Pharm Bull. 2004;27(7):1004-1009.
About the Authors
Noah Samuels, MD
Center for Integrative Complementary Medicine
Shaare Zedek Medical Center
Jerusalem, Israel
John T. Grbic, DMD, MMSc
Columbia University College of Dental Medicine
New York, New York
Aron J. Saffer, DDS, MS Jerusalem Perio Center
Jerusalem, Israel
Isaiah D. Wexler, MD, PhD
Department of Pediatrics
Hadassah University Hospital
Jerusalem, Israel
Ray C. Williams, DMD
Dean
Stony Brook University School of Dental Medicine
Stony Brook, New York