Leaky Gum Syndrome and the Mouth-Body Connection: The Impact of Oral Fluid Diagnostics
Hans-Dieter John, MSD
Despite huge improvements in dentistry over the past several decades, nearly half of all American adults still suffer from periodontal disease, the most common cause of tooth loss in adults.1 Most patients do not know they have gum disease, nor are they aware of the consequences and systemic issues that can manifest as a result of the silent inflammatory process occurring in the mouth and spreading to the rest of the body. This process by which the oral epithelium of the gums becomes damaged and unable to optimally function as a barrier against oral pathogens and inflammatory cytokines is known as leaky gum syndrome. It results in the spread of those pathogens and virulence factors from the mouth to other parts of the body.2 This process of tissue breakdown can actively occur for years before any type of periodontal disease is diagnosed by a clinician. In fact, it has been shown that, on average, periodontitis is only diagnosed 10 to 16 years after its onset, which typically is the 22- to 28-year-old age range.3
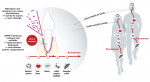
Leaky gum syndrome occurs when the gums are in a state of active periodontal degeneration. During this time, catalytic enzymes known as matrix metalloproteinases (MMPs) break down collagen, the main component comprising the periodontium, as well as occludin, which is a key molecule functioning as part of the tight junctions between epithelial cells. The structural integrity of the periodontium and that of the tight junctions is important for preventing pathogenic bacteria and viruses from entering the bloodstream through the highly vascular gum tissue. If this barrier deteriorates, these pathogens, along with other cytokines released as a part of the localized oral inflammation, can more easily enter the bloodstream and spread to the rest of the body (Figure 1). This can increase the overall inflammatory load within the body and directly cause complications in other areas. Bacteraemia of oral pathogens has been linked to atherosclerotic cardiovascular disease, diabetes, neurodegenerative diseases, rheumatoid arthritis, pancreatic cancer, colorectal cancer, liver health, and even preterm birth.4
Oral Biomarkers: Closing the Diagnostic Gap
One reason for the prevalence of periodontal disease is that current diagnostic tools are only able to assess what has occurred in the past, allowing clinicians to measure the amount of tissue that has already been destroyed. However, oral biomarkers have shown promising results in identifying the active processes occurring at the onset and early-stage progression of periodontal disease before clinical signs are visible. Oral biomarkers can detect the tissue breakdown process in real-time, identifying the disease process occurring on the molecular level and predictively foreseeing the rate of future bone loss.
The American Dental Association Science & Research Institute recently evaluated some of the most promising technologies for the future of periodontal diagnostics and noted the scientific evidence for the biochemical assay kit utilizing the biomarker active matrix metalloproteinase-8 (aMMP-8), a major mediator of tissue destruction in periodontal and peri-implant disease.5 MMP-8 is an enzyme released as part of the immune system's response to harmful bacteria in the mouth. When in its activated form, aMMP-8 functions as a collagenase to break down collagen and the tight junction protein, occludin, that connects epithelial cells.6
Clinical Relevance of Prognostic Oral Biomarkers
Successful prevention and treatment of periodontal and peri-implant disease depends largely on early detection, patient awareness, and patient adherence to treatment recommendations. Preventive measures personalized to each patient's unique immune system can stop the biochemically initiated breakdown of periodontal tissues. Compared to probing depth or radiographic diagnostics, which are post-destructive measurements, prognostic biomarkers such as aMMP-8 can open the door to truly personalized prevention and maintenance strategies.7 For example, 94% of implants that have elevated levels of aMMP-8 at prosthesis delivery go on to exhibit a high rate of bone loss in the following 6 to 24 months.8
aMMP-8 levels measured in the oral fluid can indicate the extent to which a patient's gums are "leaking" on the molecular level. As these patients are identified, clinicians and hygienists are alerted to which seemingly healthy patients need more care in order to halt this process of tissue destruction. By implementing the appropriate periodontal or hygiene therapy (along with patient compliance at home) the aMMP-8 levels in the mouth can be reduced and the tissue breakdown process halted.9
aMMP-8 has been identified as one of the most promising markers for what dental service organization Pacific Dental Services (PDS) calls the Mouth-Body Connection®, because it elucidates what is happening in the periodontal tissue, which can be a mirror of the condition of one's overall health. This diagnostic tool has even been shown to assess the "mouth-body connection" by identifying patients who may be prediabetic/diabetic and those with cardiovascular problems. Elevated levels of aMMP-8 are able to assist in the identification of patients with prediabetes or diabetes with an accuracy of 79.9%.10 In addition, patients who had experienced an acute myocardial infarction showed significantly higher levels of aMMP-8 found in their oral fluid.11
Chairside Periodontal Diagnostics in the US
While chairside aMMP-8 assessment has been used in Europe for years, this screening technology is still relatively new to US clinicians. PDS has begun implementing the technology in US dental practices to support patient education regarding the "mouth-body connection" and periodontal disease. In a Colgate Health Network Webinar on March 31 of this year, experts in periodontal disease as well as the PDS dental group shared their knowledge about the usage of oral fluid diagnostics in daily practice. This webinar can be viewed for free at the following web address: https://bit.ly/colgateoralhealthnetwork.
As early screening tools are implemented as a standard of care in dentistry, clinicians can expect to see a reduction in the prevalence of periodontal and peri-implant disease through targeted preventive care and increased patient awareness.
About the Author
Hans-Dieter John, MSD
Private Practice, Düsseldorf, Germany
References
1. Eke PI, Dye BA, Wei L, et al. Prevalence of periodontitis in adults in the United States: 2009 and 2010. J Dent Res. 2012;91(10):914-920.
2. Park DY, Park JY, Lee D, et al. Leaky gum: the revisited origin of systemic diseases. Cells. 2022;11(7):1079.
3. Thorbert-Mros S, Cassel B, Berglundh T. Age of onset of disease in subjects with severe periodontitis: a 9- to 34-year retrospective study. J Clin Periodontol. 2017;44(8):778-783.
4. Hajishengallis G, Chavakis T. Local and systemic mechanisms linking periodontal disease and inflammatory comorbidities. Nat Rev Immunol. 2021;21(7):426-440.
5. Ko TJ, Byrd KM, Kim SA. The chairside periodontal diagnostic toolkit: past, present, and future. Diagnostics (Basel). 2021;11(6):932.
6. Schubert-Unkmeir A, Konrad C, Slanina H, et al. Neisseria meningitidis induces brain microvascular endothelial cell detachment from the matrix and cleavage of occludin: a role for MMP-8. PLoS Pathog. 2010;6(4):e1000874.
7. Al-Majid A, Alassiri S, Rathnayake N, et al. Matrix metalloproteinase-8 as an inflammatory and prevention biomarker in periodontal and peri-implant diseases. Int J Dent. 2018;2018:7891323.
8. Guarnieri R, Zanza A, D'Angelo M, et al. Correlation between peri-implant marginal bone loss progression and peri-implant sulcular fluid levels of metalloproteinase-8. J Pers Med. 2022;12(1):58.
9. Alassiri S, Parnanen P, Rathnayake N, et al. The ability of quantitative, specific, and sensitive point-of-care/chair-side oral fluid immunotests for aMMP-8 to detect periodontal and peri-implant diseases. Dis Markers. 2018;2018:1306396.
10. Grigoriadis A, Sorsa T, Räisänen I, et al. "Prediabetes/diabetes can be screened at the dental office by a low-cost and fast chair-side/point-of-care aMMP-8 immunotest. Diagnostics (Basel). 2019;9(4):151.
11. Ehlers V, Willershausen I, Kraft J, et al. Gingival crevicular fluid MMP-8-concentrations in patients after acute myocardial infarction. Head Face Med. 2011;7:1.