Etiology and Prevention of Acid Erosion
David Bartlett, BDS, PhD, MRD, FDS, RCS
Abstract
Acid erosion is the chemical effect of dietary or gastric acids on enamel and dentine. Unlike dental caries, which is a bacterially mediated condition, acid erosion normally is combined with physical forms of attrition and abrasion. The clinical appearance of acid erosion in the early stages is seen characteristically as hollowed-out lesions on occlusal surfaces and on smooth surfaces as a subtle change in the tooth contour. As the condition progresses, the lesions coalesce and form widespread dentin exposure and coronal destruction. Dietary acids that are present in beverages and fruits potentially can cause acid erosion. However, dietary habits involving frequent consumption of acids are believed to be important determinants in the risk of developing clinical signs of acid erosion. Prevention using fluoride toothpastes, dietary modifications, and calcium-based products probably have a significant clinical effect in reducing the risk of developing acid erosion.
The definitions of the components of tooth wear are recognized internationally.1 Erosion is the loss of enamel and dentin caused by the action of acids unrelated to bacterial action; attrition is the loss of tooth structure caused by tooth-to-tooth contact; and abrasion is the loss of tooth structure caused by physical wear other than teeth. However, interpretations differ regarding how important each component is when analyzing a patient’s pattern of wear.2 Traditionally in Europe, the impact of acid erosion has been recognized as probably being the most important component.3 However, the impact of abrasion, particularly toothbrushing, often is emphasized in North America. Recently, the role of acids in tooth wear has become more acknowledged partly because of research and mounting clinical evidence.
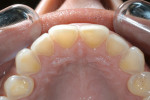
It is challenging, based solely on the appearance of teeth, to separate the various components of tooth wear. In some instances, the flattened incisal edges of incisors strongly suggest that attrition is a dominant factor. However, when there is cupping associated with the flattened surfaces, other factors will also be active, most notably erosion (Figure 1). The hollowed-out appearance of these types of lesions, in theory, also could be attributed to abrasion, but the force needed to cause this degree of wear is unlikely to occur on the occlusal surfaces. Therefore, many professionals consider acid erosion the predominant reason for cup-shaped lesions.
The knowledge that acid was responsible for erosion was first recognized in patients who had the eating disorders anorexia nervosa or bulimia nervosa.4 The appearance of periomylosis on the palatal surfaces of upper incisors has been acknowledged widely to be caused by acid regurgitation or vomiting (Figure 2). Periomylosis is the historical term used to describe the pattern of erosive tooth wear seen on the palatal surfaces of the upper incisor teeth. It was first recognized in patients with eating disorders and believed to be caused by vomiting and regurgitated gastric acids in the mouth.5 Eventually, clinical and laboratory evidence suggested the pattern similar to a palatal veneer preparation was caused by the low-pH gastric acid being either vomited or regurgitated.4 Some laboratory evidence indicates that the shape of the lesion is modified in some way by the abrasive action of the tongue.6 Therefore, a hollowed-out or scooped-out appearance strongly suggests active acid erosion.
Appearance
Probably the most easily recognizable component of tooth wear is attrition. The action of bruxing opposing tooth surfaces causes the flattening of occlusal or incisal surfaces. The physical force of the massester and temporalis muscles has been predicted to approach 911 N.7 Theoretically, applying this level of force to restorations increases the risk of fracture or chipping, particularly in those with porcelain bonded to metal. The site of attrition, by definition, will be along the incisal or occluding surfaces, whereas smooth surface lesions, in theory, can be caused by abrasion or erosion.
The etiology of cervical wear lesions has caused more debate than any other aspect of tooth wear. Early literature proposed excessive force applied by toothbrushing was a cause of cervical wear.8 However, clinical and laboratory investigative work performed simulanteously contradicted this clinical opinion and indicated that brushing of enamel caused insignificant wear. Despite this research, many clinicians continue to believe that toothbrushing is a major cause of cervical lesions. It is possible that excessive force applied while toothbrushing can cause cervical wear, but this role has not been fully investigated. More recent research suggests that the cause of cervical wear is probably a combination of erosion and abrasion.8,9 It has been known for some time that mechanical wear—in this case tooth brushing—when combined with acid disolution is synergistic.10 Recent laboratory and clinical evidence indicates that brushing within 10 to 20 minutes following a dietary acid increases wear to a signifantly greater extent than pure brushing.11
Dentin sensitivity, which often is present in association with cervical wear, has been suggested to be a direct clinical outcome of erosion.12 These researchers suggested that sensitivity is a clinical sign of acid erosion because it does not occur in patients with poor oral hygiene. Acid erosion, by definition, is caused by the action of dietary or gastric acids dissolving tooth surfaces and is quite unlike dental caries, which is a subsurface effect related to the metabolism of plaque microorganisms. This clinical evidence suggests that dentine sensitivity is a feature of clean tooth surfaces. The action of acids removes the smear layer, which opens dentinal tubules to the oral environment and increases the risk of developing sensitivity.
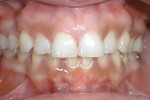
It is probably safe to conclude that the impact of normal toothbrushing in the absence of acid is unlikely to cause tooth wear.8 However, excessive pressure or brushing soon after acid consumption may increase the risk of wear significantly.11 Smooth surface lesions are seen on the facial surfaces of teeth and often noted adjacent to cervical wear lesions. On occlusal surfaces, erosive lesions appear to be hollowed-out, strongly suggesting the involvement of acids in the formation (Figure 3).3
Etiology
The evidence that dietary or gastric acids cause erosion has been extensively investigated, and its role has been subject to a number of reviews.3,13,14 The role of dietary acids is considered by many researchers probably to be the most common cause of acid erosion and may account for the finding that as much as 70% of patients have at least one tooth with wear.15,16 Although gastric acids also cause acid erosion, the prevelance of regurgitation, eating disorders, and alcoholism is insufficient to account for the prevalence data on erosion.17 A common misconception is that all acidic foods and drinks carry a high risk of acid erosion. Laboratory-based research strongly implicates that dietary acids have the potential to cause acid erosion.14,16,18 However, the pH and titrability of an acid (the volume of alkali needed for neutralization) are both important indicators of erosion risk.19 Common dietary acids and their erosion risk are shown in Table 1.20 The erosion risk from a strong acid, such as grapefruit, is higher than that of a cola drink. Although the pH is similar in both, the amount of alkali needed to neutralize the fruit means it is a stronger acid. The titrability of an acid is believed to give a stonger indication of the risk of acid erosion whereas pH—a measure of the amount of hydrogen ions in a solution—is imprecise. For example, carbonated water has a relatively low pH, approximately 4 to 5. Its erosive potential is low whereas a cola drink with the same pH would have a much greater erosive potential because of the much higher concentration of hydrogen ions.21 Therefore, in the risk assessment of someone with acid erosion, the type of acidic beverage or fruit is important to identify.
The other major risk factor with dietary acids is the frequency of consumption. Clinical evidence has suggested that the pattern of acid neutralization is unlike the pattern observed in dental caries.22 The typical drop and slow recovery seen with acid production in dental plaque is not replicated in acid erosion. Dietary acids cause an immediate drop in oral pH, commensurate with the pH of the food or drink. Then, after saliva has neutralized it, the pH returns to physiologic pH 7 within a couple of minutes. These findings, along with clinical evidence, suggest that it is the frequency of acid consumption that is important. Any dietary habit, such as holding or swilling acids before swallowng, holding fruit against the incisors, or continual snacking is indicative of an increased risk of developing erosion.22,23
The concept of abfraction has been suggested by some clinicians as a possible cause of cervical wear. The laboratory support for this hypothetical cause is varied, with some laboratory investigations supporting the view24 and others not.25 Evidence from finite-element analysis also supports this view, but the studies are computer models, which may not provide sufficient complexity to model teeth.26 Also, no clinical studies support this hypothesis.27 Despite the temptation that abfraction is a real phenomena, the evidence does not support it.28
Prevention
The epidemiologic evidence suggests that tooth wear and acid erosion are common in adults,29 children,30 and adolescents.31 Because of its widespread impact, prevention16 of tooth wear is important. The role of fluoride delivered in a toothpaste has been investigated and observed to have a beneficial effect.11,32 Most evidence on fluoride’s protective effect has been based on laboratory investigations.32-34 The fluoride ions appear to increase the microhardness of enamel and so improving its resistance to acid dissolution.35 There are some in situ studies that support the laboratory evidence concluding fluoride is protective, but no clinical interventional research to date.35,36 Also, some clinical and laboratory evidence suggests toothbrushing immediately after consuming acids increases the risk of erosion and abrasion.36 Conjunctive application of a topical mouthrinse containing fluoride also may be beneficial. Although there is no research available to support frequent application of flouride, findings from caries studies suggest that mouthrinsing at times other than during normal brushing improves the availability of fluoride and hardens tooth surfaces. In high-risk patients, there may be some justification also to consider brushing teeth at least 15 to 20 minutes after consuming an acidic beverage or fruit.36 The synergistic action of the acid and brushing increases the potential for developing erosive wear lesions.
Dietary modification is probably important in prevention. Clinical studies suggest that frequent consumption of acidic foods increases the risk of developing erosive tooth wear.23,37,38 However, the strength of this evidence continues to be based on association rather than causation. It remains challenging to properly investigate the role of dietary acids in interventional studies because of ethical issues. Therefore, in a similar way to understanding the role of refined carbohydrates in dental caries, the evidence must be based on assimilating clinical knowledge, case reports, and laboratory investigations.39 By amassing this data, the current consensus is that frequent consumption of carbohydrates causes caries. The strength of the evidence for a similar statement in acid erosion is unavailable; however, increasing clinical knowledge strongly indicates that behavior modification can be effective in preventing erosive tooth wear.21
Early laboratory evidence implies that calcium-based products have the potential to prevent erosive tooth wear.40 The mode of action is not fully understood, but these products containing casein and calcium products might be protective. The casein phosphopeptide-amorphous calcium phosphate (CPP-ACP) complex is believed to maintain a sufficiently high concentration of calcium and phosphate ions to promote enamel remineralization. Further, laboratory studies indicate CPP-ACP may be absorbed into the salivary pellicle41 and plaque42 and so provide a rich calcium reservoir, improving the potential for remineralization.
Another method to improve the resistance of teeth to acid erosion is application of dentin-bonding agents or fissure sealants to worn and eroded teeth. Clinical evidence implies that 3- to 6-month protection is afforded by a single application of a dentin-bonding agent.43 There also may be some benefit to the application of fissure sealants to teeth to provide a barrier to erosive wear, which is more resilient than a dentin-bonding agent.
Summary
The role of acids in tooth wear is recognized as an important reason why teeth wear. Increasingly, dietary habits rather than the quantity of acidic food and drinks is being recognized as the most important factor in increasing the risk. Fluoride and dentin-bonding agents appear to have the potential to increase the teeth’s ability to resist acidic attack, but behavioral changes are likely to be as effective in prevention.
References
1. Smith BGN. Toothwear: aetiology and diagnosis. Dent Update. 1989;16:204-213.
2. Bartlett DW, Phillips KM, Smith BGN. A difference of perspective—the North American and European interpretations of tooth wear. Int J Prosthodont. 1999;12(5):401-408.
3. Bartlett DW, Smith BGN. Definition, classification and clinical assessment of attrition, erosion and abrasion of enamel and dentine. In: Addy M, Embery G, Edgar WM, eds. Tooth Wear and Sensitivity–Clinical Advances in Restorative Dentistry. 1st ed. London, England: Martin Dunitz; 2000:87-93.
4. Bartlett D. Intrinsic causes of erosion. Monogr Oral Sci. 2006;20: 119-139.
5. Holst JJ, Lange F. Perimylolysis: a contribution towards the genesis of tooth wasting from non-mechanical causes. Acta Odontol Scand. 1939;1: 36-48.
6. Gregg T, Mace S, West NX, et al. A study in vitro of the abrasive effect of the tongue on enamel and dentine softened by acid erosion. Caries Res. 2004;38(6): 557-560.
7. Waltimo A, Nyström M, Könönen M. Bite force and dentofacial morphology in men with severe dental attrition. Scand J Dent Res. 1994;102(2): 92-96.
8. Mannerberg F. Appearance of tooth surface as observed in shadowed replicas in various age groups, in long-term studies, after toothbrushing, in cases of erosion and after exposure to citrus fruit juice. Odontologisk Revy. 1960;11(6 suppl):70-86.
9. Addy M, Absi EG, Adams D. Dentine hypersensitivity. The effects in vitro of acids and dietary substances on root planed and burred dentine. J Clin Periodontol. 2000;14(5):274-279.
10. Davis WB, Winter PJ. The effect of abrasion on enamel and dentine after exposure to dietary acid. Br Dent J. 1980;148:253-256.
11. Attin T, Siegel S, Buchalla W, et al. Brushing abrasion of softened and remineralised dentin: an in situ study. Caries Res.2004;38(1):62-66.
12. Absi EG, Addy M, Adams D. Dentine hypersensitivity—the effect of toothbrushing and dietary acids on dentine in vitro: an SEM study. J Oral Rehabil. 1992;19(2): 101-110.
13. Zero DT. Etiology of dental erosion—extrinsic factors. Eur J Oral Sci. 1996;104(2 pt 2):162-177.
14. Lussi A, Kohler N, Zero D, et al. A comparison of the erosive potential of different beverages in primary and permanent teeth using an in vitro model. Eur J Oral Sci. 2000;108(2):110-114.
15. Harding MA, Whelton H, O’Mullane DM, et al. Dental erosion in 5-year-old Irish school children and associated factors: a pilot study. Community Dent Health. 2003;20(3):165-170.
16. Milosevic A, Young PJ, Lennon MA. The prevalence of tooth wear in 14-year-old school children in Liverpool. Community Dent Health. 1994;11(2): 83-86.
17. Bartlett DW, Evans DF, Smith BGN. The relationship between gastro-oesophageal reflux disease and dental erosion. J Oral Rehabil. 1996;23(5): 289-297.
18. Ganss C, Klimek J, Giese K. Dental erosion in children and adolescents—a cross-sectional and longitudinal investigation using study models. Community Dent Oral Epidemiol. 2001;29(4):264-271.
19. Grenby TH. Method of assessing erosion and erosive potential. Eur J Oral Sci. 1996;104(2 pt 2):207-214.
20. Approximate pH of foods and food products. US Food and Drug Administration Center for Food Safety and Applied nutrition. https://vm.cfsan.fda.gov/~comm/lacf-phs.html. Published April 2007. Updated September 2008. Accessed: May 2009.
21. Bartlett DW. The role of erosion in tooth wear: aetiology, prevention and management. Int Dent J. 2005;55(4 suppl 1): 277-284.
22. Moazzez R, Smith BGN, Bartlett DW. Oral pH and drinking habit during ingestion of a carbonated drink in a group of adolescents with dental erosion. J Dent. 2000;28(6):395-397.
23. Millward A, Shaw L, Harrington E, et al. Continuous monitoring of salivary flow rate and pH at the surface of the dentition following consumption of acidic beverages. Caries Res. 1997;31:44-49.
24. Staninec M, Nalla RK, Hilton JF, et al. Dentin erosion simulation by cantilever beam fatigue and pH change. J Dent Res. 2005;84(4): 371-375.
25. Litonjua LA, Andreana S, Bush PJ, et al. Wedged cervical lesions produced by toothbrushing. Am J Dent. 2004;17(4):237-240.
26. Rees JS, Hammadeh M, Jagger DC. Abfraction lesion formation in maxillary incisors, canines and premolars: a finite element study. Eur J Oral Sci. 2003;111(2):149-154.
27. Estafan A, Furnari PC, Goldstein G, et al. In vivo correlation of noncarious cervical lesions and occlusal wear. J Prosthet Dent. 2005;93(3):221-226.
28. Bartlett DW, Shah P. A critical review of non-carious cervical (wear) lesions and the role of abfraction, erosion, and abrasion. J Dent Res. 2006;85(4): 306-312.
29. Van’t Spijker A, Rodrigues JM, Kreulen CM, et al. Prevalence of tooth wear in adults. Int J Prosthodont. 2009;22(1): 35-42.
30. Dugmore CR, Rock WP. The prevalence of tooth erosion in 12-year-old children. Br Dent J. 2004;196(5): 279-282.
31. Bartlett DW, Coward PY, Nikkah C, et al. The prevalence of tooth wear in a cluster sample of adolescent schoolchildren and its relationship with potential explanatory factors. Br Dent J. 1998;184(3): 125-129.
32. Bartlett DW, Smith BG, Wilson RF. Comparison of the effect of fluoride and non-fluoride toothpaste on tooth wear in vitro and the influence of enamel fluoride concentration and hardness of enamel. Br Dent J. 1994;176(9): 346-348.
33. Ganss C, Klimek J, Schäffer U, et al. Effectiveness of two fluoridation measures on erosion progression in human enamel and dentine in vitro. Caries Res. 2001;35(5):325-330.
34. Attin T, Zirkel C, Hellwig E. Brushing abrasion of eroded dentin after application of sodium fluoride solutions. Caries Res. 1998;32(5): 344-350.
35. Zero DT, Hara AT, Kelly SA, et al. Evaluation of a desensitizing test dentifrice using an in situ erosion remineralization model. J Clin Dent. 2006;17(4): 112-116.
36. Wiegand A, Egert S, Attin T. Toothbrushing before or after an acidic challenge to minimize tooth wear? An in situ/ex vivo study. Am J Dent. 2008;21(1):13-16.
37. Lussi A, Schaffner M, Holtz P, et al. Dental erosion in a population of Swiss adults. Community Dent Oral Epidemiol. 1991;19(5):286-290.
38. Lussi A, Jaeggi T, Jaeggi-Schärer S. Prediction of the erosive potential of some beverages. Caries Res. 1995;29(5):349-354.
39. Edgar WM, O’Mullane DM, eds. Saliva and Oral Health. 2nd ed. London, England: British Dental Journal; 1996.
40. Ranjitkar S, Rodriguez JM, Kaidonis JA, et al. The effect of casein phosphopeptide-amorphous calcium phosphate on erosive enamel and dentine wear by toothbrush abrasion. J Dent. 2009;37(4):250-254.
41. Schüpbach P, Neeser JR, Golliard M, et al. Incorporation of caseinoglycomacropeptide and caseinophosphopeptide into the salivary pellicle inhibits adherence of mutans streptococci. J Dent Res. 1996;75(10): 1779-1788.
42. Rose RK. Effects of an anticariogenic casein phosphopeptide on calcium diffusion in streptococcal model dental plaques. Arch Oral Biol. 2000;45(7):569-575.
43. Sundaram G, Wilson R, Watson T, et al. Clinical measurement of palatal tooth wear following coating by a resin sealing system. Oper Dent. 2007;32(6): 539-543.
About the Author
David Bartlett, BDS, PhD, MRD, FDS, RCS
Professor
Department of Prosthodontics
Kings College London Dental Institute
London Bridge
London, United Kingdom