Zirconia: Material Background and Clinical Application
Russell Giordano, DMD, CAGS, DMSc; and Carlos Eduardo Sabrosa, CD, MSD, DScD
Abstract
Zirconia is the oxidized form of zirconium and can exist in several phases, depending on the temperature. In dental ceramics, zirconia can inhibit crack growth and prevent catastrophic failure. The following case report demonstrates the use of zirconia as a framework material. Overall, zirconia has proven to be a strong and reliable framework material. In combination with CAD/CAM, this material allows for the fabrication of esthetic all-ceramic restorations in all areas of the mouth.
Ceramic materials can successfully replicate the esthetic qualities of natural teeth. However, despite their strength under compression, they are brittle, have limited tensile strength, and do not exhibit any significant deformation before failure as what can occur with metals.1-3 Clinically, this means that ceramics generally fail catastrophically, with no warning. Zirconia-based ceramic materials have properties that lend themselves to use in the mouth; they can inhibit crack growth and prevent catastrophic failure. Zirconia-based ceramics have been used in other industries for many years; dental applications present unique problems in that custom parts must be made and should look like teeth.4 Computer-aided design/computer-aided manufacturing (CAD/CAM) has enabled materials to be used that ordinarily cannot be fabricated. One of the most important of these materials is yttria partially stabilized tetragonal zirconia. Zirconia (ZrO2) is the oxidized form of zirconium (Zr) just as alumina (Al2O3) is an oxide of aluminum (Al).
Zirconia exists in three major phases: monoclinic, tetragonal, and cubic. Monoclinic is room-temperature stable.5 Above 1170°C, zirconia transforms into the tetragonal intermediate phase; at 2370°C, the material changes into a cubic phase. In pure zirconia ceramics, the cubic-to-monoclinic phase transformation occurs during cooling with approximately 5% volumetric expansion (causes cracks), which may then fracture zirconia at room temperature. Therefore, biomedical and structural/functional applications of zirconia typically do not use pure zirconia. The addition of other ceramic components may alter the presence and stability of these phases at room temperature. Zirconia may exist primarily in the tetragonal phase at room temperature by adding components such as calcia (CaO), magnesia (MgO), yttria (Y2O3), and ceria (CeO2). If the right amount of component is added, then a fully stabilized cubic phase (cubic zirconia) can be created. The addition of smaller amounts (3 weight% to 5 weight%) produces a partially stabilized zirconia. Although stabilized at room temperature, the tetragonal zirconia phase may change under stress to monoclinic with a subsequent 3% volumetric increase.6 This dimensional change diverts energy from the crack and can stop the crack. This is called transformation toughening (Figure 1).7 This helps resist catastrophic failure—even though a crack may exist in the material, the phase change prevents it from proceeding throughout the restoration. Yttria-stabilized tetragonal zirconia polycrystal (Y-TZP) has the potential to be a "universal" ceramic restorative material in that it has sufficient properties to withstand stresses in all regions of the mouth, as well as the ability to support multiple-unit restorations.
Most restorations containing zirconia frameworks are fabricated by machining a porous or partially fired block of zirconia. The framework is milled oversized and then fired at approximately 1500°C to fully densify the zirconia, producing a translucent material and strength values from 900 MPa to 1200 MPa.8 Most blocks have barcodes that tell the computer the density in order to properly mill the framework.
The use of zirconia for dental restorations in 2009 was about 50% of all restorations made completely from a ceramic. As the popularity has increased, so too has the number of suppliers of machinable blocks. There are many "brand name" manufacturers with reliable products, but there are also many questionable blocks being sold. Differences among zirconia ceramics can occur with the level of zirconia purity, grain size, trace elements, and stabilizing compounds. There are differences in the zirconia starting powders and the processing of those powders into blocks. Consequently, clinical outcomes, such as marginal fit, translucency, strength, and long-term stability, can be significantly affected. Zirconia may degrade over time.9,10 However, accelerated aging tests in steam and boiling water of two manufacturers' zirconia products (Lava™, 3M ESPE, https://www.3mespe.com; Vita In-Ceram YZ, Vita Zahnfabrik, https://www.vita-zahnfabrik.com) found no degradation.11 Other zirconia block materials may show degradation. A batch of hip implants manufactured by Saint Gobains failed catastrophically after a few years in patients' bodies.12 This may have been due to a problem with the exact composition of the zirconia in regard to the amount of yttria and alumina, leading to uncontrolled phase transformation. To help prevent instability of the crystal phases, zirconia formulations should have a small amount of alumina.
Some manufacturers use liquids with various metal salts to shade the frameworks. The colorant is applied after the framework is milled but before firing to full density. The liquid fills the pores, and the metal salt is deposited. After firing, the zirconia assumes the desired shade. Alternatively, the blocks may be fabricated, which are intrinsically shaded at the level of the starting powder. The shaded zirconia can help minimize the need for any opaque or liner and enhance the esthetic result.
One concern that is the subject of research and controversy is the issue of chipping of the veneer porcelain. In tests of zirconia itself, the veneer porcelain does not seem to fail. However, there have been numerous reports of veneer chipping. In several published studies, the veneer chipping rate was about 15% after 3 to 5 years.13-15 In a clinical trial conducted by Clinical Research Associates, up to 60% of the hand-veneered restorations demonstrated veneer chipping after 1 year and about 20% of pressed veneers showed chipping.16 It is important to note that low-fusing porcelains were used in that study. A review of various clinical trials on zirconia restorations showed a wide range of chipping at 1 to 5 years—from 5% to 25% for low-fusing porcelains such as Cercon® Ceram (DeguDent, https://www.cercon-smart-ceramics.com), Lava Ceram (3M ESPE), and IPS e.max® Ceram (Ivoclar Vivadent, https://www.ivoclarvivadent.com). Chipping was up to 54% after 1 year for Triceram® (Esprident, https://www.esprident.com) porcelain on dense zirconia.17 However, others report minor amounts of chipping problems.18,19 It is important to note that there appears to be a correlation with the firing cycle and peak firing temperature of the veneer. Laboratory studies have shown that low-fusing porcelains may be less resistant to cracking than high-fusing porcelains and the porcelain density is lower.20-22 Zirconia is a good thermal insulator, which prevents heat transfer to the veneer porcelain, preventing it from becoming fully dense if fast-fired and/or fired at a low temperature. Also, fast cooling may create stress in the porcelain, which could lead to cracking. In addition, handling of zirconia may affect the stability of the veneer. Any surface adjustment—grinding, sandblasting, and even polishing—can change the phase on the surface of the zirconia and may affect the stability and strength of the zirconia, as well as the veneer porcelain.20-22
Another area of interest is the use of zirconia as a full-contour restoration. Although zirconia has a fine microstructure, there are varied reports as to the abrasion properties and no clinical studies on the longevity of full-contour zirconia and the wear of opposing dentition. Some research shows that polished zirconia is abrasive to the opposing dentition and the glaze protects the natural tooth for some time until the glaze wears through.23 Other studies in progress demonstrate that polished zirconia is "wear-kind." Ongoing clinical trials are expected to provide a more definitive answer.
Zirconia frameworks are most often luted to the tooth structure using glass-ionomer cement. However, several primers may be used to create a bond to zirconia—Ivoclar, Kuraray, and Bisco all provide bonding products to zirconia. Glass ionomers bond weakly or not at all.24 Zirconia bonding might improve retention to the tooth structure, and these resin cements are not soluble. There again is one controversy with respect to sandblasting the zirconia. Most manufacturers recommend against the use of sandblasting on the veneer surface but allow a low-pressure (25 to 50 psi, 2/3 bar) small-particle alumina (25 μm to 50 μm) sandblasting of the interior as the final step before cementing the restoration. This may help clean the internal surface and provide some mechanical retention for nonbonded cement. Some studies have demonstrated a potential problem in sandblasting the internal surface with respect to crack growth, while others have shown an improvement in properties.11,25
The following case demonstrates the use of zirconia as a framework material. Overall, zirconia has proven to be a strong and reliable framework material. In combination with CAD/CAM, this material allows for the fabrication of esthetic all-ceramic restorations in all areas of the mouth. Zirconia is highly biocompatible, and the mechanical properties may allow for decreased coping thickness and connector sizes. Failure in all-ceramic bridges commonly occurs at the connectors, requiring larger connectors, particularly for lower-strength frameworks. However, both connector size and coping thickness of zirconia frameworks are the smallest of any all-ceramic restoration, which further enhances this material's esthetic qualities.
Case Report
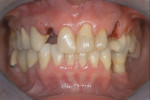
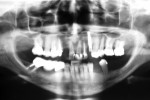
The patient presented in 2006 with teeth Nos. 7 and 11 fractured. At that time, the patient was pregnant, and in consultation with her physician, minimal intervention was suggested. Very little bone remained in the facial aspect of tooth No. 7, and it was determined that teeth Nos. 7 and 11 should be extracted (Figure 2). The existing crown on tooth No. 6 was removed, and a splinted restoration with a cantilever to restore tooth No. 7 was performed. An implant was placed immediately after the extraction of tooth No. 11 without augmentation but with immediate function (Figure 3).
The patient returned for definitive treatment approximately 1 year later. A diagnostic wax-up was used to aid in space analysis and treatment planning. Treatment was planned in a phased approach and begun on the maxillary arch. The wax-up was then used to fabricate a provisional restoration with a bis-acryl resin (Protemp™ 4, 3M ESPE) (Figure 4). After initial periodontal therapy, all endodontically treated teeth were evaluated, re-treated as necessary, and restored with fiber posts (RelyX™ Fiber Post, 3M ESPE), self-adhesive cement (RelyX Unicem Aplicap™, 3M ESPE), and composite resin (Filtek™ Silorane, 3M ESPE). All cavities and undermined dentin were also filled with Filtek Silorane.
After periodontal and endodontic evaluations, teeth Nos. 3 and 15 were extracted and teeth Nos. 3 and 7 were replaced with implants. Tooth No. 15 was not replaced because no replacement of its opposing tooth was planned.
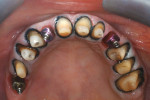
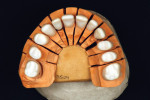
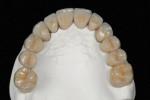
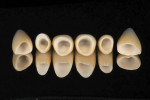
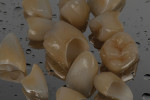
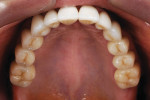
After occlusal stabilization and soft-tissue healing, the teeth and implants were impressed with a double-cord technique using retraction cords impregnated with aluminum sulfate (Gingi-Aid® 00 e2, Gingi-Pak, https://www.gingi-pak.com) (Figure 5) and polyether (Impregum™ Soft, 3M ESPE). Two casts were obtained. The first pour was sectioned and ditched whereas the second pour was retained for checking contact points in the future (Figure 6). The maxillary working and mandibular diagnostic casts were mounted in a semi-adjustable articulator, and a full-contour wax-up was fabricated. Implant wax-ups were reduced to fabricate a custom-made zirconia abutment with the Lava system (3M ESPE). A new wax-up was fabricated over the abutments to fabricate copings with a uniform 1.2-mm reduction to provide space for ceramic application. All waxed substructures were scanned with the double-scanning technique, and zirconia substructures were also fabricated with the Lava system (Figure 7). After laboratory inspection, a compatible ceramic system (IPS e.max Ceram) was used to achieve esthetics (Figure 8, Figure 9, Figure 10 and Figure 11).
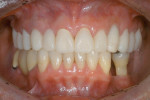
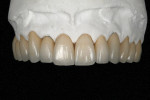
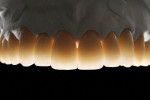
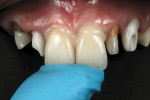
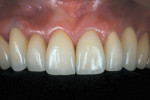
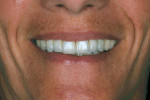
All restorations were tried and adjusted and then glazed. The internal surfaces of the restorations were treated with aluminum oxide with a 50-μm particle size at a two-bar pressure. After sandblasting, the internal surface was steam-cleaned and dried and a zirconia resin bonding primer (Kuraray, https://www.kuraray.com) was applied to the surface. The teeth were cleaned first with alcohol and then sodium hypochlorite with an abundant air-water spray and dried. A translucent shade of a self-adhesive composite resin cement (RelyX Unicem) was used for the final cementation (Figure 12). The patient was reevaluated after 2 weeks to observe soft-tissue healing (Figure 13, Figure 14 and Figure 15). The final esthetics had been obtained (Figure 16 and Figure 17). Hygiene treatment is being performed every 6 months.
Conclusion
Although ceramics are brittle materials, the phase transformation of zirconia under stress makes this ceramic highly resistant to fracture. Zirconia may be used to fabricate highly esthetic restorations anywhere in the mouth.
References
1. Proebster L, Diehl J. Slip-casting alumina ceramics for crown and bridge restorations. Quintessence Int. 1992;23(1):25-31.
2. Piconi C, Burger W, Richter HG, et al. Y-TZP ceramics for artificial joint replacements. Biomaterials.1998;19(16):1489-1494.
3. Lawn BR, Deng Y, Thompson VP. Use of contact testing in the characterization and design of all-ceramic crownlike layer structures: a review. J Prosthet Dent. 2001;86(5):495-510.
4. Green DJ, Hannink R, Swain MV. Transformation Toughening of Ceramics. Boca Raton, FL: CRC Press; 1989.
5. Piconi C, Maccauro G. Zirconia as a ceramic material. Biomaterials. 1999;20(1):1-25.
6. Giordano R. A comparison of all-ceramic restorative systems: part 2. Gen Dent. 2000;48(1):38-40, 43-45.
7. Lange FF. Transformation toughening: part 3, experimental observations in the ZrO2-Y2O3 system. J Mat Sci.1982;17:240-246.
8. Guazzato M, Albakry M, Ringer SP, et al. Strength, fracture toughness and microstructure of a selection of all-ceramic materials. Part II. Zirconia-based dental ceramics. Dent Mater.2004;20(5):449-456.
9. Chevalier J, Cales B, Drouin M. Low-temperature aging of Y-TZP ceramics. J Am Ceram Soc. 1999;82(8):2150-2154.
10. Lawson S. Environmental degradation of zirconia ceramics. J Eur Ceram Soc.1995;15(6):485-502.
11. Papanagiotou HP, Morgano SM, Giordano RA, et al. In vitro evaluation of low-temperature aging effects and finishing procedures on the flexural strength and structural stability of Y-TZP dental ceramics. J Prosthet Dent. 2006;96(3):154-164.
12. Clarke IC, Manaka M, Green SM, et al. Current status of zirconia used in total hip implants. J Bone Joint Surg Am.2003;85-A suppl:73-84.
13. Sailer I, Fèher A, Filser F, et al. Prospective clinical study of zirconia posterior fixed partial dentures: 3-year follow-up. Quintessence Int. 2006;37(9):685-693.
14. Vult von Steyern P, Carlson P, Nilner K. All-ceramic fixed partial dentures designed according to the DC-Zirkon technique. A 2-year clinical study. J Oral Rehabil.2005;32(3):180-187.
15. Sailer I, Fehér A, Filser F, et al. Five-year clinical results of zirconia frameworks for posterior fixed partial dentures. Int J Prosthodont.2007;20(4):383-388.
16. Christensen R, Eriksson KA, Ploeger BJ. Clinical performance of PFM, zirconia, and alumina three-unit posterior prostheses [abstract 1566]. J Dent Res.2008;87(spec iss B).
17. Al-Amleh B, Lyons K, Swain M. Clinical trials in zirconia: a systematic review. J Oral Rehabil.2010;37(8):641-652.
18. Blatz M, Mante F, Chiche G, et al. Clinical survival of posterior zirconia crowns in private practice [abstract 2110]. J Dent Res.2010;89(spec iss B).
19. Nathanson D, Chu S, Yamamoto H, et al. Performance of zirconia based crowns and FPDs in prosthodontic practice [abstract 2115]. J Dent Res. 2010;89(spec iss B).
20. Fahmi M, Pober R, Giordano R. Effect of surface treatment on porcelain bond strength to zirconia [abstract 1571]. J Dent Res. 2007;86(spec iss A).
21. Fahmi M, Pober R, Giordano R. Thermal shock of porcelain veneered zirconia with various surface treatment [abstract 092]. J Dent Res. 2008;87(spec iss B).
22. Arrejaie A, Giordano R, Pober R. Mechanical properties of Y-TZP/ porcelain interface with multiple surface treatments [abstract 1571]. J Dent Res. 2010;89(spec iss A).
23. Shah S, Michelson C, Beck P, et al. Wear of enamel on polished and glazed zirconia [abstract 227]. J Dent Res.2010;89(spec iss A).
24. Bulot D, Sadan A, Burgess JO, et al. Bond strength of a self-adhesive universal resin cement to Lava Zirconia after two surface treatments [abstract 578]. J Dent Res. 2005;84(spec iss A).
25. Zhang Y, Lawn BR, Malament KA, et al. Damage accumulation and fatigue life of particle-abraded ceramics. Int J Prosthodont. 2006;19(5):442-448.
About the Authors
Russell Giordano, DMD, CAGS, DMSc
Associate Professor
Director of Biomaterials
Boston University Goldman School of Dental Medicine
Boston, Massachusetts
Carlos Eduardo Sabrosa, CD, MSD, DScD
Associate Professor
Universidade Estadual do Rio de Janeiro
Rio de Janeiro, Brazil
Private Practice
Rio de Janeiro, Brazil