A Clinical and Histological Comparison of Two Different Bone Augmentation Materials in the Atrophic Pre-Maxilla
Maria Hernandez, DDS; Gregory A. Pette, DMD, MS; Andre Grenier, DMD; Carlos Villanueva, DDS; Enrique Lask, DDS; and William Parker, DDS
Abstract
This case study was conducted to compare the regenerative potential of bovine xenograft versus demineralized freeze-dried bone allograft in a thermoplastic biologic carrier in regards to achieving regeneration of bone width as well as potential bone height. The objective for the patient was to gain adequate bone volume for the placement of dental implants using the two different forms of graft material—one with a slow resorption time and the other in a gelatin matrix to provide dimensional stability. The results of this case report are inconclusive to demonstrate the superiority of one of the two graft materials over the other in reconstructing a severely atrophied maxilla. Both sites ultimately required a second guided bone regeneration (GBR) procedure prior to implant placement. It was speculated that adequate space maintenance might not have been achieved during the initial procedures.
When patients present with partially edentulous spaces in the pre-maxilla, guided bone regeneration (GBR) is often necessary to achieve an esthetic prosthetic outcome. The ability to regenerate in both the horizontal and vertical dimensions is dependent upon the basic principles of GBR. These main GBR principles are adequate space maintenance, sufficient blood supply, tension-free primary closure, and epithelial cell exclusion.1,2
Several different grafting techniques are available for GBR procedures. These options include the use of any number of available particulate or autogenous grafting materials. The use of particulate graft materials is appealing to many clinicians due to the ability to avoid a secondary surgical site and the ease of availability. Space maintenance remains one of the key components for a successful GBR procedure. Traditionally titanium-reinforced Gore-Tex® (ePTFE) (GORE-TEX®, www.gore-tex.com) membranes and/or stainless-steel tenting screws have been recommended. However, recently the use of titanium mesh in GBR has shown promising results, as the mesh provides excellent space maintenance.3
Tissue regeneration stems from the work of Melcher and Urist.4,5 Melcher discussed the compartmentalization of cells in the periodontium, and Urist discussed ectopic bone growth in muscle pouches created in animals and also introduced growth factors involved in bone regeneration, such as bone-morphogenetic protein-2.4,5 Nearly two decades later, Buser and co-workers showed successful use of autogenous particulate for GBR prior to dental implant placement.1,6 Buser also concluded that the main liability of autogenous grafts was their limited availability and quantity. Simion and co-workers compared GBR when using autogenous bone and a cell-exclusive membrane, allograft particulate with a cell exclusive membrane, synthetic graft materials with a cell exclusive membrane, a cell exclusive membrane only, and a no- treatment control.7 The autogenous graft provided the densest bone and the greatest quantity; however, the allograft was successful in bone regeneration as well. When a particulate is placed into fresh extraction sockets, it is commonly thought that the use of mineralized bone particulate (FDBA) provides far greater ridge regeneration/preservation than the use of demineralized bone particulate (DFDBA).8 This is thought to occur due to the variable amounts of bone-morphogenic proteins that have been found in DFDBA versus FDBA.8
During GBR procedures, it has been shown that there was no statistical difference in the amount of vital bone regenerated when FDBA was compared to DFDBA.9 In another study of graft materials for GBR procedures, DFDBA in a putty carrier was compared to a xenograft particulate for ridge preservation after tooth extraction.10 The xenograft particulate provided slightly greater preservation of the ridge width (0.3 +/- 0.7 vs. 0.7 +/- 1.2mm, P > 0.05). However, the DFDBA provided considerably greater vital bone histologically compared to the xenograft (61 +/- 9% vs. 26 +/- 20%, P < 0.05). While the xenograft particulate was able to provide a slightly greater amount of bone regeneration, the DFDBA produced a better quality of bone regeneration.
In a brief literature review, Chiapasco and co-workers examined various GBR techniques and concluded that it is difficult or impossible to demonstrate that a particular technique offers a better outcome when compared to the others and that every surgical procedure has its own advantages and disadvantages, which should be taken into account.11 Similarly, Esposito and co-workers concluded that priority should be given to those procedures that appear simpler, are less invasive, involve less risk of complications, and reach their goals within the shortest timeframe.12 With this brief literature review in mind, the following case study was conducted to compare the regenerative potential of bovine xenograft versus demineralized freeze-dried bone allograft in a thermoplastic biologic carrier (which aids in both handling and space maintenance during the healing period) in regards to achieving regeneration of bone width as well as potential bone height.
Materials and Methods
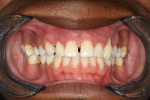
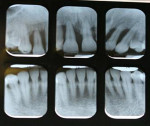
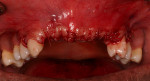
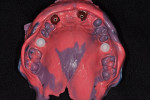
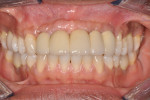
The patient was a healthy 35-year-old African-American male who presented to the Postgraduate Periodontics dental clinic at Nova Southeastern University, College of Dental Medicine, Fort Lauderdale, Florida, in 2005. His chief complaint was that “my front teeth are loose and my gums are always bleeding.” The patient was a nonsmoker and his medical history was noncontributory. Tooth Nos. 3, 7 through 11, 14, 19, and 30 had moderate-to-severe loss of periodontal attachment and migration as seen in Figure 1 and Figure 2. The upper anteriors and first molars also exhibited Miller’s Grade 2 to 3 mobility and bleeding upon probing. The patient was diagnosed with aggressive periodontitis.13 The patient received initial periodontal therapy, and upon periodontal re-evaluation, all first molars and tooth Nos. 7 through 11 were determined to have a hopeless prognosis due to advanced loss of periodontal attachment.
The definitive treatment plan included extraction of the hopeless teeth, which would be replaced by implant-supported prostheses. Single-unit implant-supported crowns were planned in the first-molar sites, while implants were to be placed in the area of Nos. 8 and 11 to support an implant-retained fixed partial denture (FPD) replacing Nos. 7 through 11.
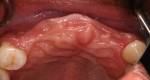
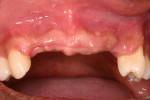
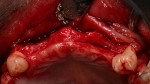
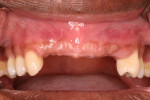
Tooth Nos. 3, 7 through 11, 14, 19, and 30 were extracted, and the patient was provided an interim upper removable partial denture. No bone grafting was done at the time of the extractions. After the area had healed from the extractions, it was determined that there was a ridge deficiency in the buccal-lingual dimension in the maxillary anterior as shown in Figure 3 and Figure 4. Options for treating such a horizontal defect include GBR, tunnel approach graft, ridge split, and block bone graft.1,14,15 Considering the size and shape of the defect, a GBR approach with delayed implant placement was chosen.
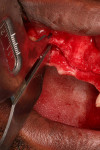
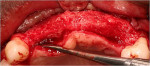
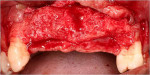
For the GBR procedure, crestal incisions were made on the edentulous ridge favoring the palatal aspect and extended sulcularly around tooth Nos. 6 and 12 with vertical releasing incisions at the distal line angles of tooth Nos. 6 and 12. A full-thickness mucoperiosteal flap was then elevated and the alveolar ridge widths were recorded using a Boley gauge prior to augmentation, as seen in Figure 5 and Figure 6. The average thickness of the residual ridge was approximately 3 mm at both future implant sites. Intra-marrow penetrations were then made on the buccal and crestal aspects of the edentulous ridges using a high-speed handpiece. Stainless steel tenting screws were then placed to provide space maintenance. The bone graft materials and collagen barrier membranes were placed, as seen in Figure 7 through Figure 9. In the area of Nos. 7 and 8, 1 cc of room temperature DFDBA in a gelatin matrix (Regenaform®, Exactech Dental Biologics, www.exac.com) was placed after being reconstituted with sterile saline according to the manufacturer’s directions. In the area of Nos. 9 through 11, 1 gram of bovine xenograft particulate (Bio-Oss®, Osteohealth, www.osteohealth.com) was placed, after being rehydrated in sterile saline. The areas were overbuilt to augment the ridge in a horizontal direction using a surgical guide, which replicated the diagnostic wax-up of the desired final prosthetic outcome. Two resorbable bilayered collagen membranes (Ossix™, OraPharma, Inc., www.orapharma.com) were adapted to cover the graft material extending at least 2 mm beyond the margin of the graft. Periosteal-releasing incisions were made to ensure tension-free primary coverage, and the surgical site was closed with 4-0 Vicryl (Ethicon, www.ethicon.com) resorbable sutures. An interim removable partial denture was relieved to avoid any pressure on the augmented sites. The patient was placed on systemic amoxicillin (500 mg 1 q8h), ibuprofen (400 mg 2 q8h prn pain), and chlorhexidine 0.12% (rinse b.i.d.) for 1 week.
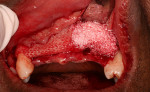
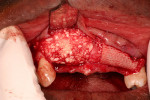
The patient’s postoperative course was uneventful. At 6 months the patient was reevaluated for implant placement using a surgical guide based upon the diagnostic wax-up (Figure 10 and Figure 11). At the surgical re-entry, measurements of the alveolar ridge were again made using a Boley gauge and recorded. Observations of the shape, consistency, and appearance of the two sites were made (Figure 12 and Figure 13). It was determined that even though significant osseous augmentation had occurred, additional augmentation in the buccal aspect would be ideal prior to implant placement. At this point, a biopsy was taken from the center buccal of each of the sites and immediately placed into a 10% neutral buffered formalin solution, then sent for histological evaluation. The sites were then re-augmented using Bio-Oss at both sites, and Ossix membranes were adapted to the ridges, as previously described. The patient’s interim removable partial denture was verified to ensure that any excessive force on the graft site was avoided.
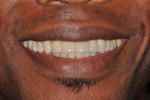
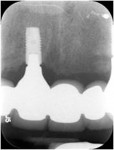
Six months after the second GBR, dental implants were placed at the Nos. 8 and 11 positions. Provisional restorations were delivered 4 months after the date of implant placement. Over the course of the next 12 months the patient was followed closely, the provisional restorations were modified, and additional implant and prosthodontic treatment was completed (Figure 14). The subgingival contours created from the provisionals were replicated by making an impression of the provisionals and fabricating custom impression copings made with acrylic resin, as seen in Figure 15. An open-tray final impression was made for the pre-maxilla, and an abutment-level impression was simultaneously made for the posterior implants, as shown in Figure 16. The final ceramo-metal FPD (x-8i-x-x-11i) prosthesis was delivered, as seen clinically in Figure 17 and Figure 18 and radiologically in Figure 19 and Figure 20.
Results
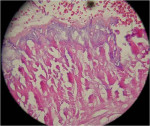
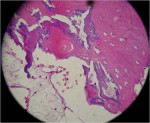
Upon reentry from the initial GBR, clinically it was noted that the ridge on the Regenaform site was more consolidated and similar to normal native bone, while the ridge on the Bio-Oss side was not as well consolidated, and it was easy to scrape off granular fragments. The Bio-Oss site felt less dense than and not similar to native bone. In the mid-crestal region of the Bio-Oss ridge, there appeared to be an area of soft tissue encasing Bio-Oss particles, which was removed. It is possible that this site was Bio-Oss particles that were still in the process of regeneration but at 6 months had not yet reached a point where they resembled bone. The Regenaform site exhibited a gain in width of 1.5 mm (from the original width of 3.6 mm at the most coronal aspect and tapered to a thinner horizontal dimension apically) and a vertical fill of the concave defect of about 2 mm. The Bio-Oss site had increased in width from 3 mm to 6.5 mm; however, the height at the apex of the crestal concavity lost an additional 1 mm. The cores were stained with hematoxylin and eosin (H&E) and examined as step serial sections under the magnifications of 5, 10, 40, and 63x. The two samples were described by the oral pathologist as “similar.” Histologically, the Regenaform sample was described as “composed of a background of fibro-vascular connective tissue showing islands of non-vital bone and trabeculae of viable new bone, attached to the graft portions and scattered osteoblasts noted in the neo-formed bone.” No osteoclasts were identified, nor was any inflammation present, as seen in Figure 21. The description of the Bio-Oss core was similar, but lacked description of a fibro-vascular connective tissue, as seen in Figure 22.
Discussion
In situations with severe alveolar ridge atrophy, bone augmentation is usually required for proper dental implant placement and adequate hard-tissue support for the surrounding soft tissues.16 Autogenous block grafting from either an intraoral or extraoral site can be used for ridge augmentation; however, possible complications from harvesting an autogenous graft must be considered.17 The degrees of morbidity associated with autogenous block graft procedures may deter patients from selecting that form of ridge augmentation.
The objective for this patient was to gain adequate bone volume for the placement of dental implants using two different forms of graft material—one with a slow resorption time and the other in a gelatin matrix to provide dimensional stability. In this case, Bio-Oss generated more horizontal bone width, whereas Regenaform gained slightly less width but showed a 2-mm gain in vertical height. It has been shown that xenograft materials can remain in augmented sites after a period of 6 months and do not undergo further resorption.18 This has been reported by histological analysis in humans showing amounts of xenograft material present with no signs of osteoclasts or resorptive activity at 6 months after placement of the xenograft material.18 With this in mind, the authors would expect that the xenograft material will provide dimensionally stable ridge contours over a long period of time.
At the 6-month re-entry there was a 2-mm to 3-mm component removed from the mid-crest of the Bio-Oss graft site that was still soft and osteoid-like. Whether or not that would have mineralized into viable bone is unknown. The second augmentation procedure was done utilizing a bovine xenograft particulate in order to ensure a slower turnover of graft material to maintain the contours of the buccal ridge for long-term implant esthetics.
There is no known concrete evidence of whether or not healing of bone grafts or long-term success rates of dental implants are different in patients exhibiting previous aggressive periodontitis versus patients with no history of periodontitis.
A slow resorbing collagen membrane was used to limit complications of potential early membrane exposure and the particular collagen membrane utilized at the time had substantial evidence supporting its success in bone regeneration.
Only two dental implants were placed for the five-unit FPD to limit having two adjacent implants, which can compromise esthetics of the final restoration. The No. 7 site was chosen as a pontic versus an implant site for two reasons: 1) the esthetics of a pontic are superior to that of a dental implant site in FPD restorations; and 2) there was a greater volume of bone width at the No. 8 and 11 sites compared to other sites to ensure that there was 1 mm to 2 mm of bone buccal to the implant platforms.
Conclusion
The results of this case report are inconclusive to demonstrate the superiority of one of the two graft materials over the other in reconstructing a severely atrophied maxilla. Both sites ultimately required a second GBR procedure prior to implant placement. It was speculated that adequate space maintenance—which is critical to the success of GBR—may not have been achieved during the initial procedures. Alternative measures to provide enhanced space maintenance during the initial GBR procedure include the adjunctive use of either longer and/or additional stainless-steel tenting screws, titanium-reinforced expanded polytetrafluoroethylene (ePTFE) membranes, titanium mesh, or utilizing a block graft technique.
While it may be controversial whether or not the dental implants could have been placed at the time of the second GBR, the clinicians chose to perform the second GBR and delay implant placement. Since adequate hard tissues were regenerated in this case, soft-tissue augmentation was not indicated and would have led to increased scar tissue formation.
Additionally, it could be speculated that the use of a removable provisional may have reduced the outcome of the GBR if it applied pressure to the healing ridge. An alternative would have been to provide the patient with a fixed provisional restoration, although this would have required full-coverage restorations for two additional teeth. When performing GBR in areas with marked alveolar resorption, it is imperative that patients be informed prior to beginning treatment that more than one grafting procedure may be indicated. This is especially true when treating situations in the esthetic region.
References
1. Buser D, Kula D, Belser U, et al. Localized ridge augmentation using guided bone regeneration. I. Surgical procedure in the maxilla. Int J Periodontics Restorative Dent. 1993;13(1):29-45.
2. Buser D, Dula K, Belser UC, et al. Localized ridge augmentation using guided bone regeneration. II. Surgical procedure in the mandible. Int J Periodontics Restorative Dent. 1995;15(1):10-29.
3. Pieri F, Corinaldesi G, Fini M, et al. Alveolar ridge augmentation with titanium mesh and a combination of autogenous bone and anorganic bovine bone: a 2-year prospective study. J Periodontol. 2008;79(1):2093-2103.
4. Melcher AH. On the repair potential of periodontal tissues. J Periodontol. 1976;47(5):256-260.
5. Urist M. Bone: Formation by autoinduction. Science. 1965;150(3698):893-899.
6. Buser D, Braegger U, Lang NP, et al. Regeneration and enlargement of jaw bone using guided tissue regeneration. Clin Oral Implants Res. 1990;1(1):22-32.
7. Simion M, Dahlin C, Trisi P, et al. Qualitative and quantitative comparative study on different filling materials used in tissue regeneration: a controlled clinical study. Int J Periodontics Restorative Dent. 1994;14(3):198-215.
8. Becker W, Becker B, Caffesse R. A comparison of demineralized freeze-dried bone and autologenous bone to induce bone formation in human extraction sockets. J Periodontol. 1994;65(12):1128-1133.
9. Cammack GV, Nevins M, Clem DS, et al. Histologic evaluation of mineralized and demineralized freeze-dried bone allograft for ridge and sinus augmentations. Int J Periodontics Restorative Dent. 2005;25(3):231-237.
10. Vance GS, Greenwell H, Miller RL, et al. Comparison of an allograft in an experimental putty carrier and a bovine-derived xenograft used in ridge preservation: a clinical and histologic study in humans. Int J Oral Maxillofac Implants. 2004;19(4): 491-497.
11. Chiapasco M, Zaniboni M, Boisco M. Augmentation procedures for the rehabilitation of deficient edentulous ridges with oral implants. Clin Oral Impl Res. 2006;17 suppl 2:136-159.
12. Esposito M, Grusovin MG, Worthington HV, et al. Interventions for replacing missing teeth: bone augmentation techniques for dental implant treatment. Cochrane Database Syst Rev. 2006;25(1):CD003607.
13. Armitage GC. Development of a classifications system for periodontal diseases and conditions. Ann Periodontol. 1999;4(1):1-6.
14. Nevins ML, Camelo M, Nevins M, et al. Minimally invasive alveolar ridge augmentation procedure (tunneling technique) using rhPDGF-BB in combination with three matrices: a case series. Int J Periodontics Restorative Dent. 2009;29(4):371-383.
15. Sethi A, Kaus T. Maxillary ridge expansion with simultaneous implant placement: 5-year results of an ongoing clinicay study. Int J Oral Maxillofac Implants. 2000;15(4):491-499.
16. Buser D, Halbritter S, Hart C, et al. Early implant placement with simultaneous guided bone regeneration following single-tooth extraction in the esthetic zone: 12-month results of a prospective study with 20 consecutive patients. J Periodontol. 2009;80(1):152-162.
17. Hunt DR, Jovanovic SA. Autogenous bone harvesting: a chin graft technique for particulate and monocortical bone blocks. Int J Periodontics Restorative Dent. 1999;19(2):165-173.
18. Molly L, Vandromme H, Quirynen M, et al. Bone formation following implantation of bone biomaterials into extraction sites. J Periodontol. 2008;79(6):1108-1115.
Disclosure
The authors received grant support from The Health Professions Division of Nova Southeastern University College of Dental Medicine, Fort Lauderdale, Florida.
About the Authors
Maria Hernandez, DDS
Associate Director
Postgraduate Periodontics
Section of Periodontology
Nova Southeastern University College of Dental Medicine
Fort Lauderdale, Florida
Gregory A. Pette, DMD, MS
Resident
Post-Graduate Periodontics and Master of Science Candidate
Section of Periodontology
Nova Southeastern University College of Dental Medicine
Fort Lauderdale, Florida
Andre Grenier, DMD
Private Practice
Periodontics
Fort Lauderdale, Florida
Carlos Villanueva, DDS
Resident in Post-Graduate Prosthodontics
Section of Prosthodontics
Nova Southeastern University College of Dental Medicine
Fort Lauderdale, Florida
Enrique Lask, DDS
Adjunct Faculty
Section of Prosthodontics
Nova Southeastern University College of Dental Medicine
Fort Lauderdale, Florida;
Private Practice
Mexico City, Mexico
William Parker, DDS
Chair
Section of Periodontology and Director Postgraduate Periodontology
Nova Southeastern University College of Dental Medicine
Fort Lauderdale, Florida