Delayed Implant Procedure Using Deproteinized Bovine Bone Mineral: A Report of 109 Consecutive Cases
Wayne A. Aldredge, DMD; and Richard Nejat, DDS
Abstract
PURPOSE: The purpose of this consecutive case series was to analyze the success rate of endosseous implants placed into deproteinized bovine bone mineral (DBBM) maxillary and mandibular grafted extraction sites during a 48-month period.
METHODS: During a 48-month period, 69 consecutive patients presented with 109 nonrestorable teeth. The teeth were extracted and the sites grafted with DBBM, and the extraction sockets were allowed to heal for an average of 6 months. Following healing, implants were placed in the grafted sites. Osseointegration was assessed based on a lack of mobility and a radiographic evaluation.
RESULTS: Implants were placed in all of the 109 grafted sites with no graft failures. No implant was mobile at uncovering. All 109 implants are currently loaded and have been in function for 6 months to 65 months with an average of 2 years for a 100% cumulative success rate. Marginal soft-tissue levels remain stable.
CONCLUSION: The delayed implant approach using DBBM is a predictable treatment modality, resulting in long-term implant stability and predictable esthetic results.
Ideal placement of endosseous implants is critical to ensure proper esthetics, facilitate oral hygiene, and provide adequate function. The long-term maintenance of hard- and soft-tissue levels surrounding endosseous implants is dependent on proper buccolingual, mesiodistal, and apicocoronal implant position.1,2 The long-term marginal soft-tissue contours and maintenance of the interdental papillae around implants are dependent on the underlying thickness and level of bone, as well as on sufficient amounts of gingival connective tissue.1,2 In a study of 2,667 implants, Spray et al determined that when 1.8 mm of bone was present buccal to the implant at placement, vertical bone levels were stable.3 As a general rule, ideal bone thickness buccal to the implant should be at least 2 mm.1,2
Post-extraction ridge resorption is well documented in the literature. Human clinical and experimental studies and literature reviews have attempted to quantify and explain the process of ridge resorption following tooth extraction.4-8 Post-extraction bone loss is often reported as unpredictable.6,8 Generally, bone loss following tooth removal is greater on the buccal than on the lingual.6 Schropp et al reported that horizontal ridge resorption following tooth extraction amounted to a mean of approximately 50%, corresponding to 6.1 mm (range 2.7 mm to 12.2 mm) for all regions of the alveolus at 12 months.5 The authors further stated that two-thirds of the horizontal bone loss occurred in the first 3 months after tooth extraction. Nevins et al demonstrated that nongrafted sockets with thin biotypes lost more than 20% of the buccal plate 71% of the time and more than 5 mm of vertical height.6
The incidence of osseointegration of immediately placed implants is high and considered predictable.8 However, recent human and experimental studies have demonstrated conclusively that the placement of an implant fixture into a fresh extraction socket does not prevent bone loss.9-11 Horizontal bone resorption occurring after placement of an implant into fresh extraction sockets was found to be similar to that seen in extraction sockets without implant placement. Botticelli et al demonstrated that when implants were placed into fresh extraction sockets, horizontal resorption (from the exterior of the buccal wall to the implant surface) approached 56% (1.9 mm) on the buccal and 30% (0.8 mm) on the lingual.9 In experimental studies, the marginal gap present at the time of immediate implant placement had resolved at reentry.9,10 However, some vertical bone loss was evident. Vertical bone loss or insufficient bone buccal to the implant surface often leads to esthetically compromised results, including a lower position of the marginal gingival, black interdental triangles, or buccal soft-tissue irregularities that may require additional corrective surgery.
Various surgical guidelines and decision trees have been presented relating to patient phenotype (biotype); the presence or absence and type of defect; implant positioning; biologic width; expected response to extraction; and type of surgical therapy planned.1,6,8,12,13 Several publications have concluded that socket grafting of various forms as well as the chosen surgical approach can minimize post-extraction bone loss.4,6 Iasella et al compared nongrafted extraction sites to extraction sites grafted with freeze-dried bone allograft.4 Grafted sites lost 1.2 mm of horizontal ridge width versus 2.7 mm of horizontal-ridge width loss in nongrafted extraction sites. In addition, the grafted sites gained 1.3 mm of bone height while nongrafted sites lost 0.9 mm of height. In another study, Nevins et al examined grafted versus nongrafted extraction sites limited to maxillary anterior teeth with thin buccal plates and prominent roots using split-thickness flaps and complete soft-tissue closure.6 In this study, grafted sites lost "less than 20% of the buccal plate in 15 of 19 test sites (79%)." In the context of these delayed implant placement studies, placing a dental implant in an ideal position with sufficient surrounding bone can be predictable. The current consecutive case series is a long-term retrospective analysis of delayed implant placement and functional loading after extraction socket grafting with deproteinized bovine bone mineral (DBBM).
Materials and Methods
In this consecutive case series study, 69 patients were treated in a private practice setting by two board-certified periodontists. All patients presented with nonrestorable teeth secondary to multiple causes, eg, fractured roots, failed endodontic procedures, advanced caries, severe periodontal involvement, or combinations of the above. With the exception of severe infection, all extraction sockets were immediately grafted with DBBM. If severe infection was present (one patient, two teeth), the teeth were extracted, the area was thoroughly debrided, and DBBM was placed following adequate soft-tissue healing. In most cases, tooth extraction was performed without flap reflection. After healing, implants were placed using either a full-thickness flap or a flapless/punch technique.
Surgical Protocol
Patients were prescribed the following: ibuprofen 800 mg every 8 hours starting the night prior to surgery; amoxicillin 500 mg, 2 grams 1 hour before surgery; amoxicillin 500 mg every 8 hours postsurgery; and chlorhexidine rinse twice daily starting the night prior to surgery. After adequate local anesthesia, the teeth were removed atraumatically to preserve the hard and soft tissues. Often, molar teeth were sectioned and treated as single-rooted teeth. Flaps were not elevated, except in rare cases requiring increased access, such as the removal of a deeply positioned root tip. Granulomatous tissue was debrided thoroughly, and the areas were irrigated with sterile saline. Cancellous DBBM (Bio-Oss®, Osteohealth, www.osteohealth.com) 0.25-mm to 1-mm particle size was placed into a sterile dappen dish and hydrated with sterile saline according to the manufacturer’s instructions.
After debridement, dehiscence or fenestration defects were treated from the internal aspect of the extraction site, using a resorbable collagen membrane (Ossix®, BIOMET 3i, www.biomet.com) to contain the graft material and prevent the in-growth of soft tissue. DBBM was then placed into the extraction socket to the level of the alveolar crest. A resorbable collagen hemostatic sponge (CollaPlug®, Zimmer Dental, www.zimmerdental.com; Helistat®, Integra LifeSciences, www.integra-ls.com) was used to form a coronal cover over the socket. To secure the collagen sponge into place, 4-0 chromic gut sutures were used. Occasionally, a resorbable collagen membrane was used as the coronal cover in lieu of the hemostatic sponge. After extraction socket grafting, all cases were provisionalized, most often with a transitional partial denture. Select cases were provisionalized with a cemented provisional restoration or an Essix™ retainer (DENTSPLY Raintree Essix, www.essix.com).
The extraction sockets were allowed to heal for an average of 6 months. After healing, a total of 109 implants were placed. No graft failures occurred, and implants were placed in all 109 grafted sites. A total of 58 implants were placed with flap reflection while 51 implants were placed using a flapless/punch technique. Both one-stage and two-stage endosseous implants were used, representing various implant designs. Implant diameters ranged from 3.3 mm to 5 mm with implant length ranging from 10 mm to 15 mm, based on restorative considerations and locations within the alveolus. All implants placed were rough-surface design. Of the 109 implants inserted, 67 were placed in the maxilla and 42 in the mandible. A total of 33 implants were put in smokers (11 patients) and 76 implants in nonsmokers (58 patients).
Results
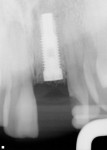
Of the 109 implants placed in 69 patients during a 4-year period, no implant failures were observed following fixture insertion and subsequent prosthetic restoration. No implant was mobile at uncovering, and all of the implants placed were restored. At the point of data compilation, all implants remained fully loaded and in function, resulting in a 100% survival rate (Table 1). As a group, the implants have been in function and loaded for an average of 2 years, ranging from 6 months to 65 months. Regardless of implant type, location within the alveolus, or smoking status, 100% of the implants are in function currently (Table 1 and Figure 1, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, Figure 8 and Figure 9).
Discussion
The primary goal of extraction socket grafting is the preservation of alveolar ridge anatomy to facilitate the placement of implants in ideal 3-dimensional positions required for highly functional and esthetic implant-supported restorations. Preservation of the ridge includes considerations for both vertical and horizontal bone dimensions. Numerous materials have been studied for the grafting of extraction sockets both prior to the placement of dental implants and for ridge preservation under traditional fixed restoration pontics.12 DBBM has been used in surgical procedures since 1989, and its use is well documented in the literature. Implant success rates in sinuses grafted with DBBM are similar to those grafted with autogenous bone.14-17 However, relatively few studies can be found in the literature examining DBBM use in extraction socket grafting compared with other indications, especially in regard to long-term implant success rates.
Nevins et al reported that the likelihood of preserving socket anatomy and subsequently placing a dental implant "under optimal conditions" in especially challenging thin-biotype patients was far greater when grafted with xenograft (DBBM).6 In examining histologic and histomorphometric data following extraction socket grafting with DBBM, Artzi et al reported a variable range of vital bone formation from the apical to the coronal third of the socket between 63% and 15%, with a mean of 46%.18 Carmagnola et al reported 14% bone formation with approximately 40% of the residual graft particles in contact with newly formed bone.19 Norton et al reported 26% bone formation with approximately 34% of the graft particles in contact with newly formed bone.20 Both Carmagnola et al and Norton et al were able to place implants into those sites, with Norton et al achieving a "baseline" survival rate of 97%.
Numerous histologic reports have demonstrated the presence of residual xenograft particles at time points from 6 months to 9 years after initial grafting.21-25 The clinical significance of this slow or nonresorptive phenomenon is an important topic of discussion in relation to osseointegration and long-term implant survival.23,24 Valentini et al demonstrated that sinus augmentation with Bio-Oss results in similar amounts of vital bone as seen in the nonaugmented, native adjacent alveolar ridge.22 In that study, histologic examination demonstrated the presence of DBBM graft particles in close contact with newly formed bone, the latter maturing over time. Because vital bone formation was no different than the adjacent nongrafted sites, the presence of the remaining graft particles resulted in less marrow space and a higher bone mineral density when compared with the surrounding native bone with its higher percentage of marrow. Importantly, histologic findings indicate only newly formed bone and not residual graft particles are in contact with the implant surface, and bone-to-implant contact is similar to that found in nongrafted native bone.21,23,25
The present study found that no implant failed in 109 consecutive extraction sockets grafted with DBBM during a loading period of 6 months to 65 months, resulting in a 100% implant survival rate. In the context of this case series, the well-documented slow resorption rate of DBBM does not appear to negatively impact implant osseointegration. Histologic findings suggesting that implants do not tend to be in contact with residual graft particles may explain, at least in part, the high implant success rate in the present case series. In addition, Hallman et al suggested that the increase in overall density of the augmented site resulting from the combination of the residual DBBM particles and bone prevents micromotion during osseointegration.21 In a histologic analysis, Esposito et al demonstrated fibrous encapsulation of implant fixtures associated with micromotion during critical stages of osseointegration.26 The overall bone mineral density is defined as the newly formed composite of bone plus the residual mineralized graft particles. Boyne described this as increasing bone density and made observations similar to Hallman regarding implant osseointegration, adding that the resulting increased bone mineral aids in the maintenance of bone even under conventional prostheses.27 In a 9-month histologic study of extraction socket grafting, Artzi et al found an average vital bone formation of 46% with approximately 31% remaining DBBM particles, resulting in a mean bone mineral density of 77%.18
The definition of implant success is especially critical when considering the esthetic zone. In addition to implant osseointegration, additional criteria have been discussed recently as guidelines for the future.28 The ability to preserve the 3-D socket volume is vital for optimal implant positioning. Based on the results of this case series for maintenance of hard- and soft-tissue levels, the goal of dimensional volume preservation was achieved. Because implant success rates are not negatively impacted by a slowly resorbing grafting material, eg, DBBM, a slowly resorbing biomaterial may be highly desirable for esthetic considerations. Cornellini et al29 demonstrated that the marginal position of gingiva surrounding immediate implants augmented with a xenograft and a resorbable membrane was significantly higher than at sites where a resorbable membrane was used alone. They demonstrated these differences at buccal, lingual, and proximal sites adjacent to implant fixtures grafted with xenograft. In another study, Hallman et al21 documented that xenografts effectively maintain the elevated sinus area for extended periods, resulting in a dimensionally stable graft. Boyne made similar conclusions in the edentulous patient over a 9-year period.27 Dimensional stability resulting from a slowly resorbing graft material, particularly in the esthetic zone, may be ideal for maintenance of alveolar volume and proper overlying soft-tissue contours.
Conclusion
In the present case series, no implant failures were observed during an implant-loading period of 6 months to 65 months. The delayed-implant approach using DBBM proved to be a predictable procedure for implant success. Additional studies are needed to evaluate potential dimensional changes post-extraction and post-implantation/restoration among other grafting materials.
About the Authors
Wayne A. Aldredge, DMD
Clinical Assistant Professor
Department of Periodontics and Implantology
Stony Brook University
Long Island, New York
Private Practice
Nutley and Holmdel, New Jersey
Richard Nejat, DDS
Clinical Assistant Professor
New York University Department of Periodontics and Implant Dentistry
Private Practice
New York Cityand Nutley, New Jersey
References
1. Buser D, Martin W, Belser UC. Optimizing esthetics for implant restorations in the anterior maxilla: anatomic and surgical considerations. Int J Oral Maxillofac Implants. 2004; 19(suppl):43-61.
2. Grunder U, Gracis S, Capelli M. Influence of the 3-D bone-to-implant relationship on esthetics. Int J Periodontics Restorative Dent. 2005;25(2):113-119.
3. Spray JR, Black CG, Morris HF, Ochi S. The influence of bone thickness on facial marginal bone response: stage 1 placement through stage 2 uncovering. Ann Periodontol. 2000;5(1):119-128.
4. Iasella JM, Greenwell H, Miller RL, et al. Ridge preservation with freeze-dried bone allograft and a collagen membrane compared to extraction alone for implant site development: a clinical and histologic study in humans. J Periodontol. 2003;74(7):990-999.
5. Schropp L, Wenzel A, Kostopoulos L, Karring T. Bone healing and soft tissue contour changes following single-tooth extraction: a clinical and radiographic 12-month prospective study. Int J Periodontics Restorative Dent. 2003;23(4):313-323.
6. Nevins M, Camelo M, De Paoli S, et al. A study of the fate of the buccal wall of extraction sockets of teeth with prominent roots. Int J Periodontics Restorative Dent. 2006;26(1):19-29.
7. Araújo MG, Lindhe J. Dimensional ridge alterations following tooth extraction. An experimental study in the dog. J Clin Periodontol. 2005;32(2):212-218.
8. Fugazzotto PA. Treatment options following single-rooted tooth removal: a literature review and proposed hierarchy of treatment selection. J Periodontol. 2005;76(5):821-831.
9. Botticelli D, Berglundh T, Lindhe J. Hard-tissue alterations following immediate implant placement in extraction sites. J Clin Periodontol. 2004;31(10):820-828.
10. Araújo MG, Wennström JL, Lindhe J. Modeling of the buccal and lingual bone walls of fresh extraction sites following implant installation. Clin Oral Implants Res. 2006;17(6):606-614.
11. Araújo MG, Sukekava F, Wennström JL, Lindhe J. Tissue modeling following implant placement in fresh extraction sockets. Clin Oral Implants Res. 2006;17(6):615-624.
12. McAllister BS, Haghighat K. Bone augmentation techniques. J Periodontol. 2007;78(3):377-396.
13. Sclar AG. Strategies for management of single-tooth extraction sites in aesthetic implant therapy. J Oral Maxillofac Surg. 2004;62(9 suppl 2):90-105.
14. Wallace SS, Froum SJ. Effect of maxillary sinus augmentation on the survival of endosseous dental implants. A systematic review. Ann Periodontal. 2003;8(1):328-343.
15. Del Fabbro M, Testori T, Francetti L, Weinstein R. Systematic review of survival rates for implants placed in the grafted maxillary sinus. Int J Periodontics Restorative Dent. 2004;24(6):565-577.
16. Esposito M, Grusovin MG, Coulthard P, Worthington HV. The efficacy of various bone augmentation procedures for dental implants: a Cochrane systematic review of randomized controlled clinical trials. Int J Oral Maxillofac Implants. 2006;21(5):696-710.
17. Sclar AG. Ridge preservation for optimum esthetics and function: the Bio-Col technique. Compend Contin Educ Dent. 1999;6(1):1-11.
18. Artzi Z, Tal H, Dayan D. Porous bovine bone mineral in healing of human extraction sockets. Part 1: histomorphometric evaluations at 9 months. J Periodontol. 2000;71(6):1015-1023.
19. Carmagnola D, Adriaens P, Berglundh T. Healing of human extraction sockets filled with Bio-Oss. Clin Oral Implants Res. 2003;14(2):137-143.
20. Norton MR, Odell EW, Thompson ID, Cook RJ. Efficacy of bovine bone mineral for alveolar augmentation: a human histologic study. Clin Oral Implants Res. 2003;14(6):775-783.
21. Hallman M, Sennerby L, Lundgren S. A clinical and histologic evaluation of implant integration in the posterior maxilla after sinus floor augmentation with autogenous bone, bovine hydroxyapatite, or a 20:80 mixture. Int J Oral Maxillofac Implants. 2002;17(5):635-643.
22. Valentini P, Abensur D, Wenz B, et al. Sinus grafting with porous bone mineral (Bio-Oss) for implant placement: a 5-year study on 15 patients. Int J Periodontics Restorative Dent. 2000;20(3):245-253.
23. Scarano A, Pecora G, Piattelli M, Piattelli A. Osseintegration in a sinus augmented with bovine porous bone mineral: histological results in an implant retrieved 4 years after insertion. A case report. J Periodontol. 2004;75(8):1161-1166.
24. Traini T, Valentini P, Iezzi G, Piattelli A. A histologic and histomorphometric evaluation of anorganic bovine bone retrieved 9 years after a sinus augmentation procedure. J Periodontol. 2007;78(5):955-961.
25. Iezzi G, Scarano A, Mangano C, et al. Histologic results from a human implant retrieved due to fracture 5 years after insertion in a sinus augmented with anorganic bovine bone. J Periodontol. 2008;79(1):192-198.
26. Esposito M, Thomsen P, Ericson LE, Lekholm U. Histopathologic observations on early oral implant failures. Int J Oral Maxillofac Implants. 1999;14(6):798-810.
27. Boyne PJ. Osseous Reconstruction of the Maxilla and the Mandible: Surgical Techniques Using Titanium Mesh and Bone Mineral. Hanover Park, IL: Quintessence Publishing Co; 1997.
28. Belser U, Buser D, Higginbottom F. Consensus statements and recommended clinical procedures regarding esthetics in implant dentistry. Int J Oral Maxillfac Implants. 2004;(19 suppl):73-74.
29. Cornelini R, Cangini F, Martuscelli G, Wennström J. Deproteinized bovine bone and biodegradable barrier membranes to support healing following immediate placement of transmucosal implants: a short-term controlled clinical trial. Int J Periodontics Restorative Dent. 2004;24(6):555-563.